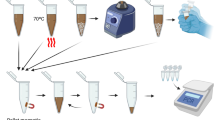
Abstract
Candida auris, an increasingly prevalent fungal pathogen, requires both rapid identification and antifungal susceptibility testing to enable proper treatment. This study introduces digital SHERLOCK (dSHERLOCK), a platform that combines CRISPR/Cas nucleic acid detection, single-template quantification and real-time kinetics monitoring. Assays implemented on this platform display excellent sensitivity to C. auris from major clades 1–4, while maintaining specificity when challenged with common environmental and pathogenic fungi. dSHERLOCK detects C. auris within 20 min in minimally processed swab samples and achieves sensitive quantification (1 c.f.u. µl−1) within 40 min. To address antifungal susceptibility testing, we develop assays that detect mutations that are commonly associated with azole and echinocandin multidrug resistance. We use machine learning and real-time monitoring of reaction kinetics to achieve highly accurate simultaneous quantification of mutant and wild-type FKS1 SNP alleles in fungal populations with mixed antifungal susceptibility, which would be misdiagnosed as completely susceptible or resistant under standard reaction conditions. Our platform’s use of commercially available materials and common laboratory equipment makes C. auris diagnostics widely deployable in global healthcare settings.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information. Example data can be downloaded from figshare at https://doi.org/10.6084/m9.figshare.29944823 (ref. 51). Should any raw data files be needed in another format, they are available from the corresponding author on reasonable request.
Code availability
The image processing and machine learning scripts have been deposited in the open-access online repository GitHub and may be accessed at https://github.com/Walt-Lab/dSherlock (ref. 52). The source code is also available as a Code Ocean Compute Capsule at https://doi.org/10.24433/CO.6935105 (ref. 53). The image processing and machine learning scripts have been deposited in GitHub at https://github.com/Walt-Lab/dSherlock (ref. 52) and in Zenodo at https://doi.org/10.5281/zenodo.17153993 (ref. 54).
References
WHO Fungal Priority Pathogens List to Guide Research, Development and Public Health Action (World Health Organization, 2022).
Fisher, M. C. et al. Tackling the emerging threat of antifungal resistance to human health. Nat. Rev. Microbiol. 20, 557–571 (2022).
Antibiotic Resistance Threats in the United States 2019 (CDC, 2019).
Mishra, S. K., Yasir, M. & Willcox, M. Candida auris: an emerging antimicrobial-resistant organism with the highest level of concern. Lancet Microbe 4, e482–e483 (2023).
Dennis, E. K., Chaturvedi, S. & Chaturvedi, V. So many diagnostic tests, so little time: review and preview of Candida auris testing in clinical and public health laboratories. Front. Microbiol. 12, 757835 (2021).
Wickes, B. L. & Wiederhold, N. P. Molecular diagnostics in medical mycology. Nat. Commun. 9, 5135 (2018).
Jeffery-Smith, A. et al. Candida auris: a review of the literature. Clin. Microbiol. Rev. 31, e00029-17 (2017).
Satoh, K. et al. Candida auris sp. nov., a novel ascomycetous yeast isolated from the external ear canal of an inpatient in a Japanese hospital. Microbiol. Immunol. 53, 41–44 (2009).
Spivak, E. S. & Hanson, K. E. Candida auris: an emerging fungal pathogen. J. Clin. Microbiol. 56, e01588-17 (2018).
Zhang, S. X. et al. Recognition of diagnostic gaps for laboratory diagnosis of fungal diseases: expert opinion from the Fungal Diagnostics Laboratories Consortium (FDLC). J. Clin. Microbiol. 59, e0178420 (2021).
Chen, J. S. et al. CRISPR-Cas12a target binding unleashes indiscriminate single-stranded DNase activity. Science 360, 436–439 (2018).
Gootenberg, J. S. et al. Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science 360, 439–444 (2018).
Gootenberg, J. S. et al. Nucleic acid detection with CRISPR-Cas13a/C2c2. Science 356, 438–442 (2017).
Kaminski, M. M., Abudayyeh, O. O., Gootenberg, J. S., Zhang, F. & Collins, J. J. CRISPR-based diagnostics. Nat. Biomed. Eng. 5, 643–656 (2021).
de Puig, H. et al. Minimally instrumented SHERLOCK (miSHERLOCK) for CRISPR-based point-of-care diagnosis of SARS-CoV-2 and emerging variants. Sci. Adv. 7, eabh2944 (2021).
Nguyen, P. Q. et al. Wearable materials with embedded synthetic biology sensors for biomolecule detection. Nat. Biotechnol. 39, 1366–1374 (2021).
Nguyen, L. T., Smith, B. M. & Jain, P. K. Enhancement of trans-cleavage activity of Cas12a with engineered crRNA enables amplified nucleic acid detection. Nat. Commun. 11, 4906 (2020).
Ooi, K. H. et al. An engineered CRISPR-Cas12a variant and DNA-RNA hybrid guides enable robust and rapid COVID-19 testing. Nat. Commun. 12, 1739 (2021).
Wang, Y. et al. Systematic evaluation of CRISPR-Cas systems reveals design principles for genome editing in human cells. Genome Biol. 19, 62 (2018).
Huyke, D. A. et al. Enzyme kinetics and detector sensitivity determine limits of detection of amplification-free CRISPR-Cas12 and CRISPR-Cas13 diagnostics. Anal. Chem. 94, 9826–9834 (2022).
Blanluet, C., Huyke, D. A., Ramachandran, A., Avaro, A. S. & Santiago, J. G. Detection and discrimination of single nucleotide polymorphisms by quantification of CRISPR-Cas catalytic efficiency. Anal. Chem. 94, 15117–15123 (2022).
Athamanolap, P. et al. Nanoarray digital polymerase chain reaction with high-resolution melt for enabling broad bacteria identification and pheno-molecular antimicrobial susceptibility test. Anal. Chem. 91, 12784–12792 (2019).
Luo, Y. et al. SAMBA: a multicolor digital melting PCR platform for rapid microbiome profiling. Small Methods 6, 2200185 (2022).
Miglietta, L. et al. Adaptive filtering framework to remove nonspecific and low-efficiency reactions in multiplex digital PCR based on sigmoidal trends. Anal. Chem. 94, 14159–14168 (2022).
Moniri, A., Miglietta, L., Holmes, A., Georgiou, P. & Rodriguez-Manzano, J. High-level multiplexing in digital PCR with intercalating dyes by coupling real-time kinetics and melting curve analysis. Anal. Chem. 92, 14181–14188 (2020).
Rolando, J. C., Jue, E., Schoepp, N. G. & Ismagilov, R. F. Real-time, digital LAMP with commercial microfluidic chips reveals the interplay of efficiency, speed, and background amplification as a function of reaction temperature and time. Anal. Chem. 91, 1034–1042 (2019).
Schoepp, N. G. et al. Rapid pathogen-specific phenotypic antibiotic susceptibility testing using digital LAMP quantification in clinical samples. Sci. Transl. Med. 9, eaal3693 (2017).
Shinoda, H. et al. Amplification-free RNA detection with CRISPR–Cas13. Commun. Biol. 4, 476 (2021).
Tian, T. et al. An ultralocalized Cas13a assay enables universal and nucleic acid amplification-free single-molecule RNA diagnostics. ACS Nano 15, 1167–1178 (2021).
Yue, H. et al. Droplet Cas12a assay enables DNA quantification from unamplified samples at the single-molecule level. Nano Lett. 21, 4643–4653 (2021).
Roh, Y. H. et al. CRISPR-enhanced hydrogel microparticles for multiplexed detection of nucleic acids. Adv. Sci. 10, 2206872 (2023).
Shinoda, H. et al. Automated amplification-free digital RNA detection platform for rapid and sensitive SARS-CoV-2 diagnosis. Commun. Biol. 5, 473 (2022).
Park, J. S. et al. Digital CRISPR/Cas-assisted assay for rapid and sensitive detection of SARS-CoV-2. Adv. Sci. 8, 2003564 (2021).
Wu, X. et al. Digital CRISPR-based method for the rapid detection and absolute quantification of nucleic acids. Biomaterials 274, 120876 (2021).
Yu, Z., Xu, L., Lyu, W. & Shen, F. Parallel multistep digital analysis SlipChip demonstrated with the quantification of nucleic acid by digital LAMP-CRISPR. Lab Chip 22, 2954–2961 (2022).
Kordalewska, M. et al. Rapid and accurate molecular identification of the emerging multidrug-resistant pathogen Candida auris. J. Clin. Microbiol. 55, 2445–2452 (2017).
Leach, L., Zhu, Y. & Chaturvedi, S. Development and validation of a real-time PCR assay for rapid detection of Candida auris from surveillance samples. J. Clin. Microbiol. 56, e01223-17 (2018).
Denancé, N., Briand, M., Gaborieau, R., Gaillard, S. & Jacques, M.-A. Identification of genetic relationships and subspecies signatures in Xylella fastidiosa. BMC Genomics 20, 239 (2019).
Procedure for Collection of Patient Swabs for Candida auris (Centers for Disease Control and Prevention, 2022).
Zhu, Y. et al. Laboratory analysis of an outbreak of Candida auris in New York from 2016 to 2018: impact and lessons learned. J. Clin. Microbiol. 58, e01503-19 (2020).
Kellner, M. J., Koob, J. G., Gootenberg, J. S., Abudayyeh, O. O. & Zhang, F. SHERLOCK: nucleic acid detection with CRISPR nucleases. Nat. Protoc. 14, 2986–3012 (2019).
Rolando, J. C., Jue, E., Barlow, J. T. & Ismagilov, R. F. Real-time kinetics and high-resolution melt curves in single-molecule digital LAMP to differentiate and study specific and non-specific amplification. Nucleic Acids Res. 48, e42 (2020).
Yao, Z. et al. Tuning the dynamic reaction balance of CRISPR/Cas12a and RPA in one pot: a key to switch nucleic acid quantification. ACS Sens. 9, 3511–3519 (2024).
Kordalewska, M. et al. Understanding echinocandin resistance in the emerging pathogen Candida auris. Antimicrob. Agents Chemother. 62, e00238-18 (2018).
Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts CLSI M27 (CLSI, 2017).
Wasylyshyn, A. & Stoneman, E. K. Management of Candida auris. JAMA 331, 611–612 (2024).
Berkow, E. L., Lockhart, S. R. & Ostrosky-Zeichner, L. Antifungal susceptibility testing: current approaches. Clin. Microbiol. Rev. 33, e00069-19 (2020).
Duvallet, C. et al. Nationwide trends in COVID-19 cases and SARS-CoV-2 RNA wastewater concentrations in the United States. ACS ES T Water 2, 1899–1909 (2022).
Addressing Foodborne Threats to Health: Policies, Practices, and Global Coordination: Workshop Summary (National Academies Press, 2006).
Jui-Cheng, Y., Fu-Juay, C. & Shyang, C. A new criterion for automatic multilevel thresholding. IEEE Trans. Image Process. 4, 370–378 (1995).
Rolando, J. C. et al. Example data for running the dSHERLOCK data analysis pipeline. figshare https://doi.org/10.6084/m9.figshare.29944823 (2025).
Rolando, J. C. et al. dSHERLOCK repository. GitHub https://github.com/Walt-Lab/dSherlock (2025).
Rolando, J. C. et al. dSHERLOCK compute capsule. Code Ocean https://doi.org/10.24433/CO.6935105 (2025).
Rolando, J. C. et al. Digital CRISPR-based diagnostics for quantification of Candida auris and resistance mutations. Source Code. Zenodo https://doi.org/10.5281/zenodo.17153993 (2025).
Acknowledgements
Digital Droplet PCR measurements were performed by the Boston Children’s Hospital Intellectual and Developmental Disabilities Research Center Molecular Genetics Core Facility. The research described in this manuscript was supported by grants from The Wyss Institute for Bioinspired Engineering (J.C.R., A.T., D.R.W.), Health Research Inc. (GR110013801, N.E.W., N.K., H.d.P., X.T., J.J.C.), the New York State Department of Health, and the Wadsworth Center Division of Infectious Diseases (WC-2019-01, N.E.W., N.K., H.d.P., X.T., J.J.C.). N.K. was supported by the Wyss Technology Development Fellowship. X.T. acknowledges funding from the NIH NIDDK (5K08DK132516).
Author information
Authors and Affiliations
Contributions
J.C.R, A.T. and N.E.W. conceived the technology, designed and conducted experiments, analysed results and prepared the manuscript. N.K. participated in study design, conducted experimental work, contributed to data analysis and provided materials essential to the study. H.d.P. and X.T. submitted the initial proposal and performed computational primer analyses. H.d.P. designed gRNA sequences and primers, conducted initial experiments and analysed the resulting data. E.C. prepared simulated swab samples, collected residual clinical composite surveillance swabs and characterized their molecular properties. V.C. helped design initial experiments, data analysis and interpretations. J.J.C. and D.R.W. jointly supervised the research. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.J.C. and D.R.W. had financial interests in and served on the board of Sherlock Biosciences Corporation, a company that develops CRISPR diagnostics; Sherlock Biosciences was acquired by OraSure Technologies. D.R.W.’s interests are reviewed and managed by Mass General Brigham and Harvard University in accordance with their conflict-of-interest policies. Harvard College and Brigham and Women’s Hospital have filed Provisional Patent 63/764,169, on behalf of J.C.R., A.T. and D.R.W. related to the multiplexed single-molecule CRISPR detection described in this manuscript. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Ruijie Dengvand, Tony Hu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary text, tables and figures.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rolando, J.C., Thieme, A., Weckman, N.E. et al. Digital CRISPR-based diagnostics for quantification of Candida auris and resistance mutations. Nat. Biomed. Eng (2026). https://doi.org/10.1038/s41551-025-01597-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41551-025-01597-0
This article is cited by
-
Rapid quantification of both fungal abundance and drug resistance via reaction kinetics
Nature Biomedical Engineering (2026)