Abstract
The presence of pharmaceutical residues in aquatic systems poses a growing environmental concern, necessitating the development of sustainable and efficient sensing materials. In this study, haloalkaliphilic archaea were employed as green biocatalysts for the synthesis of magnetite (Fe₃O₄) nanoparticles and their application in the electrochemical detection of ibuprofen under saline conditions. Two archaeal strains produced Fe₃O₄ nanoparticles with well-defined inverse spinel crystal structures, nanoscale dimensions, and superparamagnetic behavior, while exhibiting distinct surface chemistry and magnetic properties. Strain-dependent differences in crystallinity and surface functionalization were found to strongly influence electrochemical performance. RA5-derived nanoparticles exhibited higher crystallinity and enhanced charge-transfer efficiency, achieving a sensitivity of 2.105 µA mg L⁻¹ and a limit of detection of 0.927 mg L⁻¹. In contrast, A6-derived nanoparticles featured a richer organic surface corona that promoted analyte adsorption, resulting in a sensitivity of 2.2186 µA mg L⁻¹ and a detection limit of 1.05 mg L⁻¹. Both modified electrodes enabled reliable ibuprofen detection over a wide linear concentration range of 0–100 mg L⁻¹. These results demonstrate that haloalkaliphilic archaea can function as sustainable nano-factories for producing functional magnetic nanomaterials, offering an eco-friendly strategy for electrochemical monitoring of pharmaceutical contaminants in complex aquatic environments.

Similar content being viewed by others
Introduction
The rapid global expansion of the pharmaceutical industry, driven by rising healthcare demands and an aging population, has led to the abundant presence of non-steroidal anti-inflammatory drugs (NSAIDs) in aquatic ecosystems worldwide1,2. Among these compounds, Ibuprofen (IBP) has surfaced as one of the most frequently detected pharmaceutical residues in wastewater treatment plant effluents, surface waters, and even groundwater reserves. These substances are categorized as “emerging contaminants” because they are not currently regulated by standard water quality legislation, yet they pose a significant threat to environmental health and human safety3,4. Because NSAIDs are designed to be biologically active even at extremely low concentrations, their continuous discharge into the environment creates a state of “pseudo-persistence.” This long-term exposure can lead to chronic toxicity in aquatic organisms, affecting their reproductive cycles, and potentially leading to bioaccumulation within the food chain, which may eventually impact human health through the consumption of contaminated water or fish5,6. Traditional analytical techniques for the detection and quantification of pharmaceutical pollutants such as High-Performance Liquid Chromatography (HPLC), Gas Chromatography-Mass Spectrometry (GC-MS), and Capillary Electrophoresis are the gold standards for precision and sensitivity. However, these methodologies are often hindered by significant drawbacks, including high operational costs, the requirement for sophisticated and expensive instrumentation, and the need for highly trained personnel. Furthermore, these methods typically involve labor-intensive and time-consuming sample pre-treatment and extraction steps that require the use of large volumes of toxic organic solvents, which can lead to secondary environmental pollution. Consequently, there is an urgent and growing need for the development of highly sensitive, cost-effective, and sustainable sensing platforms capable of rapid, on-site, and real-time environmental monitoring7,8,9. Electrochemical sensors have emerged as a frontrunner in this field due to their remarkable sensitivity, selectivity, and rapid response times. They offer a portable and miniaturized alternative for decentralized testing, allowing for the detection of trace-level pollutants in complex matrices10,11. The performance and efficiency of an electrochemical sensor are fundamentally governed by the architecture and chemical nature of the electrode surface. By modifying electrodes with advanced nanomaterials, researchers can significantly enhance the active surface area, facilitate more efficient charge transfer, and provide an abundance of active sites for specific analyte interaction12. Among the various classes of nanomaterials explored, magnetic nanoparticles (MNPs), particularly magnetite (Fe3O4), are highly valued in the development of electrochemical platforms. Their unique superparamagnetic properties, inherent biocompatibility, and high surface-to-volume ratio make them excellent candidates for enhancing signal transduction and improving the overall limit of detection for pharmaceutical residues. Despite these advantages, the conventional chemical and physical synthesis of Fe3O4 nanoparticles such as chemical co-precipitation, hydrothermal synthesis, and microemulsion techniques often presents environmental and economic challenges. These processes frequently involve the use of toxic reducing agents, hazardous precursors, and high energy consumption, which challenges the principles of green chemistry13,14,15. This has led to a paradigm shift toward “green synthesis” or biogenic fabrication, an approach that leverages the metabolic diversity of microorganisms to produce functional nanomaterials under ambient conditions. Biogenic synthesis offers a sustainable, eco-friendly, and cost-efficient alternative, where biological entities serve as both the reducing agents for metal ions and the capping agents that stabilize the resulting nanoparticles16. In this context, haloalkaliphilic archaea microorganisms that thrive in extreme environments characterized by high salinity and alkaline pH represent a largely untapped resource as biological “factories.” These extremophiles possess specialized metabolic pathways and robust. Unlike mesophilic bacteria or fungi, haloalkaliphilic archaea utilize specialized acidic proteins and exopolysaccharides that remain stable and functional in extreme salinity and high pH, providing a robust extracellular template for Fe3O4 nucleation17. While magnetotactic bacteria rely on complex intracellular magnetosomes and fungi often require slow, enzyme-mediated reduction, these archaea leverage their naturally alkaline environment to facilitate the direct co-precipitation of Fe2+ and Fe3+ ions. Furthermore, the high ionic strength of the archaeal habitat inherently regulates the Debye length and colloidal stability, offering unique physicochemical control over nanoparticle morphology and size distribution that is unattainable in traditional freshwater or neutral-pH microbial systems18,19. These biomolecules act as natural templates, controlling the nucleation, growth, and size of the nanoparticles17. Crucially, biogenic nanoparticles are often characterized by a “bio-organic corona” a coating of biological molecules that not only prevents aggregation but also provides functional groups for further chemical interaction. This natural functionalization can significantly enhance the stability and sensitivity of the resulting sensor, potentially outperforming chemically synthesized counterparts that lack such complex surface chemistry. The present study explores the intersection of extremophilic microbiology and environmental nanotechnology by investigating the biosynthetic potential of two specific archaeal strains, Natronolimnohabitans innermongolicus A6 and Natrialba sp. RA5. These strains were isolated from the extreme hyper-saline and alkaline environment of El-Hamra Lake in Wadi El-Natrun, Egypt. The research focuses on the green synthesis of Fe3O4 nanoparticles and their subsequent application as electrochemical sensing platforms for the detection of Ibuprofen. Through a comprehensive and comparative analysis of the crystallinity, surface chemistry, and morphological characteristics of the nanoparticles produced by each strain, this investigation seeks to elucidate how strain-specific biomolecules influence the electrochemical performance of the sensors. By evaluating the sensitivity, selectivity, and kinetic mechanisms of these biogenic sensors specifically focusing on electron transfer efficiency and chemisorption at the electrode interface this work highlights a robust and sustainable approach for the electrochemical monitoring of emerging pharmaceutical pollutants, ultimately contributing to the development of eco-friendly technologies for aquatic preservation. The novelty of this work lies in the pioneering use of extremophilic archaea from hyper-saline alkaline environments to engineer strain-specific Fe3O4 nanoparticles, demonstrating that microbial-specific biomolecules can be harnessed to precisely tune the surface chemistry and crystallinity of biogenic sensors for the enhanced electrochemical detection of pharmaceutical pollutants.
Results
Isolation, screening, and molecular identification
In this study, 52 microbial isolates were recovered from various water and sediment samples collected from El-Hamra Lake, Wadi El-Natrun20. All isolates were identified as haloalkaliphilic archaea and subjected to primary screening for the synthesis of magnetic nanoparticles. Based on the screening results, two specific isolates (RA5 & A6) were selected as candidates for the green synthesis of magnetic nanoparticles and further investigations (Fig. 1). Molecular characterization of the selected isolate library identified strain A6 as Natronolimnohabitans innermongolicus and strain RA5 as Natrialba sp. Their respective sequences were deposited in GenBank under accession numbers (PX879702 & PX879555), respectively. Other studies demonstrated the production of superparamagnetic iron oxide nanoparticles by halophilic archaea with uniform size and shape, exhibiting properties suitable for biomedical applications like hyperthermia cancer therapy. Biosynthesis using haloarchaea offers advantages including eco-friendliness, cost-effectiveness, and the ability to produce nanoparticles with high purity and magnetic responsiveness21. Other halophilic prokaryotes, including bacteria and archaea from solar salterns, have also been reported to synthesize various metal nanoparticles such as silver, selenium, and manganese oxides, which show antimicrobial activity and stability22,23. While bacterial and fungal syntheses of Fe3O4 have been widely documented, haloalkaliphilic archaea offer a distinct biochemical platform. Their adaptation to hypersaline and alkaline conditions necessitates a proteome with high negative surface charge, which significantly alters the nucleation kinetics of iron oxides compared to the neutral-pH pathways found in common mesophilic bacteria and fungi. Furthermore, the inherent alkalinity of the archaeal growth medium facilitates a more direct biomimetic route to magnetite formation than that observed in fungal systems. For example Haloarchaeal ferritins (e.g., Halobacterium salinarum DpsA) use acidic outer residues to capture iron, then channel it through pores to internal ferroxidase and nucleation centers, where iron-oxide clusters form on pre-organized sites, this demonstrates that a highly negative surface charge does not directly nucleate magnetite; instead, it requires internal, specifically arranged nucleation centers24. Magnetotactic bacteria maintain a locally alkaline magnetosome lumen even at neutral external pH, using ion transporters (MamB/MamM/MamN) to export protons and create optimal conditions for magnetite formation. Thus, alkaline archaeal media may indeed be closer to these “internal” conditions than typical fungal systems, but the decisive control still lies in local pH and charge in microcompartments, not bulk medium alone25.
Red arrow indicates magnetic Fe₃O₄ nanoparticles moving toward a magnet.
Microscopic analysis of archaeal-mediated magnetic nanoparticles production
Under the Scanning Electron Microscope (SEM), N. innermongolicus and Natrialba sp. strains exhibit the distinct, often flattened morphology characteristic of haloalkaliphilic archaea adapted to extreme osmotic pressure. N. innermongolicus typically appears as short, pleomorphic coccoid shapes. In contrast, Natrialba sp. strains demonstrate extreme pleomorphism, often manifesting as flattened discs, triangles, or irregular plates, these strains frequently display non-peptidoglycan cell envelopes. Both genera belong to the family Natrialbaceae and thrive in highly saline and alkaline environments. These morphological traits are consistent with their ecological niches in extreme environments, supporting their classification and physiological adaptations (Fig. 2). However, specific SEM imaging studies directly comparing these two genera remain limited in the literature26.
Haloalkaliphilic strains A) N. innermongolicus A6 and B) Natrialba sp. RA5.
ATR-FTIR analysis of the chemical bonding and purity of green-synthesized magnetic nanoparticles
Figure 3 demonstrates the ATR‑FT/IR spectra of the biosynthesized Fe₃O₄ nanoparticles obtained from RA5 and A6 exhibit characteristic vibrational modes confirming the formation of magnetite alongside biomolecular functional groups originating from archaeal metabolites. A strong Fe–O stretching band appears at ~580–560 cm⁻¹ in both samples, validating the formation of the spinel Fe₃O₄ structure27. The RA5-derived nano-particles exhibit a sharper Fe–O band, suggesting higher crystallinity and reduced surface disorder compared to A6. A broad O–H stretching band at ~3400–3300 cm⁻¹ is present in both samples but appears more intense in RA5, indicating a higher density of surface hydroxyl groups which may enhance electron transfer during electrochemical sensing28. A6 nanoparticles display stronger aliphatic C–H stretching bands ( ~ 2920 and 2850 cm⁻¹), suggesting a thicker organic shell. Additionally, A6 exhibits more intense amide I ( ~ 1650 cm⁻¹) and amide II ( ~ 1540 cm⁻¹) bands, indicating richer proteinaceous capping29. A pronounced band at ~1100–1000 cm⁻¹ reflects polysaccharide contributions, further supporting the presence of a thicker biomolecular coating30. The ATR-FTIR results demonstrate that hydroxyl, amide, carboxylate, and polysaccharide groups constitute the primary stabilizing and electrochemically active functionalities on the Fe₃O₄ surface. These groups not only ensure colloidal stability via electrostatic and steric effects but also create an organic–inorganic interfacial layer capable of inducing local lattice strain and enhancing analyte adsorption and electron transfer. The higher intensity of these bands in A6 explains its thicker organic corona and stronger surface functionality, whereas the relatively weaker bands in RA5 are consistent with a thinner capping layer and higher crystallinity. Finally, RA5 produces cleaner, less heavily capped Fe₃O₄ nanoparticles with sharper structural bands, while A6 generates particles with thicker organic shells. These chemical differences suggest that RA5-derived nanoparticles will likely facilitate superior electron transfer, making them more suitable for electrochemical bio-sensing applications. Table 1 illustrates the FT/IR bands for both samples.
FT/IR spectra for haloalkaliphilic strains RA5 (black line) and A6 (red line).
Crystallographic characterization of green-synthesized magnetic nanoparticles using XRD
Figure 4 displays the XRD patterns of the Fe₃O₄ nanoparticles synthesized by RA5 and A6, confirming that both samples crystallize in the inverse spinel structure of magnetite (JCPDS No. 19-0629). All the characteristic diffraction peaks of magnetite are clearly observed and can be indexed to the crystallographic planes (220), (311), (400), (422), (511), and (440), demonstrating the high phase purity and crystalline nature of the biosynthesized nanoparticles. A slight shift in the peak positions relative to the standard JCPDS values is observed, which can be attributed to lattice distortion, nanoscale size effects, and strain induced by the biomolecule-mediated synthesis route. For RA5, the diffraction peaks are located at approximately 27.12°, 31.57°, 35.39°, 45.19°, 56.31°, and 66.1°, while the corresponding reflections for A6 appear at about 27.14°, 31.61°, 35.48°, 45.27°, 56.32°, and 66.15°. The extracted peak positions and full width at half maximum (FWHM) values obtained directly from the experimental diffractograms are summarized in Table 2. The crystallite sizes were calculated using the Scherrer equation (K = 0.9, λ = 1.5406 Å) based on the experimentally measured FWHM values of the most intense diffraction peaks, namely (220), (311), and (422). The calculated crystallite sizes are presented in Table 3. The obtained values fall in the range of 9.5–12.3 nm, yielding an average crystallite size of approximately 10–11 nm, which is in excellent agreement with previously reported data for biosynthesized magnetite nanoparticles31. RA5 exhibits a slightly larger crystallite size at the (220) reflection ( ≈ 12.7 nm), whereas smaller coherent domain sizes are obtained at the (311) and (422) planes ( ≈ 10.4 and ≈9.55 nm, respectively). A6 shows comparable crystallite dimensions, with values of ≈11.5 nm, ≈9.8 nm, and ≈9 nm for the (220), (311), and (422) planes, respectively, indicating that both strains promote the formation of nanocrystalline magnetite with well-defined structural order32. The slight shift observed for the low-angle diffraction peak of A6, together with the peak broadening, can be attributed to lattice strain and the presence of a denser organic capping layer on the nanoparticle surface. Such biomolecule-mediated surface coatings are known to induce microstrain and influence crystal growth kinetics, leading to subtle changes in diffraction peak positions and widths33. This interpretation is fully consistent with the ATR-FTIR results, which show stronger amide and polysaccharide bands for A6 compared to RA5, confirming the presence of a thicker organic corona. In contrast, the relatively cleaner surface of the RA5-derived nanoparticles, as evidenced by weaker organic-related vibrational bands in the FTIR spectra, correlates with sharper diffraction peaks and slightly larger coherent crystalline domains. This higher crystallinity is expected to facilitate more efficient electron transport and interfacial charge transfer, which is highly beneficial for electrochemical sensing applications34. Conversely, the stronger organic shell associated with A6 is advantageous for improving surface functionality and colloidal stability, albeit with a moderate impact on electronic coupling.
XRD spectra for haloalkaliphilic strains RA5 (black line) and A6 (red line).
Morphological characterization and elemental composition analysis of synthesized magnetic nanoparticles via SEM-EDX
Figure 5 shows the SEM micrographs which revealed distinct morphological differences between the Fe₃O₄ nanoparticles synthesized by the two archaeal strains. The RA5-derived material exhibited denser aggregates of magnetite with relatively uniform grain connectivity, forming compact clusters over the biological matrix. This suggests a more homogeneous nucleation and growth behavior, consistent with the sharper XRD peaks and the cleaner surface chemistry observed in the FT/IR spectrum. In contrast, the A6 sample presented more heterogeneous surface features, with loosely packed clusters, irregular grain boundaries, and a more textured appearance. These morphological variations align with the FT/IR evidence indicating stronger biomolecular capping (higher amide and polysaccharide bands), which typically introduces steric hindrance and leads to broader aggregation patterns. Such differences in microstructure reflect the distinct biochemical profiles of each archaeal supernatant and their influence on nano-particle formation and assembly. Similar strain-dependent morphological variation in biologically synthesized metal oxides has been reported in the literature34. Figure 6 illustrates the EDX analyses of the RA5- and A6-derived magnetite nanoparticles which revealed clear differences in their surface elemental composition that directly correlate with the biochemical environment of each archaeal strain. The RA5 sample exhibited a composition dominated by Fe (40.62%) and exceptionally high Cl and Na contents (44.39% and 10.36%, respectively). This profile reflects the extreme salinity of the haloarchaeal supernatant and indicates that RA5-derived Fe₃O₄ nanoparticles carry minimal biomolecular residues35, consistent with their compact SEM morphology and weak FT/IR amide/polysaccharide bands. In contrast, the A6 sample displayed a markedly higher oxygen percentage (23.68%) and measurable phosphorus (0.70%), indicating the presence of significant organic capping layers composed of proteins, polysaccharides, or phospholipid-associated biomolecules. Meanwhile, Fe content remained comparable between both samples ( ~ 41%), confirming similar magnetite formation efficiency. The elevated O, P, and moderate Cl and Na levels in A6, combined with its looser SEM morphology and stronger FT/IR biomolecular signatures, collectively demonstrate that A6 promotes thicker bio-organic surface functionalization, whereas RA5 produces cleaner, more crystalline magnetite surfaces with minimal biological adsorption. This compositional contrast aligns directly with the morphological and structural trends observed across SEM, FT/IR, and XRD analysis. Table 4 illustrates a comparison between the EDS parameters of AR5 and A6.
a RA5 and b A6.
a RA5 and b A6.
High-resolution morphological assessment and particle size distribution of green-synthesized magnetic nanoparticles via TEM
Figure 7 demonstrates TEM micrographs of the biosynthesized Fe₃O₄ nanoparticles revealed distinct strain-dependent differences in particle morphology, dispersion behavior, and nano-structural organization between the RA5 and A6-derived samples. The RA5 nanoparticles displayed quasi-spherical crystalline particles forming dense, tightly packed nanoclusters, with an average particle size of approximately 14.8 ± 3.2 nm based on measurements from representative fields of view. The particles exhibited strong contrast and well-defined boundaries, indicating highly crystalline magnetite domains with limited surface organic coating36. This compact nano-structuring suggests that the RA5 supernatant promotes rapid nucleation and growth of Fe₃O₄ cores with minimal steric stabilization, allowing particles to associate into coherent clusters. In contrast, the A6 sample exhibited more individually distinguishable nanoparticles with reduced aggregation and broader inter-particle spacing. The average particle size was slightly smaller, approximately 11.5 ± 2.7 nm, indicating slower crystallization kinetics and stronger surface passivation. The softer particle edges and the less compact assemblies in A6 can be attributed to the richer biomolecular capping layer, consistent with EDX results showing elevated oxygen and phosphorus levels (indicative of proteins and phospholipid residues) and the stronger FT/IR amide/polysaccharide signatures35. These biomolecular species act as natural stabilizers, limiting uncontrolled aggregation and producing finer, more dispersed Fe₃O₄ nanoparticles. The structural differences observed in TEM are fully aligned with the XRD findings: RA5 shows sharper and more intense diffraction peaks, reflecting larger coherent crystallite domains, while A6 exhibits slightly broader peaks, suggesting microstrain introduced by the organic shell. Together, these trends demonstrate that RA5 produces more crystalline but more aggregated nanoparticles, whereas A6 yields smaller, better-stabilized nanocrystals. The strain-dependent TEM characteristics directly translate into performance differences in the intended bio-sensing application. The compact nanoclusters and higher crystallinity observed in RA5 provide enhanced electron-transfer pathways, making RA5-derived Fe₃O₄ a stronger candidate for electrochemical bio-sensing, where conductivity and charge mobility are essential37. Conversely, the smaller size, better dispersion, and richer surface functionalization of A6 nanoparticles make them advantageous for surface-driven sensing mechanisms, such as biomolecule immobilization, antibody/aptamer conjugation, or binding affinity-based detection. The biomolecular corona formed naturally around A6 particles provides abundant reactive groups, potentially improving analytes interaction and sensitivity. Thus, RA5-based nanoparticles are structurally optimized for signal transduction, while A6-derived nanoparticles are more suitable for surface functionalization and bio-recognition. The complementary features of both materials highlight the significant influence of archaeal strain biochemistry on the nanoscale architecture and ultimately on the functional performance of biosynthesized Fe₃O₄ in sensing applications38.
a RA5 and b A6.
Electrocatalytic activity and sensing performance of Fe3O4 based electrodes
Vibrating Sample Magnetometry (VSM) analysis
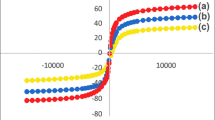
The M–H hysteresis loops of both RA5- and A6-derived Fe₃O₄ nanoparticles are shown in Fig. 8 exhibit typical S-shaped curves with almost closed hysteresis, confirming their superparamagnetic behavior at room temperature39. The magnetization curves pass very close to the origin, indicating negligible eminence and coercively, which is a hallmark of magnetite nanoparticles with dimensions below the single-domain limit. From direct graphical extraction of the VSM curves, the RA5 sample reaches magnetic saturation at an applied field of approximately ±10 kOe, with a saturation magnetization (Ms) of ≈ 63.4 emu g⁻¹. In contrast, the A6-derived nanoparticles show a slightly lower saturation magnetization of ≈ 54.8 emu g⁻¹, reaching saturation at a comparable magnetic field. The remnant magnetization (Mr) values for both samples are extremely low ( < 0.5 emu g⁻¹), while the coercivity (Hc) values are close to zero ( < 30 Oe), confirming the absence of magnetic hysteresis. The higher Ms value observed for RA5-derived nanoparticles is consistent with their higher crystallinity and cleaner magnetite surface, as previously evidenced by sharper XRD reflections and weaker biomolecular signatures in FT/IR and EDX analysis. Reduced surface spin disorder allows more Fe²⁺/Fe³⁺ magnetic moments to align coherently with the applied field, thereby enhancing the overall magnetization. Conversely, the reduced Ms of the A6 sample can be attributed to the presence of a thicker organic/biomolecular shell, as confirmed by higher oxygen and phosphorus contents in EDX and stronger amide and polysaccharide bands in FT/IR spectra. These surface-bound biomolecules introduce spin canting and partially disrupt magnetic exchange interactions at the nanoparticle surface, leading to a lower effective magnetic moment while preserving superparamagnetic behavior. The observed superparamagnetic response correlates well with the TEM-derived particle sizes ( ~ 10–15 nm) and the XRD-estimated crystallite sizes, confirming that both samples consist of single-domain magnetite nanoparticles rather than bulk magnetic phases40. From an application perspective, these magnetic characteristics are highly advantageous. Table 5 summarizes the VSM parameters. The higher saturation magnetization of RA5 nanoparticles enables faster magnetic manipulation, improved magnetic separation, and more efficient electron transfer at the electrode interface, which is beneficial for enhancing electrochemical signal intensity41. Meanwhile, the A6 nanoparticles, despite their slightly lower Ms, offer richer surface chemistry for analyte interaction and immobilization, potentially improving sensitivity and selectivity42. Thus, the VSM results highlight a clear trade-off between magnetic strength (RA5) and surface functionalization (A6), demonstrating how archaeal strain selection directly governs the balance between magnetic performance and biosensor functionality.
Hysteresis (M–H) loops of biosynthesized Fe₃O₄ nanoparticles produced using Natrialba sp. (RA5) (black line) and N. innermongolicus (A6) (red line).
Cyclic voltammetric characterization of the electrochemical interface and charge transfer with different concentrations of IBP
The electrochemical sensitivity of the prepared electrode samples toward detecting of IBP in water samples were investigated 2-electrodes electrochemical cell with different concentrations of IBP in 1% NaCl electrolyte (Table 6). Figure 9 shows the cyclic voltammograms of prepared electrodes with different concentrations of IBP (0–100 mg/L Water). It is noticeable that, the sensitivity of the prepared electrodes could be expressed as shon in Eqs. (1 – 2):
Where; I is peak current (µA), C is IBP concentration (mg/L).
CV of prepared a RA5 and b A6 electrodes with different concentrations of IBP (0–100 mg/L water).
Differential potential voltammetry (DPV) of prepared electrodes
Figure 10 presented the differential potential voltammetry (DPV) of prepared electrodes and its calibration curves of prepared electrodes of RA5 (Fig. 10a,10b) and A6 (Fig. 10c, 10d) electrodes with different concentrations of IBP (0–100 mg/L Water). For both samples, the highest sensitivity was recorded at potential of V and V for RA5 and A6 samples respectively, as shown in Fig. 10a, 10c. Calibration curves were presented in Fig. 10b, 10d.
a, b RA5 and c, d A6 electrodes with different concentrations of IBP (0–100 mg/L Water).
Electrochemical kinetics
The kinetic behavior of the prepared electrochemical electrodes sensor toward IBP were systematically evaluated using pseudo first-order and second-order kinetic models, as illustrated in Fig. 11a, d. For both sensing platforms (RA5 and A6), the linear fitting of the experimental data demonstrates that the adsorption and electrochemical interaction of IBP at the electrode surface can be quantitatively described using classical kinetic approaches. The obtained correlation coefficients (R²) provide valuable insight into the dominant sensing mechanism and the efficiency of analyte–electrode interactions26. For the RA5 sensor, the first-order kinetic plot (Fig. 11a) exhibits a moderate linearity (R² = 0.8904), suggesting that the rate of IBP interaction is partially governed by concentration-dependent surface processes, but may also be influenced by mass transfer limitations or heterogeneous active sites. In contrast, the second-order kinetic model (Fig. 11b) shows an excellent linear relationship with a significantly higher correlation coefficient (R² = 0.999), indicating that the sensing process is better described by a second-order reaction mechanism. This behavior implies that chemisorption and electron-transfer-controlled processes dominate, involving strong interactions between IBP molecules and electroactive sites on the electrode surface43. A similar kinetic trend is observed for the A6 sensor, where the first-order kinetic fitting (Fig. 9c) displays good linearity (R² = 0.9643), yet remains inferior to the second-order model. The second-order kinetic plot (Fig. 11d) yields an almost ideal linear response (R² = 0.9981), confirming that the IBP sensing process follows second-order kinetics. The higher slope observed for the first-order plot of A6 compared to RA5 suggests a faster apparent interaction rate, which can be attributed to an increased density of active sites or improved electron-transfer pathways within the A6 electrode architecture. Overall, the superior agreement with the second-order kinetic model for both RA5 and A6 electrodes indicates that the electrochemical sensing mechanism is predominantly controlled by surface-confined redox reactions rather than simple diffusion-controlled adsorption. This behavior reflects strong affinity and efficient charge transfer between IBP molecules and the electrode surface, which is highly desirable for sensitive and reliable electrochemical detection. Moreover, the enhanced kinetic performance of the A6 sensor highlights the critical role of electrode composition and structure in accelerating interfacial reaction kinetics. These kinetic findings confirm that the developed electrochemical sensor exhibits rapid and concentration-dependent response toward IBP, governed mainly by chemisorption-driven and electron-transfer-controlled processes. Such kinetic characteristics not only validate the robustness of the sensing mechanism but also support the practical applicability of the sensor for accurate and fast monitoring of pharmaceutical pollutants in electrochemical sensing platforms. The kinetic parameters summarized in Table 7 kinetics further elucidate the adsorption and electrochemical sensing behavior of IBP on the RA5 and A6 electrodes using pseudo-first-order and pseudo-second-order kinetic models. For both electrodes, a clear distinction is observed between the adequacy of the two models in describing the experimental data, as reflected by the calculated kinetic constants, equilibrium adsorption capacities (qₑ), and correlation coefficients (R²). These parameters provide quantitative insight into the nature of the IBP–electrode interaction and the rate-controlling steps of the sensing process. For the RA5 electrode, the pseudo-first-order model yields a relatively low correlation coefficient (R² = 0.8904) and a smaller calculated qₑ value (2.4238), indicating a limited ability of this model to accurately represent the experimental kinetics. In contrast, the pseudo-second-order model exhibits an excellent linearity (R² = 0.999) along with a significantly higher equilibrium adsorption capacity (qₑ = 4.7259) and rate constant (K₂ = 11.7843). This strong agreement suggests that the IBP sensing process on RA5 is predominantly governed by chemisorption involving electron sharing or transfer between IBP molecules and electroactive sites, rather than simple physisorption or diffusion-controlled processes. A similar kinetic trend is observed for the A6 electrode, where the pseudo-second-order model again provides a superior fit (R² = 0.9981) compared to the pseudo-first-order model (R² = 0.9643). Notably, the A6 electrode exhibits a much higher qₑ value in the pseudo-first-order model (12.0143), reflecting a higher apparent adsorption capacity, which can be attributed to its enhanced surface area or higher density of accessible active sites. However, the pseudo-second-order model yields a qₑ value (11.0253) that is closer to the experimentally expected capacity, reinforcing its reliability in describing the actual sensing mechanism. Comparative analysis between RA5 and A6 highlights the superior electrochemical activity of the A6 electrode, as evidenced by its higher qₑ values and larger pseudo-first-order rate constant (K₁ = 3.2002) relative to RA5 (K₁ = 1.7337). These results indicate faster interfacial kinetics and stronger IBP–electrode interactions for A6, which may arise from improved electrode composition, enhanced conductivity, or more favorable surface chemistry. Meanwhile, the higher K₂ value obtained for RA5 suggests more rapid chemisorption kinetics once IBP molecules are adsorbed onto active sites. Generally, the kinetic modeling confirms that the IBP electrochemical sensing process for both RA5 and A6 electrodes is best described by the pseudo-second-order model, underscoring the dominance of surface-controlled chemisorption and electron-transfer processes. These findings validate the robustness and efficiency of the developed sensor platforms and emphasize the critical influence of electrode structure on adsorption capacity and kinetic performance, which are key factors for achieving high sensitivity and rapid response in electrochemical sensing applications.
Pseudo 1st and 2nd order models of RA5 (a, b) and A6 (c, d) electrodes, respectively.
Proposed sensing mechanism
The electrochemical sensing performance of RA5- and A6-derived Fe₃O₄ nanoparticles toward ibuprofen arises from a synergistic interplay between the crystalline magnetite core and the archaeal-derived organic surface corona, rather than from either factor alone. As illustrated in Fig. 12, the sensing process involves two sequential and coupled steps: (i) surface preconcentration of ibuprofen molecules through the organic corona and (ii) interfacial electron transfer mediated by the Fe₃O₄ crystalline core. FTIR analysis confirmed the presence of abundant surface functional groups, including hydroxyl, amide, and polysaccharide-related moieties, which provide multiple active binding sites for ibuprofen via hydrogen bonding and electrostatic interactions. These functional groups act as a molecular recognition layer that enhances the local concentration of ibuprofen at the electrode surface. This adsorption-driven preconcentration step is more pronounced for A6-derived nanoparticles due to their denser organic corona, explaining their superior surface functionality and colloidal stability. Simultaneously, the XRD results demonstrate that both samples possess a well-defined inverse spinel magnetite structure, while RA5-derived nanoparticles exhibit slightly higher crystallinity and sharper diffraction peaks. Together with their higher saturation magnetization observed in VSM measurements, this indicates a more ordered crystal lattice and more efficient electron transport pathways within the Fe₃O₄ core. Such enhanced crystallinity facilitates faster charge transfer between the adsorbed ibuprofen molecules and the electrode, resulting in higher electrochemical signal amplification. Therefore, RA5 benefits primarily from superior electronic conductivity and charge transport efficiency. The nanoscale dimensions observed by TEM further strengthen this mechanism by providing a large surface-to-volume ratio, which increases the density of active adsorption sites and shortens the diffusion path for electrons. Consequently, the sensing response is governed by a balance between adsorption capability (controlled by the organic corona) and electron transfer efficiency (controlled by the crystalline magnetite core). In this context, A6-derived nanoparticles favor surface-driven processes due to their thicker biomolecular coating, enhancing analyte capture and interfacial stability, whereas RA5-derived nanoparticles favor charge transport processes due to their higher crystallinity and magnetic/electronic ordering. The experimentally observed electrochemical performance reflects this trade-off: RA5 shows enhanced signal intensity and faster electron transfer kinetics, while A6 offers improved surface functionality and molecular interaction efficiency. Thus, the proposed unified mechanism establishes that the sensing enhancement is not exclusively due to the inorganic Fe₃O₄ material or the biological surface corona, but rather to their cooperative action. Herein, the experimental data and calculated results are in a good agreement with the pseudo-second-order kinetic model indicates that the IBP sensing process is governed by interfacial electron-transfer–controlled chemisorption rather than simple diffusion or weak physisorption. In this model, the reaction rate is proportional to the square of the number of available active sites, implying that charge transfer occurs through strong electronic coupling between IBP molecules and the Fe₃O₄ nanoparticle-modified electrode surface. At the electrode–nanoparticle interface, the mixed Fe²⁺/Fe³⁺ redox centers of magnetite facilitate rapid electron hopping and act as efficient charge-transfer mediators during IBP oxidation. The involvement of surface functional groups originating from the biogenic organic corona further promotes inner-sphere electron transfer by stabilizing IBP adsorption and lowering the activation energy for interfacial charge exchange. Consequently, the dominance of pseudo-second-order kinetics reflects a sensing mechanism controlled by surface-confined electron transfer processes rather than mass-transport limitations, confirming the key role of the Fe₃O₄ nanoparticle interface in governing the electrochemical response. To place the analytical performance of the developed archaeal Fe₃O₄-based sensors in a broader context, a comparison with previously reported electrochemical sensors employing Fe₃O₄ nanoparticles and related nanocomposites is summarized in Table 8.
Electrochemical sensing mechanism of ibuprofen with green-synthesized Fe3O4.
As shown in Table 8, Fe₃O₄-based electrochemical platforms reported in the literature exhibit a wide range of detection limits and linear ranges depending on the electrode architecture and target analyte. The archaeal-derived Fe₃O₄ nanoparticles developed in this study demonstrate competitive analytical performance, particularly under saline conditions, highlighting the advantage of biologically functionalized magnetic nanomaterials for pharmaceutical monitoring in complex aquatic matrices. Overall, this study highlights the potential of haloalkaliphilic archaea as sustainable bio-factories for the green synthesis of magnetite nanoparticles with tunable structural and surface properties. Strain-dependent differences in crystallinity and surface chemistry enable effective control over the electrochemical performance of Fe₃O₄-based sensors for ibuprofen detection under saline conditions, supported by comprehensive physicochemical characterization. The proposed approach contributes to sustainable water quality monitoring and aligns with key United Nations Sustainable Development Goals, including SDG 6 (Clean Water and Sanitation), SDG 3 (Good Health and Well-Being), SDG 12 (Responsible Consumption and Production), and SDG 9 (Industry, Innovation and Infrastructure). While the sensing mechanism is mainly supported by indirect evidence, further validation using advanced surface-sensitive techniques, evaluation in real environmental samples, and benchmarking against chemically synthesized Fe₃O₄ nanoparticles will strengthen future practical implementation.
Discussion
This study demonstrates a sustainable and green biotechnological route for the fabrication of magnetite (Fe₃O₄) nanoparticles using haloalkaliphilic archaea isolated from the extreme environment of El-Hamra Lake. By exploiting the distinct metabolic characteristics of Natrialba sp. RA5 and Natronolimnobius innermongolicus A6, the synthesis process enabled strain-dependent control over crystallinity, surface chemistry, and magnetic behavior of the resulting nanoparticles. The key novelty of this work lies in advancing green synthesis toward a precision bioengineering approach, where the choice of extremophilic archaeal strain acts as a biological architect governing both the crystalline magnetite core and the composition of the bio-organic surface corona. Comprehensive physicochemical characterization revealed clear structure–property relationships. RA5-derived Fe₃O₄ exhibited higher crystallinity and more compact nanostructures, facilitating efficient electron transfer, whereas A6-derived nanoparticles possessed a thicker biomolecular corona rich in proteins and polysaccharides, enhancing surface functionality and analyte interaction. When integrated into electrochemical sensing platforms, both materials enabled effective ibuprofen detection under saline conditions, with kinetic analysis confirming a surface-controlled chemisorption mechanism consistent with strong analyte–surface affinity and efficient charge transport. Beyond demonstrating sensing performance, this work bridges extremophilic microbiology and environmental nanotechnology, highlighting archaeal-mediated synthesis as an eco-friendly strategy for producing functional magnetic nanomaterials. Future research will focus on extending this biosynthetic approach to other multifunctional nanomaterials, incorporating advanced electrochemical and surface-sensitive techniques to further elucidate sensing mechanisms, validating performance in real environmental water samples, assessing long-term stability and reusability, and scaling up the process toward portable or in situ sensing devices for comprehensive water quality monitoring in saline and extreme environments.
Methods
Isolation source and screening haloalkaliphilic archaeal isolates for magnetic nanoparticle synthesis
The study involved collecting samples from the hypersaline El-Hamra Lake in Wadi El-Natrun for the isolation of haloalkaliphilic archaea. The specific isolation protocol utilized a culture medium with the following composition in g/L: casamino acids, 5; KH2PO4, 1; MgSO4 7H2O, 0.2; NaCl, 200; and Na2CO3, 18, supplemented with 1 ml/L of a trace metal solution containing ZnSO4 7H2O, 0.1, MnCl2 4H2O, (0.03), H3BO3 (0.3), CoCl2 6H2O, (0.2), CuCl22H2O, (0.01), NiCl2 6H2O, (0.02), and Na2MoO4 H2O, (0.03), following established methods20. Samples were appropriately diluted to fit the instrument’s calibration range, which was established using blank and standard solutions. To test the halophilic archaeal isolates’ potential for magnetic nanoparticles synthesis, a seed culture was grown to an OD ~ 0.9, and 1 ml inoculum was transferred to 250 ml flasks containing 100 ml of basal medium adjusted to pH 11. These cultures were incubated at 37ﹾC while shaking at 200 rpm for 7 days, after which the cell-free supernatant was collected and processed for magnetic nanoparticle synthesis and characterization using the following methods
Preparation of the inoculums
The inoculums for all subsequent experiments were prepared from a pre-cultures grown for 7 days. Growth was monitored spectrophotometrically by measuring the optical density at 600 nm (OD600). The cultures, which had reached the late logarithmic phase (achieving an OD600 of 0.99), were then used to inoculate the main experimental media at a final concentration of 10% (v/v), unless otherwise specified33.
Green synthesis of magnetic nanoparticles
The biosynthesis of magnetite nanoparticles (Fe3O4) will be conducted by reacting equal volume of a 2:1 molar of FeCl3 and FeSO4 solution with the archaeal supernatant. The metal precursor solution will be added dropwise to the archaeal supernatant while the mixture is continuously stirred at 500 rpm at temperature 37°C and incubated for 48 h to ensure uniform reaction kinetics and particles formation. Following the reaction, the formation of black precipitates (indicating magnetite) will be observed. These biosynthesized nanoparticles will be collected via centrifugation at 10,000 rpm for 20 min s. To purify the product, the collected nanoparticles will be washed sequentially: twice with absolute ethanol to remove organic impurities, and then once with distilled water to remove residual salts. Finally, the purified magnetic nanoparticles will be dried under vacuum conditions at 60°C for 12 h to obtain the final powder product (based on the method described by Yew et al.,33.
Microscopic examination of magnetic nanoparticles producing haloalkaliphilic archaeal strains
The two isolates (RA5 & A6) cultured broth samples were prepared for Scanning Electron Microscopy (SEM) analysis. The samples were metalized with a uniform thin gold film via sputter deposition using a JEOL JFC-1100 E sputtering device (JEOL, USA) for a duration of 12 min. The morphology was subsequently characterized using a JEOL JSM 5300 scanning electron microscope (JEOL, USA) operating at an accelerating voltage of 20 kV. All electron microscopy was conducted in the Centre Laboratory, City of Scientific Research and Technological Applications20.
Molecular identification of magnetic nanoparticles producing haloalkaliphilic archaeal strains
Molecular identification of the magnetic nanoparticle-synthesizing haloalkaliphilic archaeal isolates was performed using 16S rRNA gene analysis. Genomic DNA was prepared using a simplified and rapid protocol in which individual colonies were picked directly from agar plates and subjected to a brief cell disruption step. The 16S rRNA genes were amplified by polymerase chain reaction (PCR) using universal archaeal primers, generating amplicons of approximately 1500 bp. The resulting sequences were assembled and aligned using the BioEdit Sequence Alignment Program with the ClustalW module. Taxonomic identification was achieved by comparing the obtained sequences with reference sequences in the NCBI database using the BLAST algorithm (https://www.ncbi.nlm.nih.gov), enabling accurate species-level assignment of the isolates20.
ATR‑FT/IR
Fourier-transform infrared spectroscopy (FT/IR) was performed using an ATR (attenuated total reflectance) configuration to investigate the surface functional groups of the biosynthesised magnetite nanoparticles. Spectra were recorded using a Thermo Scientific Nicolet iS10 FT/IR spectrometer equipped with a diamond ATR crystal. Approximately 2–3 mg of the dried Fe₃O₄ nanoparticles produced by the isolates (RA5) and (A6) were directly placed onto the ATR crystal, and uniform contact was ensured by applying controlled pressure using the built‑in pressure arm. All spectra were collected in the range of 4000–400 cm⁻¹ with a resolution of 4 cm⁻¹ and averaged over 32 scans. Background spectra were recorded before each measurement and automatically subtracted. All spectra were baseline‑corrected and normalized for comparison44,45.
XRD analysis
X-ray diffraction (XRD) patterns of the biosynthesized Fe₃O₄ nanoparticles were recorded using a Bruker D8 Advance diffractometer equipped with a Cu Kα radiation source (λ = 1.5406 Å) operating at 40 kV and 40 mA. Samples from both archaeal strains (RA5) and (A6) were finely ground and mounted onto a zero-background silicon holder46. Diffraction data were collected over the 2θ range of 20–90°, with a step size of 0.02° and a counting time of 0.5 s per step. Instrumental broadening was determined using a standard silicon reference, and the corrected full width at half maximum (FWHM) values were used to calculate the average crystallite size. The crystallite size (D) of the magnetite nanoparticles was estimated using the Debye-Scherrer equation as shown in Eq. (3):
Where D is the crystallite size, K is the shape factor (taken as 0.9), λ is the wavelength of the X-ray source (1.5406 Å), β is the full width at half maximum (FWHM) of the most intense diffraction peak in radians, and θ is the Bragg diffraction angle.
For each sample, the (220), (311), and (400) reflections of magnetite were used for crystallite size evaluation, and the final crystallite size was calculated as the average of the three values.
SEM and EDX analysis
The surface morphology and elemental composition of the biosynthesized Fe₃O₄ nanoparticles produced by the isolates (RA5) and (A6) were examined using a JEOL JSM-IT200 Scanning Electron Microscope equipped with a secondary electron detector (SED). Samples were sputter-coated with a thin Au layer to prevent surface charging, and images were acquired under high-vacuum mode at an accelerating voltage of 20 kV, a working distance of 10 mm, and a fixed magnification of ×1000 for both samples to ensure a direct morphological comparison47. Elemental analyses were performed using an Oxford Instruments X-act Energy-Dispersive X-ray Spectroscopy (EDS) system attached to the SEM. Spectra were collected using the ZAF standard-less quantification method with a live time of 30 s. The detector was calibrated prior to analysis using a pure copper standard. Elemental mass percentages were obtained from representative regions of each sample48,49.
Transmission electron microscopy (TEM)
The morphology and nanoscale structural features of the biosynthesized Fe₃O₄ nanoparticles obtained from the isolates (RA5) and (A6) were investigated using Transmission Electron Microscopy (TEM). TEM images were acquired on a JEOL JEM-2100 transmission electron microscope operated at an accelerating voltage of 200 kV. Nanoparticle suspensions were sonicated for 5 min, and a drop of the diluted dispersion was deposited onto a carbon-coated copper grid (300-mesh) and allowed to dry under ambient conditions prior to imaging. The particle morphology, aggregation behavior, and approximate size range were evaluated from representative micrographs collected for each sample47.
Vibrating sample magnetometry (VSM)
The magnetic properties of the biosynthesized Fe₃O₄ nanoparticles obtained from Natrialba sp. RA5 and Natronolimnobius innermongolicus (A6) were investigated using a Lake Shore 7407 Vibrating Sample Magnetometer (VSM). Measurements were performed at room temperature (300 K) under an applied magnetic field ranging from −15 kOe to +15 kOe. Approximately 10 mg of dried nanoparticle powder was placed in a non-magnetic sample holder, and the magnetization (M) as a function of the applied magnetic field (H) was recorded. The magnetic parameters saturation magnetization (Ms), remanent magnetization (Mr), and coercivity (Hc) were determined directly from the M–H hysteresis loops50.
Electrode setup
A mixture of 20 mg of biosynthesized material with 10 mg PVDF, 10 mg graphite and 2 ml (DMF) solution were used for ink creation. To setup the ink working electrode, the ink was well mixed using ultrasonic device for 15 min at ambient condition. After which, the stainless steel and nickel substrates of 10×10 mm were treated by putting a drop of mentioned ink and letting the substrates dried at 65°C during overnight51.
Electrochemical study
Cyclic electrochemical voltametric (CV) and linear differential potential voltammetry (DPC) analysis were employed to maintain the sensing performance activities of the prepared electrodes toward the important pollutant such as IBP using a computer-controlled potentiostat (Metrohm Autolab, model: 87,070). Platinum (Pt) rod was placed as the counter electrode, where the working materials electrodes were the fabricated electrodes in a 100 mL aqueous solution contained 1% NaCl52, using a stock solution of 10000 ppm IBP. All electrochemical measurements were repeated three times using independently prepared electrodes. The relative standard deviation (RSD) was below 5%, indicating good reproducibility of the sensing response.
Mathematical model
The sensing mechanism is studied through two kinetic models: 1st order pseudo model and the 2nd order pseudo model. The 1st order kinetic equation represented by Eq. (4)53.
Where, Me, Md is sensing current (A) at concentration (D) and at equilibrium state correspondingly, and k1 sensing rate.
The 2nd order pseudo model was shown in Eq. (5)54:
Where, K2 is the sensing rate. Me and K2 were calculated from a slope and interception of plot.
Data availability
All datasets generated and/or analysed during the current study are available in the NCBI repository,(https://submit.ncbi.nlm.nih.gov) under accession numbers (PX879702 & PX879555).
References
Parolini, M. Toxicity of the Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) acetylsalicylic acid, paracetamol, diclofenac, ibuprofen and naproxen towards freshwater invertebrates: A review. Sci. Total Environ. 740, 140043 (2020).
Pires, P. et al. Non-steroidal anti-inflammatory drugs in the aquatic environment and bivalves: The state of the art. Toxics 12, 415 (2024).
Di Baccio, D. et al. Response of Lemna gibba L. to high and environmentally relevant concentrations of ibuprofen: Removal, metabolism and morpho-physiological traits for biomonitoring of emerging contaminants. Sci. Total Environ. 584, 363–373 (2017).
Trianda, Y., Adityosulindro, S. & Moersidik, S. S. Ibuprofen as an Emerging Contaminant of Concern: Occurrence in Southeast Asia Water Environment. in E3S Web of Conferences. (2024). EDP Sciences.
Godoi, F. G. et al. Review of the nonsteroidal anti-inflammatory drug consumption, occurrence, potential impacts on environmental health, and insights into regulatory decision-making Brazilian aquatic ecosystems. ACS omega 10, 26250–26265 (2025).
Mezzelani, M. et al. Long-term exposure of Mytilus galloprovincialis to diclofenac, Ibuprofen and Ketoprofen: Insights into bioavailability, biomarkers and transcriptomic changes. Chemosphere 198, 238–248 (2018).
Alqarni, A. M. Analytical methods for the determination of pharmaceuticals and personal care products in solid and liquid environmental matrices: A review. Molecules 29, 3900 (2024).
Całus-Makowska, K., Grosser, A. & Grobelak, A. Pharmaceutical contamination in wastewater treatment plants: occurrence, challenges in detection and insights on high-performance liquid chromatography as an effective analytical tool in environmental matrices—a review. Desalination Water Treat. 305, 129–154 (2023).
De Oliveira, M. et al. Pharmaceuticals residues and xenobiotics contaminants: occurrence, analytical techniques and sustainable alternatives for wastewater treatment. Sci. Total Environ. 705, 135568 (2020).
Harini, U. & Ponraj, C. Graphene-based electrochemical sensors for detecting environmental pollutants. J. Electrochem. Soc. 171, 027521 (2024).
He, Q. et al. Research on the construction of portable electrochemical sensors for environmental compounds quality monitoring. Mater. Today Adv. 17, 100340 (2023).
Cho, I.-H., Kim, D. H. & Park, S. Electrochemical biosensors: Perspective on functional nanomaterials for on-site analysis. Biomater. Res. 24, 6 (2020).
Tipsawat, P. et al. Magnetite (Fe3O4) nanoparticles: Synthesis, characterization and electrochemical properties. Appl. Surf. Sci. 446, 287–292 (2018).
Teymourian, H., Salimi, A. & Khezrian, S. Fe3O4 magnetic nanoparticles/reduced graphene oxide nanosheets as a novel electrochemical and bioeletrochemical sensing platform. Biosens. Bioelectron. 49, 1–8 (2013).
Mollarasouli, F. et al. Magnetic nanoparticles in developing electrochemical sensors for pharmaceutical and biomedical applications. Talanta 226, 122108 (2021).
Zokhtareh, R. et al. A novel sensing platform for electrochemical detection of metronidazole antibiotic based on green-synthesized magnetic Fe3O4 nanoparticles. Environ. Res. 216, 114643 (2023).
Moopantakath, J. et al. Bioactive molecules from haloarchaea: Scope and prospects for industrial and therapeutic applications. Front. Microbiol. 14, 1113540 (2023).
Rabani, G. et al. Extracellular synthesis of iron oxide nanoparticles using an extract of bacillus circulans: characterization and in vitro antioxidant activity. J. Chem. 2023, 4659034 (2023).
Berg, J. S. et al. Microbial diversity involved in iron and cryptic sulfur cycling in the ferruginous, low-sulfate waters of Lake Pavin. PLoS One 14, e0212787 (2019).
Hegazy, G. E. et al. In vitro dual (anticancer and antiviral) activity of the carotenoids produced by haloalkaliphilic archaeon Natrialba sp. M6. Sci. Rep. 10, 5986 (2020).
Salem, N. F. et al. Large scale production of superparamagnetic iron oxide nanoparticles by the haloarchaeon Halobiforma sp. N1 and their potential in localized hyperthermia cancer therapy. Nanotechnology 32, 09LT01 (2021).
Abdollahnia, M. et al. Exploring the potentials of halophilic prokaryotes from a solar saltern for synthesizing nanoparticles: The case of silver and selenium. PLoS One 15, e0229886 (2020).
Alvares, J. J. et al. Characterization of Mn3O4-MnO2 nanocomposites biosynthesized by cell lysate of Haloferax alexandrinus GUSF-1. J. Basic Microbiol. 63, 996–1006 (2023).
Zeth, K. et al. Iron-oxo clusters biomineralizing on protein surfaces: structural analysis of Halobacterium salinarum DpsA in its low-and high-iron states. Proc. Natl. Acad. Sci. 101, 13780–13785 (2004).
Ben-Shimon, S., Stein, D. & Zarivach, R. Current view of iron biomineralization in magnetotactic bacteria. J. Struct. Biol. X, 100052 (2021).
Zhao, B. et al. Natronolimnobius aegyptiacus sp. nov., an extremely halophilic alkalithermophilic archaeon isolated from the athalassohaline Wadi An Natrun, Egypt. Int. J. Syst. Evolut. Microbiol. 68, 498–506 (2018).
Begum, S. K. et al. Green synthesis of magnetite (Fe3O4) and hematite (Fe2O3) nanoparticles using Moringa oleifera and Psidium guajava leaf extracts for sustainable applications. Sci. Rep. 15, 36465 (2025).
Guo, H. & Barnard, A. S. Naturally occurring iron oxide nanoparticles: morphology, surface chemistry and environmental stability. J. Mater. Chem. A 1, 27–42 (2013).
Mondal, A. et al. Biogenic synthesis of metal/metal oxide nanostructured materials. Curr. Pharm. Biotechnol. 22, 1782–1793 (2021).
Sathiyanarayanan, G., Dineshkumar, K. & Yang, Y.-H. Microbial exopolysaccharide-mediated synthesis and stabilization of metal nanoparticles. Crit. Rev. Microbiol. 43, 731–752 (2017).
Poinern, G. E. J. et al. Banksia Ashbyi-engineered facile green synthesis of magnetite nanoparticles: Characterization, and determination of micro-strain, stress, and physical parameters by X-ray-based Williamson-Hall analysis. AIMS Mater. Sci. 11, 1096–1124 (2024).
Sidhu, A. K., Verma, N. & Kaushal, P. Role of biogenic capping agents in the synthesis of metallic nanoparticles and evaluation of their therapeutic potential. Front. Nanotechnol. 3, 801620 (2022).
Yew, Y. P. et al. Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: A review. Arab. J. Chem. 13, 2287–2308 (2020).
Oyebamiji, A. K., et al., A comprehensive review on mycosynthesis of nanoparticles, characteristics, applications, and limitations. Plasmonics. 1-19 (2025).
Mourdikoudis, S., Pallares, R. M. & Thanh, N. T. Characterization techniques for nanoparticles: comparison and complementarity upon studying nanoparticle properties. Nanoscale 10, 12871–12934 (2018).
Jayarambabu, N. et al. Biogenic synthesized Fe3O4 nanoparticles affect on growth parameter of maize (Zea mays L.). Dig. J. Nanomaterials Biostructures 13, 903–913 (2018).
Wang, J. et al. Insights into the crystallinity-dependent photochemical productions of reactive oxygen species from iron minerals. Environ. Sci. Technol. 58, 10623–10631 (2024).
He, Y. et al. Magnetite–graphene for the direct electrochemistry of hemoglobin and its biosensing application. Electrochim. Acta 56, 2471–2476 (2011).
Friebe, J. & Zacharias, P. Review of magnetic material degradation characteristics for the design of premagnetized inductors. IEEE Trans. Magn. 50, 40–48 (2013).
Goya, G. F. et al. Static and dynamic magnetic properties of spherical magnetite nanoparticles. J. Appl. Phys. 94, 3520–3528 (2003).
Kodama, R. Magnetic nanoparticles. J. Magn. Magn. Mater. 200, 359–372 (1999).
Singh, A. and V. Kumar, Iron oxide nanoparticles in biosensors, imaging and drug delivery applications—a complete tool, in Internet of Things and Big Data Applications: Recent Advances and Challenges. 2020, Springer. p. 243-252.
Youssef, M. E., Assessing the performance of novel and sustainable electrochemical sensor based on cobalt sulfide/upcycled reduced graphene oxide from plastic waste. Electrochem. Commun. 177 (2025).
Lesiak, B. et al. Surface study of Fe3O4 nanoparticles functionalized with biocompatible adsorbed molecules. Front. Chem. 7, 642 (2019).
Sandhya, J. & Kalaiselvam, S. Biogenic synthesis of magnetic iron oxide nanoparticles using inedible borassus flabellifer seed coat: characterization, antimicrobial, antioxidant activity and in vitro cytotoxicity analysis. Mater. Res. Express 7, 015045 (2020).
Radu, T. et al. X-ray photoelectron spectroscopic characterization of iron oxide nanoparticles. Appl. Surf. Sci. 405, 337–343 (2017).
Susithra, V. et al. Citrus sinensis assisted biogenic synthesis and physicochemical properties of Fe3O4 nanoparticles for antibacterial activity. Ceram. Int. 50, 10225–10231 (2024).
Vinayagam, R. et al. Structural characterization of green synthesized magnetic mesoporous Fe3O4NPs@ ME. Mater. Chem. Phys. 262, 124323 (2021).
Abouelkheir, S. S. & Mourad, M. M. Anxiety of microbially synthesized Fe3O4-SPIONs on embryonic/larval ontogeny in red tilapia (Oreochromis sp. Appl. Microbiol. Biotechnol. 109, 3 (2025).
Eldeeb, B. A., El-Raheem, W. M. A. & Elbeltagi, S. Green synthesis of biocompatible Fe3O4 magnetic nanoparticles using Citrus Sinensis peels extract for their biological activities and magnetic-hyperthermia applications. Sci. Rep. 13, 19000 (2023).
You, H. et al. Sustainable Production of Biomass-Derived Graphite and Graphene Conductive Inks from Biochar. Small 20, 2406669 (2024).
Nam, P. T. et al. Fabrication and characterization of a NiO–ZnO/PANI-CNTs composite for sensing of methanol in an aqueous environment. RSC Adv. 13, 36060–36070 (2023).
Elessawy, N. A. et al. Novel sodium alginate/polyvinylpyrrolidone/TiO2 nanocomposite for efficient removal of cationic dye from aqueous solution. Appl. Sci. 11, 9186 (2021).
Gouda, M. H., Elessawy, N. A. & Toghan, A. Novel crosslinked sulfonated PVA/PEO doped with phosphated titanium oxide nanotubes as effective green cation exchange membrane for direct borohydride fuel cells. Polymers 13, 2050 (2021).
Elnouby, M. S., Zahran, H. F. & Younes, S. M. Advanced Schiff base bio-electrode as sensitive sensor for ibuprofen in environmentally contaminant water. J. Mater. Sci.: Mater. Electron. 35, 2267 (2024).
Gaya, E. et al. Fe3O4-nanoparticle-modified sensor for the detection of dopamine, uric acid and ascorbic acid. Chemosensors 11, 79 (2023).
Sobahi, N. et al. Electrochemical sensing of H2O2 by employing a flexible Fe3O4/graphene/carbon cloth as working electrode. Materials 16, 2770 (2023).
Gai, K., et al. Preparation of Ag-Fe3O4 nanoparticles sensor and application in detection of methomyl. in E3S Web of Conferences. (2019). EDP Sciences.
Sarno, M. and E. Ponticorvo, Fe 3 O 4/Graphene Electrode for the ELectrochemical Detection of 4-Ntrophenol. CET J.-Chem. Eng. Transac. 79 (2020)
Abdelrazig, S. et al. Metabolic characterisation of Magnetospirillum gryphiswaldense MSR-1 using LC-MS-based metabolite profiling. RSC Adv. 10, 32548–32560 (2020).
Webster, D. P. et al. An arsenic-specific biosensor with genetically engineered Shewanella oneidensis in a bioelectrochemical system. Biosens. Bioelectron. 62, 320–324 (2014).
Dang, Q. et al. A Highly Sensitive and Rapid Enzyme Biosensor Based on AuNPs/CS@ cMWCNTS for Detecting Fusarium Acid. J. Electrochem. Soc. 171, 087521 (2024).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
G.E.H. H.O. and M.E.: Conceptualization, data curation, formal analysis, investigation, methodology, supervision, validation, visualization, writing-original draft, writing-review and editing. T.H.T.: Conceptualization, data curation, formal analysis. N.A.S and Y.R.A.: Supervision, writing-review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors approved for the publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hegazy, G.E., Oraby, H., Elnouby, M. et al. Haloalkaliphilic archaea-mediated green synthesis of superparamagnetic Fe₃O₄ nanoparticles for electrochemical detection of ibuprofen in saline environments. npj Clean Water 9, 30 (2026). https://doi.org/10.1038/s41545-026-00569-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41545-026-00569-4