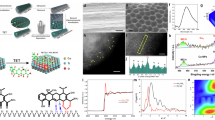
Abstract
The development of biosensors that can detect specific analytes continuously, in vivo, in real time has proven difficult due to biofouling, probe degradation and signal drift that often occur in vivo. By drawing inspiration from intestinal mucosa that can protect host cell receptors in the presence of the gut microbiome, we develop a synthetic biosensor that can continuously detect specific target molecules in vivo. The biomimetic multicomponent sensor features the hierarchical nano-bio interface design with three-dimensional bicontinuous nanoporous structure, polymer coating and aptamer switches, balancing small-molecule sensing and surface protection in complex biological environments. Our system is stable for at least 1 month in undiluted serum in vitro or 1 week implanted within the blood vessels of free-moving rats, retaining over 50% baseline signal and reproducible calibration curves. We demonstrate that the implanted system can intravenously track pharmacokinetics in real time even after 4 days of continuous exposure to flowing blood within rat femoral vein. In this way, our work provides a generalizable design foundation for biosensors that can continuously operate in vivo for extended durations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the findings of this study are available within the paper and its supplementary information files. Extra data are available from the corresponding author upon request. Source data for Figs. 1–6 are provided with this paper.
References
Sohn, E. Diagnosis: frontiers in blood testing. Nature 549, S16–S18 (2017).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Kim, J., Campbell, A. S., de Ávila, B. E.-F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406 (2019).
Ye, C. et al. A wearable aptamer nanobiosensor for non-invasive female hormone monitoring. Nat. Nanotechnol. 19, 330–337 (2024).
Sempionatto, J. R. et al. An epidermal patch for the simultaneous monitoring of haemodynamic and metabolic biomarkers. Nat. Biomed. Eng. 5, 737–748 (2021).
Wang, M. et al. A wearable electrochemical biosensor for the monitoring of metabolites and nutrients. Nat. Biomed. Eng. 6, 1225–1235 (2022).
Tavallaie, R. et al. Nucleic acid hybridization on an electrically reconfigurable network of gold-coated magnetic nanoparticles enables microRNA detection in blood. Nat. Nanotechnol. 13, 1066–1071 (2018).
Drummond, T. G., Hill, M. G. & Barton, J. K. Electrochemical DNA sensors. Nat. Biotechnol. 21, 1192–1199 (2003).
Nakatsuka, N. et al. Aptamer–field-effect transistors overcome Debye length limitations for small-molecule sensing. Science 362, 319–324 (2018).
Flynn, C. D. et al. Biomolecular sensors for advanced physiological monitoring. Nat. Rev. Bioeng. 1, 560–575 (2023).
Wang, L. et al. Rapid and ultrasensitive electromechanical detection of ions, biomolecules and SARS-CoV-2 RNA in unamplified samples. Nat. Biomed. Eng. 6, 276–285 (2022).
Yin, F. et al. DNA-framework-based multidimensional molecular classifiers for cancer diagnosis. Nat. Nanotechnol. 18, 677–686 (2023).
Swensen, J. S. et al. Continuous, real-time monitoring of cocaine in undiluted blood serum via a microfluidic, electrochemical aptamer-based sensor. J. Am. Chem. Soc. 131, 4262–4266 (2009).
Vallée-Bélisle, A., Ricci, F. & Plaxco, K. W. Thermodynamic basis for the optimization of binding-induced biomolecular switches and structure-switching biosensors. Proc. Natl Acad. Sci. USA 106, 13802–13807 (2009).
Ferguson, B. S. et al. Real-time, aptamer-based tracking of circulating therapeutic agents in living animals. Sci. Transl. Med. 5, 213ra165 (2013).
Arroyo-Currás, N. et al. Real-time measurement of small molecules directly in awake, ambulatory animals. Proc. Natl Acad. Sci. USA 114, 645–650 (2017).
Parolo, C. et al. Real-time monitoring of a protein biomarker. ACS Sens. 5, 1877–1881 (2020).
Sabaté del Río, J., Henry, O. Y. F., Jolly, P. & Ingber, D. E. An antifouling coating that enables affinity-based electrochemical biosensing in complex biological fluids. Nat. Nanotechnol. 14, 1143–1149 (2019).
Song, E., Li, J., Won, S. M., Bai, W. & Rogers, J. A. Materials for flexible bioelectronic systems as chronic neural interfaces. Nat. Mater. 19, 590–603 (2020).
Shaver, A. & Arroyo-Currás, N. The challenge of long-term stability for nucleic acid-based electrochemical sensors. Curr. Opin. Electrochem. 32, 100902 (2022).
Leslie, D. C. et al. A bioinspired omniphobic surface coating on medical devices prevents thrombosis and biofouling. Nat. Biotechnol. 32, 1134–1140 (2014).
Jaffer, I. H. & Weitz, J. I. The blood compatibility challenge. Part 1: blood-contacting medical devices: the scope of the problem. Acta Biomater. 94, 2–10 (2019).
Frutiger, A. et al. Nonspecific binding—fundamental concepts and consequences for biosensing applications. Chem. Rev. 121, 8095–8160 (2021).
Chan, D. et al. Combinatorial polyacrylamide hydrogels for preventing biofouling on implantable biosensors. Adv. Mater. 34, 2109764 (2022).
Li, S. et al. Implantable hydrogel-protective DNA aptamer-based sensor supports accurate, continuous electrochemical analysis of drugs at multiple sites in living rats. ACS Nano 17, 18525–18538 (2023).
Li, H. et al. A biomimetic phosphatidylcholine-terminated monolayer greatly improves the in vivo performance of electrochemical aptamer-based sensors. Angew. Chem. Int. Ed. 56, 7492–7495 (2017).
Watkins, Z., Karajic, A., Young, T., White, R. & Heikenfeld, J. Week-long operation of electrochemical aptamer sensors: new insights into self-assembled monolayer degradation mechanisms and solutions for stability in serum at body temperature. ACS Sens. 8, 1119–1131 (2023).
Daggumati, P., Matharu, Z., Wang, L. & Seker, E. Biofouling-resilient nanoporous gold electrodes for DNA sensing. Anal. Chem. 87, 8618–8622 (2015).
Seo, J. W. et al. Real-time monitoring of drug pharmacokinetics within tumor tissue in live animals. Sci. Adv. 8, eabk2901 (2022).
Peterson, L. W. & Artis, D. Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat. Rev. Immunol. 14, 141–153 (2014).
Wagner, C. E., Wheeler, K. M. & Ribbeck, K. Mucins and their role in shaping the functions of mucus barriers. Annu. Rev. Cell Dev. Biol. 34, 189–215 (2018).
Luo, L. Principles of Neurobiology (Garland Science, 2020).
Latz, E. et al. Ligand-induced conformational changes allosterically activate Toll-like receptor 9. Nat. Immunol. 8, 772–779 (2007).
Bansil, R. & Turner, B. S. The biology of mucus: composition, synthesis and organization. Adv. Drug Deliv. Rev. 124, 3–15 (2018).
Sun, W. W. et al. Nanoarchitecture and dynamics of the mouse enteric glycocalyx examined by freeze-etching electron tomography and intravital microscopy. Commun. Biol. 3, 5 (2020).
Shaver, A., Curtis, S. D. & Arroyo-Currás, N. Alkanethiol monolayer end groups affect the long-term operational stability and signaling of electrochemical, aptamer-based sensors in biological fluids. ACS Appl. Mater. Interfaces 12, 11214–11223 (2020).
Companion Handbook to the WHO Guidelines for the Programmatic Management of Drug-resistant Tuberculosis (World Health Organization, 2014).
Chien, J. C., Baker, S. W., Soh, H. T. & Arbabian, A. Design and analysis of a sample-and-hold CMOS electrochemical sensor for aptamer-based therapeutic drug monitoring. IEEE J. Solid State Circuits 55, 2914–2929 (2020).
Lowe, S., O’Brien-Simpson, N. M. & Connal, L. A. Antibiofouling polymer interfaces: poly(ethylene glycol) and other promising candidates. Polym. Chem. 6, 198–212 (2015).
Sharma, S., Johnson, R. W. & Desai, T. A. XPS and AFM analysis of antifouling PEG interfaces for microfabricated silicon biosensors. Biosens. Bioelectron. 20, 227–239 (2004).
Shaver, A. et al. Nuclease hydrolysis does not drive the rapid signaling decay of DNA aptamer-based electrochemical sensors in biological fluids. Langmuir 37, 5213–5221 (2021).
Chandrasekaran, A. R. Nuclease resistance of DNA nanostructures. Nat. Rev. Chem. 5, 225–239 (2021).
Armstrong, J. K., Wenby, R. B., Meiselman, H. J. & Fisher, T. C. The hydrodynamic radii of macromolecules and their effect on red blood cell aggregation. Biophys. J. 87, 4259–4270 (2004).
Lin, P.-H. & Li, B.-R. Antifouling strategies in advanced electrochemical sensors and biosensors. Analyst 145, 1110–1120 (2020).
Laktionov, M. Y., Zhulina, E. B., Richter, R. P. & Borisov, O. V. Polymer brush in a nanopore: effects of solvent strength and macromolecular architecture studied by self-consistent field and scaling theory. Polymers 13, 3929 (2021).
Fu, K. et al. Accelerated electron transfer in nanostructured electrodes improves the sensitivity of electrochemical biosensors. Adv. Sci. 8, 2102495 (2021).
White, R. J., Rowe, A. A. & Plaxco, K. W. Re-engineering aptamers to support reagentless, self-reporting electrochemical sensors. Analyst 135, 589–594 (2010).
Maganzini, N., Thompson, I., Wilson, B. & Soh, H. T. Pre-equilibrium biosensors as an approach towards rapid and continuous molecular measurements. Nat. Commun. 13, 7072 (2022).
Jaffer, I. H., Fredenburgh, J. C., Hirsh, J. & Weitz, J. I. Medical device-induced thrombosis: what causes it and how can we prevent it? J. Thromb. Haemost. 13, S72–S81 (2015).
Weber, M. et al. Blood-contacting biomaterials: in vitro evaluation of the hemocompatibility. Front. Bioeng. Biotechnol. 6, 99 (2018).
Bard, A. J., Faulkner, L. R. & White, H. S. Electrochemical Methods: Fundamentals and Applications (John Wiley & Sons, 2022).
Das, J. et al. Reagentless biomolecular analysis using a molecular pendulum. Nat. Chem. 13, 428–434 (2021).
Acknowledgements
This work was supported by the Helmsley Trust, Wellcome LEAP SAVE programme, Stanford Material and Child Health Research Institute pilot grant and the National Institutes of Health (NIH, OT2OD025342). Part of this work was performed at the Stanford Nano Shared Facilities (SNSF), Stanford Nanofabrication Facility (SNF) and Stanford University Cell Sciences Imaging Core Facility (RRID:SCR_017787), supported by the National Science Foundation under award ECCS-2026822 and ECCS-1542152. We thank the Canary Center at Stanford for Cancer Early Detection for providing facilities and the Stanford Veterinary Service Center for their assistance with animal care and training. Viability and cell proliferation assays were performed by N. Quispe Calla at Stanford Transgenic, Knockout and Tumor Model Center. Flow cytometry analysis for this project was done on instruments in the Stanford Shared FACS Facility. Y.C. was supported by the T. S. Lo Graduate Fellowship at Stanford University. Z.O. was supported by the Wu Tsai Neurosciences Institute at Stanford University. Y.C. also thanks E. Sullivan of the Technical Communications Program at Stanford for thoughtful feedback on writing, and E. Bagley and N. Torres for lab management support.
Author information
Authors and Affiliations
Contributions
Y.C. and K.X.F. conceived the project. H.T.S. supervised the project. Y.C. and K.X.F. initiated the idea of using a hierarchical nanoarchitecture for long-term stable biosensing in complex biological matrices. Y.C., K.X.F. and H.T.S. designed the experiments. Y.C. performed the experiments, and collected and analysed the data. Y.C. and Z.O. conducted the optical experiments and image analysis. R.C. performed animal surgeries and trained Y.C. Y.C., J.W.K and J.-C.C. worked on improving intravenous microwire probes. V.K. and H.Y.Y.N. advised on electrochemical sensing. Y.C., K.X.F., M.E and H.T.S. co-wrote the paper. All authors discussed the results and commented on the paper.
Corresponding author
Ethics declarations
Competing interests
Y.C., K.X.F. and H.T.S. are listed as co-inventors on a patent application related to this work filed at the US Patent and Trademark Office (No. PCT/US2024/060103). All other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Yi Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1, Notes 1–5, Figs. 1–29, Videos 1 and 2, and References.
Supplementary Video 1 (download MP4 )
Intravenous implantation. Successful intravenous access was confirmed on the basis of blood flow after retracting the implanted probes.
Supplementary Video 2 (download MP4 )
Multiday implantation in free-moving rat. The rat was able to move freely while the implanted electrode probes were constantly subjected to blood flow and mechanical strain.
Source data
Source Data Fig. 1 (download XLSX )
Source data.
Source Data Fig. 2 (download XLSX )
Source data.
Source Data Fig. 3 (download XLSX )
Source data.
Source Data Fig. 4 (download XLSX )
Source data.
Source Data Fig. 5 (download XLSX )
Source data.
Source Data Fig. 6 (download XLSX )
Source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Fu, K.X., Cotton, R. et al. A biochemical sensor with continuous extended stability in vivo. Nat. Biomed. Eng 9, 1517–1530 (2025). https://doi.org/10.1038/s41551-025-01389-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41551-025-01389-6
This article is cited by
-
Toward continuous monitoring systems: emerging trends of on-chip sensors in organ-on-a-chip
Microsystems & Nanoengineering (2026)
-
Next-generation biosensing for in situ monitoring
Nature Sensors (2026)
-
Printing technologies for monitoring crop health
Nature Communications (2026)
-
Mechanics and bio-interface engineering in flexible biosensors for continuous health monitoring
npj Flexible Electronics (2026)
-
Wireless in-body sensing through genetically engineered bacteria
Nature Communications (2025)