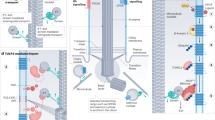
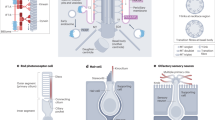
Abstract
Hair cells in the human ear contain cilia of varying lengths that sense varied acoustic signals. Here, inspired by this, we report an artificial cilia-based sound-decoding device capable of directly recognizing and responding to sound frequencies without relying on electricity and algorithms. We create 3D-printed micrometre-sized (40–200 μm) artificial cilia-based arrays with varying length-to-diameter ratios (30–100) that can sense and decode sound frequency signals (100–6,000 Hz), including piano music and human voices, on the basis of acoustic resonance. The artificial cilia can also vibrate accordingly in water to initiate subsequent tasks such as controlling drug release profiles of two distinct therapeutics (insulin and glucagon) in an acoustic-frequency-responsive manner to treat type 1 diabetic mice. This cochlear cilia-inspired device holds potential for broad applications such as recognizing complicated physiological sounds and performing various tasks in personalized voice interactions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are presented in the Article and its Supplementary Information. Source data are provided with this paper.
References
Suvilehto, J. T., Cekaite, A. & Morrison, I. The why, who and how of social touch. Nat. Rev. Psychol. 2, 606–621 (2023).
Cohen, P. R. & Oviatt, S. L. The role of voice input for human–machine communication. Proc. Natl Acad. Sci. USA 92, 9921–9927 (1995).
Yan, W. et al. Single fibre enables acoustic fabrics via nanometre-scale vibrations. Nature 603, 616–623 (2022).
Lee, S. et al. An ultrathin conformable vibration-responsive electronic skin for quantitative vocal recognition. Nat. Commun. 10, 2468 (2019).
Yang, Q. et al. Mixed-modality speech recognition and interaction using a wearable artificial throat. Nat. Mach. Intell. 5, 169–180 (2023).
Ma, P., Petridis, S. & Pantic, M. Visual speech recognition for multiple languages in the wild. Nat. Mach. Intell. https://doi.org/10.1038/s42256-022-00550-z (2022).
Koenecke, A. et al. Racial disparities in automated speech recognition. Proc. Natl Acad. Sci. USA 117, 7684–7689 (2020).
Young, T., Hazarika, D., Poria, S. & Cambria, E. Recent trends in deep learning based natural language processing. IEEE Comput. Intell. Mag. 13, 55–75 (2018).
Pataranutaporn, P. et al. AI-generated characters for supporting personalized learning and well-being. Nat. Mach. Intell. 3, 1013–1022 (2021).
Chen, C., Ding, S. & Wang, J. Digital health for aging populations. Nat. Med. 29, 1623–1630 (2023).
Raucci, U. et al. Voice-controlled quantum chemistry. Nat. Comput. Sci. 1, 42–45 (2021).
Sezgin, E., Huang, Y., Ramtekkar, U. & Lin, S. Readiness for voice assistants to support healthcare delivery during a health crisis and pandemic. npj Digit. Med. 3, 122 (2020).
Zhang, A., Xing, L., Zou, J. & Wu, J. C. Shifting machine learning for healthcare from development to deployment and from models to data. Nat. Biomed. Eng. 6, 1330–1345 (2022).
Hansen, J. H. & Hasan, T. Speaker recognition by machines and humans: a tutorial review. IEEE Signal Process Mag. 32, 74–99 (2015).
Haeb-Umbach, R. et al. Far-field automatic speech recognition. Proc. IEEE 109, 124–148 (2020).
LeCun, Y., Bengio, Y. & Hinton, G. Deep learning. Nature 521, 436–444 (2015).
Fettiplace, R. & Hackney, C. M. The sensory and motor roles of auditory hair cells. Nat. Rev. Neurosci. 7, 19–29 (2006).
Hudspeth, A. Integrating the active process of hair cells with cochlear function. Nat. Rev. Neurosci. 15, 600–614 (2014).
Zheng, J. et al. Prestin is the motor protein of cochlear outer hair cells. Nature 405, 149–155 (2000).
Ren, T., He, W. & Kemp, D. Reticular lamina and basilar membrane vibrations in living mouse cochleae. Proc. Natl Acad. Sci. USA 113, 9910–9915 (2016).
Wei, X. et al. Biomimetic design strategies for biomedical applications. Matter 7, 826–854 (2024).
Martin, P. & Hudspeth, A. Active hair-bundle movements can amplify a hair cell’s response to oscillatory mechanical stimuli. Proc. Natl Acad. Sci. USA 96, 14306–14311 (1999).
Fisher, J. A., Nin, F., Reichenbach, T., Uthaiah, R. C. & Hudspeth, A. The spatial pattern of cochlear amplification. Neuron 76, 989–997 (2012).
Schneider, M. E., Belyantseva, I. A., Azevedo, R. B. & Kachar, B. Rapid renewal of auditory hair bundles. Nature 418, 837–838 (2002).
Kozlov, A. S., Risler, T. & Hudspeth, A. Coherent motion of stereocilia assures the concerted gating of hair-cell transduction channels. Nat. Neurosci. 10, 87–92 (2007).
Hakizimana, P. & Fridberger, A. Inner hair cell stereocilia are embedded in the tectorial membrane. Nat. Commun. 12, 2604 (2021).
Hakizimana, P., Brownell, W. E., Jacob, S. & Fridberger, A. Sound-induced length changes in outer hair cell stereocilia. Nat. Commun. 3, 1094 (2012).
Singla, V. & Reiter, J. F. The primary cilium as the cell’s antenna: signaling at a sensory organelle. Science 313, 629–633 (2006).
Anvarian, Z., Mykytyn, K., Mukhopadhyay, S., Pedersen, L. B. & Christensen, S. T. Cellular signalling by primary cilia in development, organ function and disease. Nat. Rev. Nephrol. 15, 199–219 (2019).
Mill, P., Christensen, S. T. & Pedersen, L. B. Primary cilia as dynamic and diverse signalling hubs in development and disease. Nat. Rev. Genet. https://doi.org/10.1038/s41576-023-00587-9 (2023).
Gilpin, W., Bull, M. S. & Prakash, M. The multiscale physics of cilia and flagella. Nat. Rev. Phys. 2, 74–88 (2020).
Katoh, T. A. et al. Immotile cilia mechanically sense the direction of fluid flow for left-right determination. Science 379, 66–71 (2023).
Gu, H. et al. Magnetic cilia carpets with programmable metachronal waves. Nat. Commun. 11, 2637 (2020).
Kim, Y. & Zhao, X. Magnetic soft materials and robots. Chem. Rev. 122, 5317–5364 (2022).
Kim, Y., Parada, G. A., Liu, S. & Zhao, X. Ferromagnetic soft continuum robots. Sci. Rob. 4, eaax7329 (2019).
Dai, B. et al. Artificial asymmetric cilia array of dielectric elastomer for cargo transportation. ACS Appl. Mater. Interfaces 10, 42979–42984 (2018).
Wang, W. et al. Cilia metasurfaces for electronically programmable microfluidic manipulation. Nature 605, 681–686 (2022).
Gelebart, A. H., Mc Bride, M., Schenning, A. P., Bowman, C. N. & Broer, D. J. Photoresponsive fiber array: toward mimicking the collective motion of cilia for transport applications. Adv. Funct. Mater. 26, 5322–5327 (2016).
Li, S. et al. Self-regulated non-reciprocal motions in single-material microstructures. Nature 605, 76–83 (2022).
Milana, E., Gorissen, B., Peerlinck, S., De Volder, M. & Reynaerts, D. Artificial soft cilia with asymmetric beating patterns for biomimetic low‐Reynolds‐number fluid propulsion. Adv. Funct. Mater. 29, 1900462 (2019).
Milana, E. et al. Metachronal patterns in artificial cilia for low Reynolds number fluid propulsion. Sci. Adv. 6, eabd2508 (2020).
Dillinger, C., Nama, N. & Ahmed, D. Ultrasound-activated ciliary bands for microrobotic systems inspired by starfish. Nat. Commun. 12, 6455 (2021).
Orbay, S., Ozcelik, A., Bachman, H. & Huang, T. J. Acoustic actuation of in situ fabricated artificial cilia. J. Micromech. Microeng. 28, 025012 (2018).
He, X. et al. Synthetic homeostatic materials with chemo-mechano-chemical self-regulation. Nature 487, 214–218 (2012).
Zarzar, L. D., Kim, P. & Aizenberg, J. Bio‐inspired design of submerged hydrogel‐actuated polymer microstructures operating in response to pH. Adv. Mater. 23, 1442–1446 (2011).
Zhang, S., Zhang, R., Wang, Y., Onck, P. R. & den Toonder, J. M. Controlled multidirectional particle transportation by magnetic artificial cilia. ACS Nano 14, 10313–10323 (2020).
Pokroy, B., Kang, S. H., Mahadevan, L. & Aizenberg, J. Self-organization of a mesoscale bristle into ordered, hierarchical helical assemblies. Science 323, 237–240 (2009).
Ren, Z. et al. Soft-robotic ciliated epidermis for reconfigurable coordinated fluid manipulation. Sci. Adv. 8, eabq2345 (2022).
Sarlo, R., Najem, J. S. & Leo, D. J. Flow field sensing with bio-inspired artificial hair cell arrays. Sensor Actuat. B 236, 805–814 (2016).
Alfadhel, A., Li, B., Zaher, A., Yassine, O. & Kosel, J. A magnetic nanocomposite for biomimetic flow sensing. Lab Chip 14, 4362–4369 (2014).
den Toonder, J. M. & Onck, P. R. Microfluidic manipulation with artificial/bioinspired cilia. Trends Biotechnol. 31, 85–91 (2013).
Dong, X. et al. Bioinspired cilia arrays with programmable nonreciprocal motion and metachronal coordination. Sci. Adv. 6, eabc9323 (2020).
Lu, H. et al. A bioinspired multilegged soft millirobot that functions in both dry and wet conditions. Nat. Commun. 9, 3944 (2018).
Müller, U. & Barr-Gillespie, P. G. New treatment options for hearing loss. Nat. Rev. Drug Discov. 14, 346–365 (2015).
Roccio, M. et al. Molecular characterization and prospective isolation of human fetal cochlear hair cell progenitors. Nat. Commun. 9, 4027 (2018).
Schwander, M., Kachar, B. & Müller, U. Review series: the cell biology of hearing. J. Cell Biol. 190, 9 (2010).
Titze, I. R. & Martin, D. W. Principles of Voice Production (Acoustical Society of America, 1998).
Kinsler, L. E., Frey, A. R., Coppens, A. B. & Sanders, J. V. Fundamentals of Acoustics (John Wiley & Sons, 2000).
Durrer, J. et al. A robot-assisted acoustofluidic end effector. Nat. Commun. 13, 6370 (2022).
Lee, T.-R. et al. Resonant behavior and microfluidic manipulation of silicone cilia due to an added mass effect. Soft Matter 7, 4325–4334 (2011).
Grillet, N. High-resolution imaging of the mouse-hair-cell hair bundle by scanning electron microscopy. STAR Protoc. 3, 101213 (2022).
Rao, S. S. & Yap, F. F. Mechanical Vibrations vol. 4 (Addison-Wesley, 1995).
Gurtin, M. E. An Introduction to Continuum Mechanics (Academic Press, 1982).
Ji, K. et al. An orally administered glucose-responsive polymeric complex for high-efficiency and safe delivery of insulin in mice and pigs. Nat. Nanotechnol. 19, 1880–1891 (2024).
Xu, J. et al. A bioinspired polymeric membrane-enclosed insulin crystal achieves long-term, self-regulated drug release for type 1 diabetes therapy. Nat. Nanotechnol. https://doi.org/10.1038/s41565-025-01860-0 (2025).
Acknowledgements
This work was supported by the grants from “Pioneer” and “Leading Goose” R&D Program of Zhejiang (grant no. 2024C03085) and Zhejiang University’s start-up packages, National Natural Science Foundation of China (grant nos. 82100911 and 32201082), National Natural Science Fund for Excellent Young Scientist Fund Program (Overseas) (grant no. 588020-X42208/061), Zhejiang Provincial Natural Science Foundation (grant no. LQ18H070004). We thank S. Liu from the Core Facilities, Zhejiang University School of Medicine, for her technical support in Confocal Microscopy. We thank G. Zhu and D. Song in the Center of Cryo-Electron Microscopy (CCEM), Zhejiang University, for their assistance with SEM. We appreciate S. Yang from Hangzhou Liuxia Primary School for her guidance on music theory.
Author information
Authors and Affiliations
Contributions
Z.G., J.W. and X.W. conceived and designed the study. X.W., H.W., Y.W., W.Z., C.C., K.L., L.H., J.R., J.X., Y.Y., Y.H. and T.Z. conducted experiments and collected related data. X.W., H.W., X.L., J.G., P.W., C.X., J.W. and Z.G. analysed the data and wrote the paper. T.J.H. edited the paper.
Corresponding authors
Ethics declarations
Competing interests
Z.G. is the co-founder of Zcapsule Inc. and μZen Inc. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Daniel Ahmed and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–13 and Videos 1 and 2.
Supplementary Video 1 (download MP4 )
The vibration of artificial cilia in response to sound stimuli in the air recorded by the high-speed camera.
Supplementary Video 2 (download MP4 )
The vibration of artificial cilia array upon the acoustic stimulation by PZT in the water.
Supplementary Data (download XLSX )
Statistical source data for the Supplementary Information.
Source data
Source Data Figs. 2–5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, X., Wang, H., Wang, Y. et al. An artificial cilia-based array system for sound frequency decoding and resonance-responsive drug release. Nat. Biomed. Eng (2025). https://doi.org/10.1038/s41551-025-01505-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41551-025-01505-6