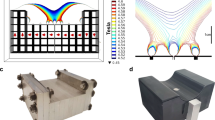
Abstract
Serial assessment of the biomechanical properties of tissues can be used to aid the early detection and management of pathophysiological conditions, to track the evolution of lesions and to evaluate the progress of rehabilitation. However, current methods are invasive, can be used only for short-term measurements, or have insufficient penetration depth or spatial resolution. Here we describe a stretchable ultrasonic array for performing serial non-invasive elastographic measurements of tissues up to 4 cm beneath the skin at a spatial resolution of 0.5 mm. The array conforms to human skin and acoustically couples with it, allowing for accurate elastographic imaging, which we validated via magnetic resonance elastography. We used the device to map three-dimensional distributions of the Young’s modulus of tissues ex vivo, to detect microstructural damage in the muscles of volunteers before the onset of soreness and to monitor the dynamic recovery process of muscle injuries during physiotherapies. The technology may facilitate the diagnosis and treatment of diseases affecting tissue biomechanics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the paper and its Supplementary Information. The data generated in this study are available from figshare at https://doi.org/10.6084/m9.figshare.22197139.v1. Source data are provided with this paper.
References
Fung, Y.-C. & Skalak, R. Biomechanics. Mechanical properties of living tissues. J. Appl. Mech. 49, 464 (1982).
Habibi, H. A. et al. In vivo assessment of placental elasticity in intrauterine growth restriction by shear-wave elastography. Eur. J. Radiol. 97, 16–20 (2017).
Huh, J. H. et al. Obesity is more closely related with hepatic steatosis and fibrosis measured by transient elastography than metabolic health status. Metabolism 66, 23–31 (2017).
Lee, G. Y. & Lim, C. T. Biomechanics approaches to studying human diseases. Trends Biotechnol. 25, 111–118 (2007).
Mehrara, E., Forssell-Aronsson, E., Ahlman, H. & Bernhardt, P. Specific growth rate versus doubling time for quantitative characterization of tumor growth rate. Cancer Res. 67, 3970–3975 (2007).
Wellman, P., Howe, R. D., Dalton, E. & Kern, K. A. Breast Tissue Stiffness in Compression is Correlated to Histological Diagnosis (Harvard BioRobotics, 1999).
Wang, Y. et al. Assessment of tumor stiffness with shear wave elastography in a human prostate cancer xenograft implantation model. J. Ultrasound Med. 36, 955–963 (2017).
Furukawa, H., Iwata, R. & Moriyama, N. Growth rate of pancreatic adenocarcinoma: initial clinical experience. Pancreas 22, 366–369 (2001).
Hasegawa, M. et al. Growth rate of small lung cancers detected on mass CT screening. Br. J. Radiol. 73, 1252–1259 (2000).
Winer-Muram, H. T. et al. Volumetric growth rate of stage I lung cancer prior to treatment: serial CT scanning. Radiology 223, 798–805 (2002).
Nakajima, T. et al. Simple tumor profile chart based on cell kinetic parameters and histologic grade is useful for estimating the natural growth rate of hepatocellular carcinoma. Hum. Pathol. 33, 92–99 (2002).
Saito, Y. et al. Multiple regression analysis for assessing the growth of small hepatocellular carcinoma: the MIB-1 labeling index is the most effective parameter. J. Gastroenterol. 33, 229–235 (1998).
Katyan, A., Mittal, M. K., Mani, C. & Mandal, A. K. Strain wave elastography in response assessment to neo-adjuvant chemotherapy in patients with locally advanced breast cancer. Br. J. Radiol. 92, 20180515 (2019).
Botar-Jid, C. et al. The contribution of ultrasonography and sonoelastography in assessment of myositis. Med. Ultrason. 12, 120–126 (2010).
Brandenburg, J. E. et al. Quantifying passive muscle stiffness in children with and without cerebral palsy using ultrasound shear wave elastography. Dev. Med. Child Neurol. 58, 1288–1294 (2016).
Gao, J. et al. Quantitative ultrasound imaging to assess the biceps brachii muscle in chronic post-stroke spasticity: preliminary observation. Ultrasound Med. Biol. 44, 1931–1940 (2018).
Illomei, G., Spinicci, G., Locci, E. & Marrosu, M. Muscle elastography: a new imaging technique for multiple sclerosis spasticity measurement. Neurol. Sci. 38, 433–439 (2017).
Lv, F. et al. Muscle crush injury of extremity: quantitative elastography with supersonic shear imaging. Ultrasound Med. Biol. 38, 795–802 (2012).
Lacourpaille, L. et al. Time-course effect of exercise-induced muscle damage on localized muscle mechanical properties assessed using elastography. Acta Physiol. 211, 135–146 (2014).
Drakonaki, E. Ultrasound elastography for imaging tendons and muscles. J. Ultrason. 12, 214 (2012).
Brandenburg, J. E. et al. Ultrasound elastography: the new frontier in direct measurement of muscle stiffness. Arch. Phys. Med. Rehabil. 95, 2207–2219 (2014).
Gao, J., Du, L.-J., He, W., Li, S. & Cheng, L.-G. Ultrasound strain elastography in assessment of muscle stiffness in acute levodopa challenge test: a feasibility study. Ultrasound Med. Biol. 42, 1084–1089 (2016).
Wen, H., Bennett, E., Epstein, N. & Plehn, J. Magnetic resonance imaging assessment of myocardial elastic modulus and viscosity using displacement imaging and phase-contrast velocity mapping. Magn. Reason. Med. 54, 538–548 (2005).
Mercuri, M. Noninvasive imaging protocols to detect and monitor carotid atherosclerosis progression. Am. J. Hypertens. 7, 23S–29S (1994).
Qian, X. et al. Multi-functional ultrasonic micro-elastography imaging system. Sci. Rep. 7, 1230 (2017).
Montaldo, G., Tanter, M., Bercoff, J., Benech, N. & Fink, M. Coherent plane-wave compounding for very high frame rate ultrasonography and transient elastography. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 56, 489–506 (2009).
Francois Dord, J. et al. in Ultrasound Elastography for Biomedical Applications and Medicine (eds Nenadic, I. Z. et al.) 129–142 (Wiley, 2018).
Goenezen, S. et al. Linear and nonlinear elastic modulus imaging: an application to breast cancer diagnosis. IEEE Trans. Med. Imaging 31, 1628–1637 (2012).
Pellot-Barakat, C., Frouin, F., Insana, M. F. & Herment, A. Ultrasound elastography based on multiscale estimations of regularized displacement fields. IEEE Trans. Med. Imaging 23, 153–163 (2004).
Jiang, J. & Hall, T. J. A coupled subsample displacement estimation method for ultrasound-based strain elastography. Phys. Med. Biol. 60, 8347 (2015).
Kallel, F. & Ophir, J. A least-squares strain estimator for elastography. Ultrason. Imaging 19, 195–208 (1997).
Cloostermans, M. & Thijssen, J. A beam corrected estimation of the frequency dependent attenuation of biological tissues from backscattered ultrasound. Ultrason. Imaging 5, 136–147 (1983).
Bader, K. B., Crowe, M. J., Raymond, J. L. & Holland, C. K. Effect of frequency-dependent attenuation on predicted histotripsy waveforms in tissue-mimicking phantoms. Ultrasound Med. Biol. 42, 1701–1705 (2016).
Shung, K. K. Diagnostic Ultrasound: Imaging and Blood Flow Measurements (CRC Press, 2015).
Hu, H. et al. Stretchable ultrasonic transducer arrays for three-dimensional imaging on complex surfaces. Sci. Adv. 4, eaar3979 (2018).
Wang, C. et al. Monitoring of the central blood pressure waveform via a conformal ultrasonic device. Nat. Biomed. Eng. 2, 687–695 (2018).
Mi, X., Qin, L., Liao, Q. & Wang, L. Electromechanical coupling coefficient and acoustic impedance of 1–1–3 piezoelectric composites. Ceram. Int. 43, 7374–7377 (2017).
Haller, M. & Khuri-Yakub, B. Micromachined 1–3 composites for ultrasonic air transducers. Rev. Sci. Instrum. 65, 2095–2098 (1994).
Chen, R. et al. Eco-friendly highly sensitive transducers based on a new KNN-NTK-FM lead-free piezoelectric ceramic for high-frequency biomedical ultrasonic imaging applications. IEEE Trans. Biomed. Eng. 66, 1580–1587 (2018).
Arumugam, V., Naresh, M. D. & Sanjeevi, R. Effect of strain-rate on the fracture-behavior of skin. J. Biosci. 19, 307–313 (1994).
Wang, Y., Bayer, M., Jiang, J. & Hall, T. J. Large-strain 3-D in vivo breast ultrasound strain elastography using a multi-compression strategy and a whole-breast scanning system. Ultrasound Med. Biol. 45, 3145–3159 (2019).
Céspedes, I. & Ophir, J. Reduction of image noise in elastography. Ultrason. Imaging 15, 89–102 (1993).
Varghese, T. & Ophir, J. An analysis of elastographic contrast-to-noise ratio. Ultrasound Med. Biol. 24, 915–924 (1998).
Kennedy, K. M. et al. Quantitative micro-elastography: imaging of tissue elasticity using compression optical coherence elastography. Sci. Rep. 5, 15538 (2015).
Ding, H. et al. Ultrasonic imaging of muscle-like phantoms using bimorph pmuts toward wearable muscle disorder diagnostics. In 2018 IEEE Micro Electro Mechanical Systems (MEMS) (eds Despont, M. & Ducrée, J.) 396–399 (IEEE, 2018).
Dietrich, C. F. et al. Strain elastography—how to do it? Ultrasound Int. Open 3, E137 (2017).
Taylor, K. et al. Ultrasound elastography as an adjuvant to conventional ultrasound in the preoperative assessment of axillary lymph nodes in suspected breast cancer: a pilot study. Clin. Radiol. 66, 1064–1071 (2011).
DeWall, R. J. Ultrasound elastography: principles, techniques, and clinical applications. Crit. Rev. Biomed. Eng. 41, 1–19 (2013).
Krouskop, T. A., Wheeler, T. M., Kallel, F., Garra, B. S. & Hall, T. Elastic moduli of breast and prostate tissues under compression. Ultrason. Imaging 20, 260–274 (1998).
Lopata, R. G. et al. Performance evaluation of methods for two-dimensional displacement and strain estimation using ultrasound radio frequency data. Ultrasound Med. Biol. 35, 796–812 (2009).
Hÿtch, M., Snoeck, E. & Kilaas, R. Quantitative measurement of displacement and strain fields from HREM micrographs. Ultramicroscopy 74, 131–146 (1998).
Papadacci, C., Bunting, E. A. & Konofagou, E. E. 3D quasi-static ultrasound elastography with plane wave in vivo. IEEE Trans. Med. Imaging 36, 357–365 (2016).
Hayakawa, M., Hatano, T., Tsuji, A., Nakajima, F. & Ogawa, Y. Patients with renal cysts associated with renal cell carcinoma and the clinical implications of cyst puncture: a study of 223 cases. Urology 47, 643–646 (1996).
Sigrist, R. M., Liau, J., El Kaffas, A., Chammas, M. C. & Willmann, J. K. Ultrasound elastography: review of techniques and clinical applications. Theranostics 7, 1303 (2017).
Oberai, A. A., Gokhale, N. H. & Feijóo, G. R. Solution of inverse problems in elasticity imaging using the adjoint method. Inverse Prob. 19, 297 (2003).
Ferreira, E. R., Oberai, A. A. & Barbone, P. E. Uniqueness of the elastography inverse problem for incompressible nonlinear planar hyperelasticity. Inverse Prob. 28, 065008 (2012).
Song, D., Hugenberg, N. & Oberai, A. A. Three-dimensional traction microscopy with a fiber-based constitutive model. Comput. Methods Appl. Mech. Eng. 357, 112579 (2019).
Yu, X. et al. Needle-shaped ultrathin piezoelectric microsystem for guided tissue targeting via mechanical sensing. Nat. Biomed. Eng. 2, 165–172 (2018).
Risholm, P., Ross, J., Washko, G. R. & Wells, W. M. Probabilistic elastography: estimating lung elasticity. In Biennial International Conference on Information Processing in Medical Imaging (eds Székely, G. & Hahn, H. K.) 699–710 (Springer, 2011).
Gokhale, N. H., Barbone, P. E. & Oberai, A. A. Solution of the nonlinear elasticity imaging inverse problem: the compressible case. Inverse Prob. 24, 045010 (2008).
Brouwer, I., Ustin, J., Bentley, L., Dhruv, A. & Tendick, F. in Medicine Meets Virtual Reality (eds Westwood, J. D. et al.) 69–74 (IOS Press, 2001).
Van Houten, E. E., Doyley, M. M., Kennedy, F. E., Weaver, J. B. & Paulsen, K. D. Initial in vivo experience with steady-state subzone-based MR elastography of the human breast. J. Magn. Reson. 17, 72–85 (2003).
Xie, Y., Thomas, L., Hug, F., Johnston, V. & Coombes, B. K. Quantifying cervical and axioscapular muscle stiffness using shear wave elastography. J. Electromyogr. Kinesiol. 48, 94–102 (2019).
Van Sligtenhorst, C., Cronin, D. S. & Brodland, G. W. High strain rate compressive properties of bovine muscle tissue determined using a split Hopkinson bar apparatus. J. Biomech. 39, 1852–1858 (2006).
Ishiwatari, N., Fukuoka, M. & Sakai, N. Effect of protein denaturation degree on texture and water state of cooked meat. J. Food Eng. 117, 361–369 (2013).
Cipryan, L. & Litschmannova, M. Intra-day and inter-day reliability of heart rate variability measurement. J. Sports Sci. 31, 150–158 (2013).
Koo, T. K. & Li, M. Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 15, 155–163 (2016).
Sorbie, G. G. et al. Intra-session and inter-day reliability of the Myon 320 electromyography system during sub-maximal contractions. Front. Physiol. 9, 309 (2018).
Pearcey, G. E. et al. Foam rolling for delayed-onset muscle soreness and recovery of dynamic performance measures. J. Athl. Train. 50, 5–13 (2015).
Hotfiel, T. et al. Advances in delayed-onset muscle soreness (DOMS): part I: pathogenesis and diagnostics. Sportverletz. Sportschaden 32, 243–250 (2018).
Kishimoto, K., Sakuraba, K., Kubota, A. & Fujita, S. The effect of concentric and eccentric exercise on muscle hardness. Juntendo Med. J. 64, 371–378 (2018).
Niitsu, M., Michizaki, A., Endo, A., Takei, H. & Yanagisawa, O. Muscle hardness measurement by using ultrasound elastography: a feasibility study. Acta Radiol. 52, 99–105 (2011).
Murayama, M., Nosaka, K., Inami, T., Shima, N. & Yoneda, T. Biceps brachii muscle hardness assessed by a push-in meter in comparison to ultrasound strain elastography. Sci. Rep. 10, 20308 (2020).
Peake, J. M., Neubauer, O., Della Gatta, P. A. & Nosaka, K. Muscle damage and inflammation during recovery from exercise. J. Appl. Physiol. 122, 559–570 (2017).
Nelson, N. Delayed onset muscle soreness: is massage effective? J. Bodyw. Mov. Ther. 17, 475–482 (2013).
Lippold, O. The tremor in fatigue. Ciba Found. Symp. 82, 234–248 (1981).
Lee, S.-Y. & Jo, M.-E. Comparison of maximum voluntary isometric contraction of the biceps on various posture and respiration conditions for normalization of electromyography data. J. Phys. Ther. Sci. 28, 3007–3010 (2016).
Clarkson, P. M., Nosaka, K. & Braun, B. Muscle function after exercise-induced muscle damage and rapid adaptation. Med. Sci. Sports Exerc. 24, 512–520 (1992).
Huang, Z. L. et al. Three-dimensional integrated stretchable electronics. Nat. Electron. 1, 473–480 (2018).
Yang, Y. et al. Stretchable nanolayered thermoelectric energy harvester on complex and dynamic surfaces. Nano Lett. 20, 4445–4453 (2020).
Kwok, K. W., Chan, H. & Choy, C. L. Lead zirconate titanate/poly (vinylidene fluoride-trifluoroethylene) 1–3 composites for ultrasonic transducer applications. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 46, 626–637 (1999).
Madsen, E. L., Hobson, M. A., Shi, H., Varghese, T. & Frank, G. R. Tissue-mimicking agar/gelatin materials for use in heterogeneous elastography phantoms. Phys. Med. Biol. 50, 5597 (2005).
Li, X. et al. 80-MHz intravascular ultrasound transducer using PMN-PT free-standing film. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 58, 2281–2288 (2011).
Chen, Z. et al. High-frequency ultrasonic imaging with lead-free (Na, K)(Nb, Ta) O3 single crystal. Ultrason. Imaging 39, 348–356 (2017).
Webster, R., Button, T., Meggs, C., MacLennan, D. & Cochran, S. P3K-5 passive materials for high frequency ultrasound components. In 2007 IEEE Ultrasonics Symposium Proceedings (ed. Passini Yuhas, M.) 1925–1928 (IEEE, 2007).
Sayers, C. & Tait, C. Ultrasonic properties of transducer backings. Ultrasonics 22, 57–60 (1984).
Varghese, T. & Ophir, J. A theoretical framework for performance characterization of elastography: the strain filter. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 44, 164–172 (1997).
Tang, A., Cloutier, G., Szeverenyi, N. M. & Sirlin, C. B. Ultrasound elastography and MR elastography for assessing liver fibrosis: part 2, diagnostic performance, confounders, and future directions. Am. J. Roentgenol. 205, 33–40 (2015).
Acknowledgements
We thank Z. Wu for the guidance on ultrasonic imaging algorithm and data processing, N. Szeverenyi for the help with MRE testing, S. Sinha for the discussions about eccentric exercise experiments, Y. Wang and R. Kou for the help with Young’s modulus characterizations by a mechanical apparatus, and S. Xiang for the feedback on paper preparation. The material is based on research sponsored by Air Force Research Laboratory (AFRL) under agreement number FA8650-18-2-5402. The US Government is authorized to reproduce and distribute reprints for government purposes notwithstanding any copyright notation thereon. The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsements, either expressed or implied, of Air Force Research Laboratory or the US Government. This work was partially supported by the National Institutes of Health (NIH) grants 1R21EB025521-01, 1R21EB027303-01A1, 3R21EB027303-02S1 and 1R01EB033464-01, and the Center for Wearable Sensors at the University of California, San Diego. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
Ho.H., Y.M., X.G., D.S. and S.X. designed the research. Ho.H., Y.M., M.L. and Ha.H. performed the experiments. Y.M., X.G. and D.S. performed the data processing and simulations. Ho.H., Y.M. and X.G. analysed the data. Ho.H., D.S., X.G. and S.X. wrote the paper. All authors provided constructive and valuable feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Antoine Nordez, Xiaoning Jiang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Simulation results of uniform and nonuniform compressions.
(a) A synthetic specimen with an inclusion that has a shear modulus 10 times higher than that of the surrounding matrix. The synthetic “measured” displacement fields based on (b) uniform compression and (c) non-uniform compression on the specimen. (d) and (e) show the strain distributions from uniform and non-uniform compression, respectively. The strain distributions vary upon different applied loads, which indicates that the strain-based elastography is qualitative only reflecting a relative stiffness of each component. The reconstructed modulus distributions obtained from solving inverse elasticity problems based on (f) uniform compression and (g) non-uniform compression. (h) Quantitative analysis of modulus contrasts and their deviations from the ground truth in (a).
Extended Data Fig. 2 Validating the device’s reliability on six subjects.
(a) Strain mapping results of upper arms of six subjects for the evaluation of the intra-session reliability. (b) Strain mapping results of upper arms of six subjects for the evaluation of the inter-day reliability.
Extended Data Fig. 3 Serial surveillance of delayed-onset muscle soreness in multiple subjects.
Serial monitoring results of normalized modulus contrast of the biceps brachii muscle before and after the eccentric exercise. All six subjects did two rounds of experiments. In the first round, all subjects took the natural recovery after doing the exercise. In the second round, subjects in (a), (b) took natural recovery, subjects in (c), (d) took massotherapy, and subjects in (e), (f) took hyperthermia. For each test, we mapped the modulus distribution of the muscle. Then, we calculated the mean and standard deviations of the biceps brachii area. In each physiotherapy session, data are normalized by the modulus contrast of the biceps brachii muscle before exercise.
Extended Data Fig. 4 Validation by a clinical standard method on a larger sample size.
(a) Changes in the maximal voluntary contraction torque (black) and normalized modulus contrast (blue) of the biceps brachii muscle measured by the stretchable ultrasonic array before and after the eccentric exercise of 16 subjects. The points and error bars of the blue curves indicate the mean and standard deviation of modulus contrast of the biceps brachii muscle of every test. For each test, we mapped the modulus distribution of the muscle. Then, we calculated the mean and standard deviations of the biceps brachii area. In each physiotherapy session, data were normalized by the modulus contrast of the biceps brachii muscle before exercise. As a clinical standard approach, the maximal voluntary contraction can validate the muscle strength and muscle damage. (b) P-values of the clinical standard method and the stretchable ultrasonic array, which were calculated by single-sided paired t-test. Error bars are standard deviations of the data of 16 subjects (n = 16).
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes, Figures, Tables, References and Video.
Supplementary type (download MP4 )
Simulation illustrating the principle of ultrasound elastography.
Source data
Source Data for Fig. 1 (download XLSX )
Source data.
Source Data for Fig. 2 (download XLSX )
Source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, H., Ma, Y., Gao, X. et al. Stretchable ultrasonic arrays for the three-dimensional mapping of the modulus of deep tissue. Nat. Biomed. Eng 7, 1321–1334 (2023). https://doi.org/10.1038/s41551-023-01038-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41551-023-01038-w
This article is cited by
-
Mechanomedicine
Nature Reviews Bioengineering (2026)
-
Wearable Ultrasound Devices for Therapeutic Applications
Nano-Micro Letters (2026)
-
Silicon nanocolumn-based disposable and flexible ultrasound patches
Nature Communications (2025)
-
Wearable flexible ultrasound microneedle patch for cancer immunotherapy
Nature Communications (2025)
-
Ultrasound brain stimulation technologies for targeted therapeutics
Nature Electronics (2025)