Abstract
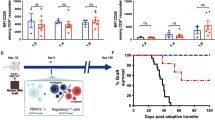
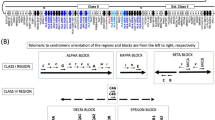
Immune rejection caused by mismatches in human leucocyte antigens (HLAs) remains a major obstacle to the success of allogeneic cell therapies. Current strategies for the generation of ‘universal’ immune-compatible cells, particularly the editing of HLA class I (HLA-I) genes or the modulation of proteins that inhibit natural killer cells, often result in genomic instability or cellular cytotoxicity. Here we show that a β2-microglobulin super-enhancer (B2M-SE) that is responsive to interferon-γ is a critical regulator of the expression of HLA-I on mesenchymal stromal cells (MSCs). Targeted epigenetic repression of B2M-SE in MSCs reduced the surface expression of HLA-I below the threshold required to activate allogenic T cells while maintaining levels sufficient to evade cytotoxicity mediated by natural killer cells. In a humanized mouse model, the epigenetically edited MSCs demonstrated improved survival by evading the immune system, allowing them to exert enhanced therapeutic effects on LPS-induced acute lung injury. Targeted epigenetic repression of B2M-SE may facilitate the development of off-the-shelf cell sources for allogeneic cell therapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the article and its Supplementary Information. The RNA-seq, ChIP–seq and ATAC–seq datasets generated during the study are available from the Genome Sequence Archive for Human (https://ngdc.cncb.ac.cn/gsa-human, HRA003235). The raw and analysed datasets generated during the study are available for research purposes from the corresponding authors on reasonable request. Source data are provided with this paper.
References
Global Observatory on Donation and Transplantation. Organ Donation and Transplantation Activities 2019 Report (WHO–ONT, 2021); http://www.transplant-observatory.org/wp-content/uploads/2021/06/GODT2019-data_web_updated-June-2021.pdf
Lanza, R., Russell, D. W. & Nagy, A. Engineering universal cells that evade immune detection. Nat. Rev. Immunol. 19, 723–733 (2019).
Yamanaka, S. Pluripotent stem cell-based cell therapy—promise and challenges. Cell Stem Cell 27, 523–531 (2020).
Kobayashi, K. S. & van den Elsen, P. J. NLRC5: a key regulator of MHC class I-dependent immune responses. Nat. Rev. Immunol. 12, 813–820 (2012).
Maggs, L., Sadagopan, A., Moghaddam, A. S. & Ferrone, S. HLA class I antigen processing machinery defects in antitumor immunity and immunotherapy. Trends Cancer 7, 1089–1101 (2021).
Vyas, J. M., Van der Veen, A. G. & Ploegh, H. L. The known unknowns of antigen processing and presentation. Nat. Rev. Immunol. 8, 607–618 (2008).
Blander, J. M. Regulation of the cell biology of antigen cross-presentation. Annu. Rev. Immunol. 36, 717–753 (2018).
Poggi, A. & Zocchi, M. R. Antigen presenting cells and stromal cells trigger human natural killer lymphocytes to autoreactivity: evidence for the involvement of natural cytotoxicity receptors (NCR) and NKG2D. Clin. Dev. Immunol. 13, 325–336 (2006).
Martinet, L. & Smyth, M. J. Balancing natural killer cell activation through paired receptors. Nat. Rev. Immunol. 15, 243–254 (2015).
Deuse, T. et al. Hypoimmunogenic derivatives of induced pluripotent stem cells evade immune rejection in fully immunocompetent allogeneic recipients. Nat. Biotechnol. 37, 252–258 (2019).
Han, X. et al. Generation of hypoimmunogenic human pluripotent stem cells. Proc Natl Acad. Sci. USA 116, 10441–10446 (2019).
Gornalusse, G. G. et al. HLA-E-expressing pluripotent stem cells escape allogeneic responses and lysis by NK cells. Nat. Biotechnol. 35, 765–772 (2017).
Wang, B. et al. Generation of hypoimmunogenic T cells from genetically engineered allogeneic human induced pluripotent stem cells. Nat. Biomed. Eng. 5, 429–440 (2021).
Jo, S. et al. Endowing universal CAR T-cell with immune-evasive properties using TALEN-gene editing. Nat. Commun. 13, 3453 (2022).
Shi, L. et al. Generation of hypoimmunogenic human pluripotent stem cells via expression of membrane-bound and secreted β2m-HLA-G fusion proteins. Stem Cells 38, 1423–1437 (2020).
Parent, A. V. et al. Selective deletion of human leukocyte antigens protects stem cell-derived islets from immune rejection. Cell Rep. 36, 109538 (2021).
Xu, H. et al. Targeted disruption of HLA genes via CRISPR–Cas9 generates iPSCs with enhanced immune compatibility. Cell Stem Cell 24, 566–578.e7 (2019).
Haga, K. et al. Permanent, lowered HLA class I expression using lentivirus vectors with shRNA constructs: averting cytotoxicity by alloreactive T lymphocytes. Transplant. Proc. 38, 3184–3188 (2006).
Anikeeva, N., Fischer, N. O., Blanchette, C. D. & Sykulev, Y. Extent of MHC clustering regulates selectivity and effectiveness of T cell responses. J. Immunol. 202, 591–597 (2019).
Hnisz, D. et al. Super-enhancers in the control of cell identity and disease. Cell 155, 934–947 (2013).
Whyte, W. A. et al. Master transcription factors and mediator establish super-enhancers at key cell identity genes. Cell 153, 307–319 (2013).
Dowen, J. M. et al. Control of cell identity genes occurs in insulated neighborhoods in mammalian chromosomes. Cell 159, 374–387 (2014).
Adam, R. C. et al. Pioneer factors govern super-enhancer dynamics in stem cell plasticity and lineage choice. Nature 521, 366–370 (2015).
Lovén, J. et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 153, 320–334 (2013).
Cavalli, G. et al. MHC class II super-enhancer increases surface expression of HLA-DR and HLA-DQ and affects cytokine production in autoimmune vitiligo. Proc Natl Acad. Sci USA 113, 1363–1368 (2016).
Majumder, P. et al. A super enhancer controls expression and chromatin architecture within the MHC class II locus. J. Exp. Med. 217, e20190668 (2020).
Ankrum, J. A., Ong, J. F. & Karp, J. M. Mesenchymal stem cells: immune evasive, not immune privileged. Nat. Biotechnol. 32, 252–260 (2014).
Lohan, P., Treacy, O., Griffin, M. D., Ritter, T. & Ryan, A. E. Anti-donor immune responses elicited by allogeneic mesenchymal stem cells and their extracellular vesicles: are we still learning? Front. Immunol. 8, 1626 (2017).
Gao, J., Dennis, J. E., Muzic, R. F., Lundberg, M. & Caplan, A. I. The dynamic in vivo distribution of bone marrow-derived mesenchymal stem cells after infusion. Cells Tissues Organs 169, 12–20 (2001).
Toma, C., Wagner, W. R., Bowry, S., Schwartz, A. & Villanueva, F. Fate of culture-expanded mesenchymal stem cells in the microvasculature: in vivo observations of cell kinetics. Circ. Res. 104, 398–402 (2009).
Lee, R. H. et al. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell 5, 54–63 (2009).
Kidd, S. et al. Direct evidence of mesenchymal stem cell tropism for tumor and wounding microenvironments using in vivo bioluminescent imaging. Stem Cells 27, 2614–2623 (2009).
Zangi, L. et al. Direct imaging of immune rejection and memory induction by allogeneic mesenchymal stromal cells. Stem Cells 27, 2865–2874 (2009).
von Bahr, L. et al. Analysis of tissues following mesenchymal stromal cell therapy in humans indicates limited long-term engraftment and no ectopic tissue formation. Stem Cells 30, 1575–1578 (2012).
Griffin, M. D. et al. Anti-donor immune responses elicited by allogeneic mesenchymal stem cells: what have we learned so far? Immunol. Cell Biol. 91, 40–51 (2013).
Roemeling-van Rhijn, M. et al. Human allogeneic bone marrow and adipose tissue derived mesenchymal stromal cells induce CD8+ cytotoxic T cell reactivity. J. Stem Cell Res. Ther. 3, 004 (2013).
Li, Y. & Lin, F. Mesenchymal stem cells are injured by complement after their contact with serum. Blood 120, 3436–3443 (2012).
Panes, J. et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet 388, 1281–1290 (2016).
Deng, L. et al. Intrathecal injection of allogenic bone marrow-derived mesenchymal stromal cells in treatment of patients with severe ischemic stroke: study protocol for a randomized controlled observer-blinded trial. Transl. Stroke Res. 10, 170–177 (2019).
Schweizer, M. T. et al. A phase I study to assess the safety and cancer-homing ability of allogeneic bone marrow-derived mesenchymal stem cells in men with localized prostate cancer. Stem Cells Transl. Med. 8, 441–449 (2019).
Hashemian, S. R. et al. Mesenchymal stem cells derived from perinatal tissues for treatment of critically ill COVID-19-induced ARDS patients: a case series. Stem Cell Res. Ther. 12, 91 (2021).
Filippakopoulos, P. et al. Selective inhibition of BET bromodomains. Nature 468, 1067–1073 (2010).
Gilbert, L. A. et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154, 442–451 (2013).
Fulco, C. P. et al. Systematic mapping of functional enhancer-promoter connections with CRISPR interference. Science 354, 769–773 (2016).
Oh, J. Y. et al. MHC class I enables MSCs to evade NK-cell-mediated cytotoxicity and exert immunosuppressive activity. Stem Cells 40, 870–882 (2022).
Ziegler, S. F., Ramsdell, F. & Alderson, M. R. The activation antigen CD69. Stem Cells 12, 456–465 (1994).
Prager, I. & Watzl, C. Mechanisms of natural killer cell-mediated cellular cytotoxicity. J. Leukoc. Biol. 105, 1319–1329 (2019).
Zhu, J. et al. Mesenchymal stem cells alleviate LPS-induced acute lung injury by inhibiting the proinflammatory function of Ly6C(+) CD8(+) T cells. Cell Death Dis. 11, 829 (2020).
Han, B. et al. Long pentraxin PTX3 deficiency worsens LPS-induced acute lung injury. Intensive Care Med. 37, 334–342 (2011).
Lu, Z. et al. Mesenchymal stem cells activate Notch signaling to induce regulatory dendritic cells in LPS-induced acute lung injury. J. Transl. Med. 18, 241 (2020).
Borsari, B. et al. Enhancers with tissue-specific activity are enriched in intronic regions. Genome Res. 31, 1325–1336 (2021).
Amabile, A. et al. Inheritable silencing of endogenous genes by hit-and-run targeted epigenetic editing. Cell 167, 219–232.e14 (2016).
Gonzalez, S. et al. Amplification of RNAi–targeting HLA mRNAs. Mol. Ther. 11, 811–818 (2005).
Deuse, T. et al. Human leukocyte antigen I knockdown human embryonic stem cells induce host ignorance and achieve prolonged xenogeneic survival. Circulation 124, S3–S9 (2011).
Soland, M. A. et al. Modulation of human mesenchymal stem cell immunogenicity through forced expression of human cytomegalovirus us proteins. PLoS ONE 7, e36163 (2012).
Figueiredo, C., Horn, P. A., Blasczyk, R. & Seltsam, A. Regulating MHC expression for cellular therapeutics. Transfusion 47, 18–27 (2007).
Bern, M. D. et al. Inducible down-regulation of MHC class I results in natural killer cell tolerance. J. Exp. Med. 216, 99–116 (2019).
Nuñez, J. K. et al. Genome-wide programmable transcriptional memory by CRISPR-based epigenome editing. Cell 184, 2503–2519.e17 (2021).
Wang, Y. et al. Exosomes from embryonic mesenchymal stem cells alleviate osteoarthritis through balancing synthesis and degradation of cartilage extracellular matrix. Stem Cell Res. Ther. 8, 189 (2017).
Wang, Y. et al. The plasticity of mesenchymal stem cells in regulating surface HLA-I. iScience 15, 66–78 (2019).
Wang, F. et al. A comparison of CRISPR/Cas9 and siRNA-mediated ALDH2 gene silencing in human cell lines. Mol. Genet. Genomics 293, 769–783 (2018).
Wang, F. et al. Retinol binding protein 4 mediates MEHP-induced glucometabolic abnormalities in HepG2 cells. Toxicology 424, 152236 (2019).
Xiao, X. et al. Guidance of super-enhancers in regulation of IL-9 induction and airway inflammation. J. Exp. Med. 215, 559–574 (2018).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10, R25 (2009).
Zhang, Y. et al. Model-based analysis of ChIP-seq (MACS). Genome Biol. 9, R137 (2008).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-seq data with or without a reference genome. BMC Bioinformatics 12, 323 (2011).
Chen, S. et al. PP2A-mTOR-p70S6K/4E-BP1 axis regulates M1 polarization of pulmonary macrophages and promotes ambient particulate matter induced mouse lung injury. J. Hazard. Mater. 424, 127624 (2022).
Li, D. et al. Caloric restriction attenuates C57BL/6 J mouse lung injury and extra-pulmonary toxicity induced by real ambient particulate matter exposure. Part. Fibre Toxicol. 17, 22 (2020).
Acknowledgements
We thank Y. Huang, Y. Xing, Y. Li and J. Wang from the Core Facility, Zhejiang University School of Medicine for their technical support. We thank Y. J. Wang from the Blood Center of Zhejiang Province for technical support. We thank J. L. Jia, W. L. Liu, L. R. Lu, X. Chen, Z. Yin, S. F. Zhang, Y. Ping, Y. Shen, Q. Sun and Y. Gu for helpful discussions. We thank Z. J. Cai for providing the K562 cell line. H.L. discloses support for the research described in this study from the National Key Research and Development Program of China (grant number 2023YFB3813003) and the National Natural Science Foundation of China (grant number 32371424). H.W.O. discloses support for the research described in this study from the National Natural Science Foundation of China (grant numbers T2121004 and 92268203). X.H.Z. discloses support for the publication of this study from the Zhejiang Province Science and Technology Innovation Talent Plan (grant number 2023R5238), the Zhejiang Province Key Research and Development Program (grant number 2023C03099) and the National Natural Science Foundation of China (grant number 92049117).
Author information
Authors and Affiliations
Contributions
H.L., H.W.O. and X.H.Z. were responsible for the conception, supervision, interpretation and financial support of the project, and wrote the article with the assistance of all the co-authors. F.W. designed and performed the majority of the experiments, analysed the data and prepared the figures and article. R.L. led the bioinformatic analysis and annotated the B2M-SE with cooperation from F.W., H.L. and C.P. J.Y.X. performed the molecular experiments with assistance from S.C.J. X.X.B., Y.W. and Z.S. constructed the humanized mouse model and performed animal experiments with assistance from X.W. and W.G. X.R.C. performed the ALI experiments with assistance from S.C. K.Z. performed the differentiation experiments of ES cells into MSCs and identified the MSCs. B.C.H. prepared the article. J.Z. supervised the study. All authors have approved the article for submission and publication.
Corresponding authors
Ethics declarations
Competing interests
China National Intellectual Property Administration has filed for patent protection on the technology described in this study (CN113801881B and CN113846063B; with authors H.L., F.W. and R.L). The other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–6.
Supplementary Data 1 (download XLSX )
Statistics for Supplementary Figs. 1–6.
Supplementary Data 2 (download XLSX )
Raw data for Supplementary Fig. 1.
Supplementary Data 3 (download XLSX )
Raw data for Supplementary Fig. 2.
Supplementary Data 4 (download XLSX )
Raw data for Supplementary Fig. 3.
Supplementary Data 5 (download XLSX )
Raw data for Supplementary Fig. 4.
Supplementary Data 6 (download XLSX )
Raw data for Supplementary Fig. 5.
Supplementary Data 7 (download XLSX )
Raw data for Supplementary Fig. 6.
Supplementary Table 1 (download XLSX )
List of oligonucleotide sequences.
Supplementary Table 2 (download XLSX )
Key resources list.
Source data
Source Data Fig. 2 (download XLSX )
Source data.
Source Data Fig. 3 (download XLSX )
Source data.
Source Data Fig. 4 (download XLSX )
Source data.
Source Data Fig. 5 (download XLSX )
Source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, F., Li, R., Xu, J.Y. et al. Downregulating human leucocyte antigens on mesenchymal stromal cells by epigenetically repressing a β2-microglobulin super-enhancer. Nat. Biomed. Eng 8, 1682–1699 (2024). https://doi.org/10.1038/s41551-024-01264-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41551-024-01264-w
This article is cited by
-
Mesenchymal stem cell therapies for ARDS: translational promise and challenges
Stem Cell Research & Therapy (2025)
-
Low dose IFNγ remodels enhancer landscape to potentiate mesenchymal stromal cell activation
Stem Cell Research & Therapy (2025)
-
Generation of transchromosomic mice harboring HLA-A/B/C and human B2M via mouse artificial chromosome and triple BAC integration
Scientific Reports (2025)