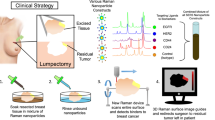
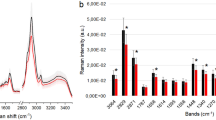
Abstract
Radical prostatectomy is a standard curative approach for high-risk prostate cancer, yet accurately defining tumour margins during surgery remains a major challenge. Intraoperative assessment of prostate tumour malignancy—particularly those with high aggressiveness catalogued in Gleason grade group (GG) ≥ 3—is crucial to prevent positive surgical margins and minimize postoperative complications. Here we develop a surface-enhanced Raman scattering (SERS)-based navigation system for intraoperative localization of high-grade malignant regions by simultaneously accessing tissue acidity and prostate-specific antigen (PSA) enzymatic activity. This system integrates a sampling pen for automated biomarker extraction from tissue surfaces, a nano-imprinted SERS array producing a ratiometric Raman signal in response to acidity and PSA activity, and a two-dimensional deep-learning model for rapid Raman spectral interpretation. We show that the system can intraoperatively identify GG ≥ 3 malignancies in fresh prostate tissues from 144 Chinese patients with an area under the receiver operating characteristic curve of 0.89. This SERS-based navigation system holds strong potential to enhance surgical precision, minimize tumour residue and ultimately improve patient outcomes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the paper and its Supplementary Information. Source data are provided with this paper and available from figshare at https://doi.org/10.6084/m9.figshare.30001840 (ref. 58). Any other datasets generated and analysed during the study are available for research purposes from the corresponding authors upon request.
Code availability
The code supporting this study is available from https://github.com/zz9912/CNN_Raman_pH_detection.git.
References
Gandaglia, G. et al. Epidemiology and prevention of prostate cancer. Eur. Urol. Oncol. 4, 877–892 (2021).
Siegel, R. L., Miller, K. D., Wagle, N. S. & Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 73, 17–48 (2023).
Gandaglia, G. et al. Adjuvant and salvage radiotherapy after radical prostatectomy in prostate cancer patients. Eur. Urol. 72, 689–709 (2017).
Preisser, F. et al. Rates of positive surgical margins and their effect on cancer-specific mortality at radical prostatectomy for patients with clinically localized prostate cancer. Clin. Genitourin. Cancer 17, e130–e139 (2019).
Costello, A. J. Considering the role of radical prostatectomy in 21st century prostate cancer care. Nat. Rev. Urol. 17, 177–188 (2020).
Patel, V. R. et al. Pentafecta: a new concept for reporting outcomes of robot-assisted laparoscopic radical prostatectomy. Eur. Urol. 59, 702–707 (2011).
Bravi, C. A. et al. Positive surgical margins after anterior robot-assisted radical prostatectomy: assessing the learning curve in a multi-institutional collaboration. Eur. Urol. Oncol. 7, 821–828 (2024).
Zhang, L. et al. Positive surgical margin is associated with biochemical recurrence risk following radical prostatectomy: a meta-analysis from high-quality retrospective cohort studies. World J. Surg. Oncol. 16, 124 (2018).
Preisser, F. et al. Impact of positive surgical margin length and Gleason grade at the margin on biochemical recurrence in patients with organ-confined prostate cancer. Prostate 79, 1832–1836 (2019).
Viers, B. R. et al. Primary Gleason grade 4 at the positive margin is associated with metastasis and death among patients with Gleason 7 prostate cancer undergoing radical prostatectomy. Eur. Urol. 66, 1116–1124 (2014).
Lysenko, I. et al. MP64-11 prognostic impact of Gleason grade at the margin in patients undergoing radical prostatectomy. J. Urol. 206, e1114 (2021).
Miyamoto, H. Intraoperative pathology consultation during urological surgery: impact on final margin status and pitfalls of frozen section diagnosis. Pathol. Int. 71, 567–580 (2021).
Denmeade, S. R. et al. Specific and efficient peptide substrates for assaying the proteolytic activity of prostate-specific antigen. Cancer Res. 57, 4924–4930 (1997).
Catalona, W. J. et al. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. N. Engl. J. Med. 324, 1156–1161 (1991).
Denmeade, S. R., Sokoll, L. J., Chan, D. W., Khan, S. R. & Isaacs, J. T. Concentration of enzymatically active prostate-specific antigen (PSA) in the extracellular fluid of primary human prostate cancers and human prostate cancer xenograft models. Prostate 48, 1–6 (2001).
Aihara, M. et al. Prostate specific antigen and Gleason grade: an immunohistochemical study of prostate cancer. J. Urol. 151, 1558–1564 (1994).
Ahrens, M. J. et al. PSA enzymatic activity: a new biomarker for assessing prostate cancer aggressiveness. Prostate 73, 1731–1737 (2013).
Pretlow, T. G. et al. Tissue concentrations of prostate-specific antigen in prostatic carcinoma and benign prostatic hyperplasia. Int. J. Cancer 49, 645–649 (1991).
Nguyen, T. T. et al. Aurora kinase A inhibition reverses the Warburg effect and elicits unique metabolic vulnerabilities in glioblastoma. Nat. Commun. 12, 5203 (2021).
Niland, S., Riscanevo, A. X. & Eble, J. A. Matrix metalloproteinases shape the tumor microenvironment in cancer progression. Int. J. Mol. Sci. 23, 146 (2021).
Sushentsev, N. et al. Hyperpolarised 13C-MRI identifies the emergence of a glycolytic cell population within intermediate-risk human prostate cancer. Nat. Commun. 13, 466 (2022).
Jones, R. R., Hooper, D. C., Zhang, L., Wolverson, D. & Valev, V. K. Raman techniques: fundamentals and frontiers. Nanoscale Res. Lett. 14, 231 (2019).
Pinto, M. et al. Integration of a Raman spectroscopy system to a robotic-assisted surgical system for real-time tissue characterization during radical prostatectomy procedures. J. Biomed. Opt. 24, 025001 (2019).
Xu, H., Bjerneld, E. J., Käll, M. & Börjesson, L. Spectroscopy of single hemoglobin molecules by surface enhanced Raman scattering. Phys. Rev. Lett. 83, 4357 (1999).
Li, J. F. et al. Shell-isolated nanoparticle-enhanced Raman spectroscopy. Nature 464, 392–395 (2010).
Fleischmann, M., Hendra, P. J. & McQuillan, A. J. Raman spectra of pyridine adsorbed at a silver electrode. Chem. Phys. Lett. 26, 163–166 (1974).
Hakonen, A. et al. Dimer-on-mirror SERS substrates with attogram sensitivity fabricated by colloidal lithography. Nanoscale 7, 9405–9410 (2015).
Le Ru, E. C. & Auguié, B. Enhancement factors: a central concept during 50 years of surface-enhanced Raman spectroscopy. ACS Nano 18, 9773–9783 (2024).
Cong, S. et al. Noble metal-comparable SERS enhancement from semiconducting metal oxides by making oxygen vacancies. Nat. Commun. 6, 7800 (2015).
Bi, X., Czajkowsky, D. M., Shao, Z. & Ye, J. Digital colloid-enhanced Raman spectroscopy by single-molecule counting. Nature 628, 771–775 (2024).
Langer, J. et al. Present and future of surface-enhanced Raman scattering. ACS Nano 14, 28–117 (2019).
Yin, H. et al. Emergence of responsive surface-enhanced Raman scattering probes for imaging tumor-associated metabolites. Adv. Healthc. Mater. 11, e2200030 (2022).
Kneipp, K. et al. Single molecule detection using surface-enhanced Raman scattering (SERS). Phys. Rev. Lett. 78, 1667 (1997).
Gaggero, S. et al. IL-2 is inactivated by the acidic pH environment of tumors enabling engineering of a pH-selective mutein. Sci. Immunol. 7, eade5686 (2022).
Kuppusamy, P. et al. Noninvasive imaging of tumor redox status and its modification by tissue glutathione levels. Cancer Res. 62, 307–312 (2002).
Ye, H. et al. Recent advances on reactive oxygen species-responsive delivery and diagnosis system. Biomacromolecules 20, 2441–2463 (2019).
Jin, Z. et al. Intelligent SERS navigation system guiding brain tumor surgery by intraoperatively delineating the metabolic acidosis. Adv. Sci. 9, 2104935 (2022).
Neves, T. B. & Andrade, G. F. SERS characterization of the indocyanine-type dye IR-820 on gold and silver nanoparticles in the near infrared. J. Spectrosc. https://doi.org/10.1155/2015/805649 (2015).
Gutkin, S., Green, O., Raviv, G., Shabat, D. & Portnoy, O. Powerful chemiluminescence probe for rapid detection of prostate specific antigen proteolytic activity: forensic identification of human semen. Bioconjug. Chem. 31, 2488–2493 (2020).
Choi, J. H. et al. A novel Au-nanoparticle biosensor for the rapid and simple detection of PSA using a sequence-specific peptide cleavage reaction. Biosens. Bioelectron. 49, 415–419 (2013).
Eckmann, J.-P., Kamphorst, S. O. & Ruelle, D. Recurrence plots of dynamical systems. World Sci. Ser. Nonlinear Sci. Ser. A 16, 441–446 (1995).
Selvaraju, R. R. et al. Grad-CAM: visual explanations from deep networks via gradient-based localization. In Proc. IEEE International Conference on Computer Vision 618–626 (IEEE, 2017).
Bracarda, S. et al. Cancer of the prostate. Crit. Rev. Oncol. Hematol. 56, 379–396 (2005).
Grignon, D. J. Unusual subtypes of prostate cancer. Mod. Pathol. 17, 316–327 (2004).
Estrella, V. et al. Acidity generated by the tumor microenvironment drives local invasion. Cancer Res. 73, 1524–1535 (2013).
Zhang, J. et al. Nondestructive tissue analysis for ex vivo and in vivo cancer diagnosis using a handheld mass spectrometry system. Sci. Transl. Med. 9, eaan3968 (2017).
Adam, C. et al. Photodynamic diagnosis using 5-aminolevulinic acid for the detection of positive surgical margins during radical prostatectomy in patients with carcinoma of the prostate: a multicentre, prospective, phase 2 trial of a diagnostic procedure. Eur. Urol. 55, 1281–1288 (2009).
Olde Heuvel, J. et al. 68Ga-PSMA Cerenkov luminescence imaging in primary prostate cancer: first-in-man series. Eur. J. Nucl. Med. Mol. Imaging 47, 2624–2632 (2020).
Darr, C. et al. Intraoperative 68Ga-PSMA Cerenkov luminescence imaging for surgical margins in radical prostatectomy: a feasibility study. J. Nucl. Med. 61, 1500–1506 (2020).
Lajunen, T. et al. The effect of light sensitizer localization on the stability of indocyanine green liposomes. J. Contr. Release 284, 213–223 (2018).
Aubertin, K. et al. Mesoscopic characterization of prostate cancer using Raman spectroscopy: potential for diagnostics and therapeutics. BJU Int. 122, 326–336 (2018).
Tira, C., Tira, D., Simon, T. & Astilean, S. Finite-difference time-domain (FDTD) design of gold nanoparticle chains with specific surface plasmon resonance. J. Mol. Struct. 1072, 137–143 (2014).
Grimault, A. S., Vial, A. & Lamy de la Chapelle, M. Modeling of regular gold nanostructures arrays for SERS applications using a 3D FDTD method. Appl. Phys. B 84, 111–115 (2006).
Haynes, W. M., Lide, D. R. & Bruno, T. J. CRC Handbook of Chemistry and Physics 95th edn (CRC Press, 2014).
Swietach, P., Vaughan-Jones, R. D., Harris, A. L. & Hulikova, A. The chemistry, physiology and pathology of pH in cancer. Philos. Trans. R. Soc. B 369, 20130099 (2014).
Chesler, M. Regulation and modulation of pH in the brain. Physiol. Rev. 83, 1183–1221 (2003).
Epstein, J. I. et al. The 2014 International Society of Urological Pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma: definition of grading patterns and proposal for a new grading system. Am. J. Surg. Pathol. 40, 244–252 (2016).
Jin, Z. Ziyi_jin_NBME _source_data. figshare https://doi.org/10.6084/m9.figshare.30001840.v1 (2025).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2023YFA1801200 to Cong Li), National Science Fund for Distinguished Young Scholars (82025019 to Cong Li), National Natural Science Foundation of China (82227806 and 92159304 to Cong Li and 62273099 to H.W.), Shanghai Health Commission Emerging Cross Disciplinary Research Project (2022JC003 to Cong Li), Science and Technology Commission of Shanghai Municipality (23TS1401100 and 23J21901700 to Cong Li), AI for Science Initiative of Fudan University (FudanX24AI049 to Changle Li and FudanX24AI038 to J.Y.), Postdoctoral Fellowship Program of the China Postdoctoral Science Foundation (grant GZC20251908 to Z.J.) and 2025 Medical Engineering Integration Project of Fudan University (to H.W.).
Author information
Authors and Affiliations
Contributions
Z.J., H. Zheng, J.Y., H. Yang, H.W. and Cong Li conceived of and designed the research. Z.J., S.C., X.D., Z.Z., H. Zeng, P.Z., H. Yin and Changle Li performed the experiments. C.H., J.H. and J.C. provided clinical patient specimens. Z.J. and Z.Z. wrote the manuscript. Cong Li supervised the overall project. All authors discussed the results and assisted with editing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Pola Goldberg Oppenheimer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–16, Tables 1–7, methods and results.
Supplementary Video 1 (download MP4 )
Video demonstrating the sampling module.
Supplementary Video 2 (download MP4 )
Video demonstrating the sampling pen tip.
Supplementary Video 3 (download MP4 )
Video demonstrating the detection module.
Supplementary Data 1 (download XLSX )
Patient information.
Supplementary Data 2 (download XLSX )
Source data for Supplementary Figs. 3, 6–8 and 11–16.
Source data
Source Data Figs. 2, 3, 6 and 7 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download ZIP )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, Z., Chen, S., Dong, X. et al. Label-free navigation system for grading prostate tumour malignancy in situ via tissue pH and prostate-specific antigen activity. Nat. Biomed. Eng (2025). https://doi.org/10.1038/s41551-025-01561-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41551-025-01561-y