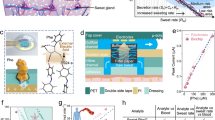
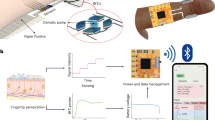
Abstract
Wearable non-invasive biosensors for the continuous monitoring of metabolites in sweat can detect a few analytes at sufficiently high concentrations, typically during vigorous exercise so as to generate sufficient quantity of the biofluid. Here we report the design and performance of a wearable electrochemical biosensor for the continuous analysis, in sweat during physical exercise and at rest, of trace levels of multiple metabolites and nutrients, including all essential amino acids and vitamins. The biosensor consists of graphene electrodes that can be repeatedly regenerated in situ, functionalized with metabolite-specific antibody-like molecularly imprinted polymers and redox-active reporter nanoparticles, and integrated with modules for iontophoresis-based sweat induction, microfluidic sweat sampling, signal processing and calibration, and wireless communication. In volunteers, the biosensor enabled the real-time monitoring of the intake of amino acids and their levels during physical exercise, as well as the assessment of the risk of metabolic syndrome (by correlating amino acid levels in serum and sweat). The monitoring of metabolites for the early identification of abnormal health conditions could facilitate applications in precision nutrition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. Source data for Figs. 4 and 5 and for Supplementary Figs. 36 and 39–41 are provided with this paper. All raw and analysed datasets generated during the study are available from the corresponding author on request. Source data are provided with this paper.
References
Trumbo, P., Schlicker, S., Yates, A. A. & Poos, M. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J. Am. Diet. Assoc. 102, 1621–1630 (2002).
Wu, G. Amino acids: metabolism, functions, and nutrition. Amino Acids 37, 1–17 (2009).
Lynch, C. J. & Adams, S. H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 10, 723–736 (2014).
Wang, T. J. et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 17, 448–453 (2011).
Mayers, J. R. et al. Elevation of circulating branched-chain amino acids is an early event in human pancreatic adenocarcinoma development. Nat. Med. 20, 1193–1198 (2014).
Kau, A. L., Ahern, P. P., Griffin, N. W., Goodman, A. L. & Gordon, J. I. Human nutrition, the gut microbiome and the immune system. Nature 474, 327–336 (2011).
Fernstrom, J. D. & Wurtman, R. J. Brain serotonin content: physiological dependence on plasma tryptophan levels. Science 173, 149–152 (1971).
Fernstrom, J. D. & Fernstrom, M. H. Tyrosine, phenylalanine, and catecholamine synthesis and function in the brain. J. Nutr. 137, 1539S–1547S (2007).
Dierckx, T. et al. The metabolic fingerprint of COVID-19 severity. Preprint at medRxiv (2020) https://doi.org/10.1101/2020.11.09.20228221
Grant, W. B. et al. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 12, 988–1006 (2020).
Belanger, M. J. et al. COVID-19 and disparities in nutrition and obesity. N. Engl. J. Med. 383, e69 (2020).
Stefan, N., Birkenfeld, A. L. & Schulze, M. B. Global pandemics interconnected—obesity, impaired metabolic health and COVID-19. Nat. Rev. Endocrinol. 17, 135–149 (2021).
Wishart, D. S. Emerging applications of metabolomics in drug discovery and precision medicine. Nat. Rev. Drug Discov. 15, 473–484 (2016).
Dettmer, K., Aronov, P. A. & Hammock, B. D. Mass spectrometry-based metabolomics. Mass Spectrom. Rev. 26, 51–78 (2007).
Ray, T. R. et al. Bio-integrated wearable systems: A comprehensive review. Chem. Rev. 119, 5461–5533 (2019).
Hollander, J. E. & Carr, B. G. Virtually perfect? Telemedicine for COVID-19. N. Engl. J. Med. 382, 1679–1681 (2020).
Niu, S. et al. A wireless body area sensor network based on stretchable passive tags. Nat. Electron. 2, 361–368 (2019).
Meng, K. et al. A wireless textile-based sensor system for self-powered personalized health care. Matter 2, 896–907 (2020).
Wang, C. et al. Monitoring of the central blood pressure waveform via a conformal ultrasonic device. Nat. Biomed. Eng. 2, 687–695 (2018).
Ni, X. et al. Automated, multiparametric monitoring of respiratory biomarkers and vital signs in clinical and home settings for COVID-19 patients. Proc. Natl Acad. Sci. USA 118, e2026610118 (2021).
Jeong, H. et al. Differential cardiopulmonary monitoring system for artifact-canceled physiological tracking of athletes, workers, and COVID-19 patients. Sci. Adv. 7, eabg3092 (2021).
Sempionatto, J. R. et al. An epidermal patch for the simultaneous monitoring of haemodynamic and metabolic biomarkers. Nat. Biomed. Eng. 5, 737–748 (2021).
Xu, C., Yang, Y. & Gao, W. Skin-interfaced sensors in digital medicine: from materials to applications. Matter 2, 1414–1445 (2020).
Heikenfeld, J. et al. Accessing analytes in biofluids for peripheral biochemical monitoring. Nat. Biotechnol. 37, 407–419 (2019).
Bariya, M., Nyein, H. Y. Y. & Javey, A. Wearable sweat sensors. Nat. Electron. 1, 160–171 (2018).
Yang, Y. & Gao, W. Wearable and flexible electronics for continuous molecular monitoring. Chem. Soc. Rev. 48, 1465–1491 (2019).
Kim, J., Campbell, A. S., de Ávila, B. E.-F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406 (2019).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Bandodkar, A. J. et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 5, eaav3294 (2019).
Emaminejad, S. et al. Autonomous sweat extraction and analysis applied to cystic fibrosis and glucose monitoring using a fully integrated wearable platform. Proc. Natl Acad. Sci. USA 114, 4625–4630 (2017).
Torrente-Rodríguez, R. M. et al. Investigation of cortisol dynamics in human sweat using a graphene-based wireless mHealth System. Matter 2, 921–937 (2020).
Koh, A. et al. A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci. Transl. Med. 8, 366ra165 (2016).
Ray, T. R. et al. Soft, skin-interfaced sweat stickers for cystic fibrosis diagnosis and management. Sci. Transl. Med. 13, eabd8109 (2021).
Lee, H. et al. A graphene-based electrochemical device with thermoresponsive microneedles for diabetes monitoring and therapy. Nat. Nanotech. 11, 566–572 (2016).
Lee, H. et al. Wearable/disposable sweat-based glucose monitoring device with multistage transdermal drug delivery module. Sci. Adv. 3, e1601314 (2017).
Kim, J. et al. Noninvasive alcohol monitoring using a wearable tattoo-based iontophoretic-biosensing system. ACS Sens. 1, 1011–1019 (2016).
Kim, J. et al. A skin‐interfaced, miniaturized microfluidic analysis and delivery system for colorimetric measurements of nutrients in sweat and supply of vitamins through the skin. Adv. Sci. 9, 2103331 (2022).
Sempionatto, J. R. et al. Wearable and mobile sensors for personalized nutrition. ACS Sens. 6, 1745–1760 (2021).
Yang, Y. et al. A laser-engraved wearable sensor for sensitive detection of uric acid and tyrosine in sweat. Nat. Biotechnol. 38, 217–224 (2020).
Yu, Y. et al. Biofuel-powered soft electronic skin with multiplexed and wireless sensing for human-machine interfaces. Sci. Robot. 5, eaaz7946 (2020).
Wang, J. Electrochemical biosensors: towards point-of-care cancer diagnostics. Biosens. Bioelectron. 21, 1887–1892 (2006).
Uzun, L. & Turner, A. P. Molecularly-imprinted polymer sensors: realising their potential. Biosens. Bioelectron. 76, 131–144 (2016).
Parlak, O., Keene, S. T., Marais, A., Curto, V. F. & Salleo, A. Molecularly selective nanoporous membrane-based wearable organic electrochemical device for noninvasive cortisol sensing. Sci. Adv. 4, eaar2904 (2018).
Kaspar, H., Dettmer, K., Gronwald, W. & Oefner, P. J. Automated GC–MS analysis of free amino acids in biological fluids. J. Chromatogr. B 870, 222–232 (2008).
Riedl, B., Nischik, M., Birklein, F., Neundörfer, B. & Handwerker, H. O. Spatial extension of sudomotor axon reflex sweating in human skin. J. Auton. Nerv. Syst. 69, 83–88 (1998).
Newsholme, E. A. & Blomstrand, E. Branched-chain amino acids and central fatigue. J. Nutr. 136, 274S–276S (2006).
Ahlqvist, E. et al. Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol. 6, 361–369 (2018).
Layman, D. K. & Walker, D. A. Potential importance of leucine in treatment of obesity and the metabolic syndrome. J. Nutr. 136, 319S–323S (2006).
Kitson, F. G., Larsen, B. S. & McEwen, C. N. Mass Spectrometry: A Practical Guide, pp. 3–23 (Academic Press, San Diego, 1996).
Torrente-Rodríguez, R. M. et al. Rapid endoglin determination in serum samples using an amperometric magneto-actuated disposable immunosensing platform. J. Pharm. Biomed. 129, 288–293 (2016).
Acknowledgements
This project was supported by the National Institutes of Health grant R01HL155815, Office of Naval Research grants N00014-21-1-2483 and N00014-21-1-2845, the Translational Research Institute for Space Health through NASA NNX16AO69A, NASA Cooperative Agreement 80NSSC20M0167, High Impact Pilot Research Award T31IP1666 and grant R01RG3746 from the Tobacco-Related Disease Research Program, Caltech-City of Hope Biomedical Initiative Pilot Grant and the Rothenberg Innovation Initiative Program at California Institute of Technology. J.T. was supported by the National Science Scholarship (NSS) from the Agency of Science Technology and Research (A*STAR) Singapore. We gratefully acknowledge critical support and infrastructure provided for this work by the Kavli Nanoscience Institute at Caltech. This project benefitted from the use of instrumentation made available by the Caltech Environmental Analysis Center, and we gratefully acknowledge support on GC–MS from N. Dalleska. We also gratefully acknowledge Z. Wang for the contribution to mobile app development, R. M. Torrente-Rodríguez for the insulin assay optimization and S. Bao for the valuable inputs.
Author information
Authors and Affiliations
Contributions
W.G., M.W., Y.Y. and J.M. initiated the concept and designed the studies; W.G. supervised the work; M.W., Y.Y. and J.M. led the experiments and collected the overall data; Y.S., J.T., D.M., C.Y. and C.X. contributed to sensor characterization, validation and sample analysis; N.H. contributed to the signal processing and app development. J.S.M., T.K.H. and Z.L. contributed to the design of the human studies. W.G., M.W., Y.Y. and J.M. co-wrote the paper. All authors contributed to the data analysis and provided feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary notes, figures, tables and references.
Supplementary Video 1 (download MP4 )
On-body demonstration with the designed sensing patch and app.
Supplementary Video 2 (download MP4 )
Simulation and validation of microfluidic sweat refreshing.
Supplementary Video 3 (download MP4 )
Iontophoresis-induced microfluidic sweat sampling at rest.
Source data
Source data (download XLSX )
Source data for Figs. 4 and 5 and Supplementary Figs. 36 and 39–41.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Yang, Y., Min, J. et al. A wearable electrochemical biosensor for the monitoring of metabolites and nutrients. Nat. Biomed. Eng 6, 1225–1235 (2022). https://doi.org/10.1038/s41551-022-00916-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41551-022-00916-z
This article is cited by
-
Predictive design of stretchable electrodes with strain-insensitive performance via robotics- and machine learning-integrated workflow
Nature Communications (2026)
-
Challenges and opportunities of wearable molecular sensors in endocrinology and metabolism
Nature Reviews Endocrinology (2026)
-
Universal modulus-free transfer of scalable laser-induced graphene for electronic skins
Nature Communications (2026)
-
Molecularly imprinted polymer-based electrochemical sensors for amino acid detection: towards wearable sensing
npj Biosensing (2026)
-
Noninvasive On-Skin Biosensors for Monitoring Diabetes Mellitus
Nano-Micro Letters (2026)