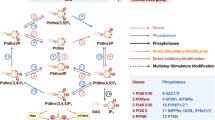
Abstract
Cell motility and adhesion are fundamental components for diverse physiological functions, including embryonic development, immune responses and tissue repair. Dysregulation of these processes can lead to a range of diseases, including cancer. Cell motility and adhesion are complex and often require regulation by an intricate network of signalling pathways, with phosphatidylinositol phosphates (PIPs) having a central role. PIPs are derived from phosphatidylinositol phosphorylation and are instrumental in mediating membrane dynamics, intracellular trafficking, cytoskeletal organization and signal transduction, all of which are crucial for cellular responses to environmental stimuli. Here we discuss the mechanisms through which PIPs modulate cell motility and adhesion by examining their roles at focal adhesions, within the cytoskeleton, at protein scaffolds and in the nucleus. By providing a comprehensive overview of PIP signalling, this Review underscores their significance in maintaining cellular homeostasis and highlights their potential as therapeutic targets in diseases characterized by aberrant cell motility and adhesion.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gumbiner, B. M. Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell 84, 345–357 (1996).
Conway, J. R. W. & Jacquemet, G. Cell matrix adhesion in cell migration. Essays Biochem. 63, 535–551 (2019).
Shewan, A., Eastburn, D. J. & Mostov, K. Phosphoinositides in cell architecture. Cold Spring Harb. Perspect. Biol. 3, a004796 (2011).
Hokin, M. R. & Hokin, L. E. Enzyme secretion and the incorporation of P32 into phospholipides of pancreas slices. J. Biol. Chem. 203, 967–977 (1953).
Di Paolo, G. & De Camilli, P. Phosphoinositides in cell regulation and membrane dynamics. Nature 443, 651–657 (2006).
Posor, Y., Jang, W. & Haucke, V.Phosphoinositides as membrane organizers. Nat. Rev. Mol. Cell Biol. 23, 797–816 (2022).
Chen, M. et al. The nuclear phosphoinositide response to stress. Cell Cycle 19, 268–289 (2020).
Manning, B. D. & Toker, A. AKT/PKB signaling: navigating the network. Cell 169, 381–405 (2017).
Liu, P. et al. PtdIns(3,4,5)P3-dependent activation of the mTORC2 kinase complex. Cancer Discov. 5, 1194–1209 (2015).
Currie, R. A. et al. Role of phosphatidylinositol 3,4,5-trisphosphate in regulating the activity and localization of 3-phosphoinositide-dependent protein kinase-1. Biochem. J. 337, 575–583 (1999).
Li, J. et al. Phosphorylation of ACAP1 by Akt regulates the stimulation-dependent recycling of integrin β1 to control cell migration. Dev. Cell 9, 663–673 (2005).
Kwon, T., Kwon, D. Y., Chun, J., Kim, J. H. & Kang, S. S. Akt protein kinase inhibits Rac1–GTP binding through phosphorylation at serine 71 of Rac1. J. Biol. Chem. 275, 423–428 (2000).
Zhu, Q. S. et al. Vimentin is a novel AKT1 target mediating motility and invasion. Oncogene 30, 457–470 (2011).
Bilanges, B., Posor, Y. & Vanhaesebroeck, B. PI3K isoforms in cell signalling and vesicle trafficking. Nat. Rev. Mol. Cell Biol. 20, 515–534 (2019).
Vanhaesebroeck, B., Perry, M. W. D., Brown, J. R., André, F. & Okkenhaug, K.PI3K inhibitors are finally coming of age. Nat. Rev. Drug Discov. 20, 741–769 (2021).
Thapa, N. et al. Phosphatidylinositol-3-OH kinase signalling is spatially organized at endosomal compartments by microtubule-associated protein 4. Nat. Cell Biol. 22, 1357–1370 (2020).
Xu, Q. et al. Phosphatidylinositol phosphate kinase PIPKIγ and phosphatase INPP5E coordinate initiation of ciliogenesis. Nat. Commun. 7, 10777 (2016).
Ling, K., Doughman, R. L., Firestone, A. J., Bunce, M. W. & Anderson, R. A. Type I gamma phosphatidylinositol phosphate kinase targets and regulates focal adhesions. Nature 420, 89–93 (2002).
Schumacher, S., Vazquez Nunez, R., Biertumpfel, C. & Mizuno, N. Bottom-up reconstitution of focal adhesion complexes. FEBS J. 289, 3360–3373 (2022).
Chen, S. et al. Roles of focal adhesion proteins in skeleton and diseases. Acta Pharm. Sin. B 13, 998–1013 (2023).
Hynes, R. O. Integrins: bidirectional, allosteric signaling machines. Cell 110, 673–687 (2002).
Legerstee, K. & Houtsmuller, A. B. A layered view on focal adhesions. Biology 10, 1189 (2021).
Geiger, B. & Bershadsky, A. Assembly and mechanosensory function of focal contacts. Curr. Opin. Cell Biol. 13, 584–592 (2001).
Winograd-Katz, S. E., Fassler, R., Geiger, B. & Legate, K. R. The integrin adhesome: from genes and proteins to human disease. Nat. Rev. Mol. Cell Biol. 15, 273–288 (2014).
Zaidel-Bar, R., Itzkovitz, S., Ma’ayan, A., Iyengar, R. & Geiger, B. Functional atlas of the integrin adhesome. Nat. Cell Biol. 9, 858–867 (2007).
Zaidel-Bar, R. & Geiger, B. The switchable integrin adhesome. J. Cell Sci. 123, 1385–1388 (2010).
Di Paolo, G. et al. Recruitment and regulation of phosphatidylinositol phosphate kinase type 1γ by the FERM domain of talin. Nature 420, 85–89 (2002).
Gilmore, A. P. & Burridge, K. Regulation of vinculin binding to talin and actin by phosphatidyl-inositol-4-5-bisphosphate. Nature 381, 531–535 (1996).
Senju, Y. & Lappalainen, P.Regulation of actin dynamics by PI(4,5)P2 in cell migration and endocytosis. Curr. Opin. Cell Biol. 56, 7–13 (2019).
Mandal, K. Review of PIP2 in cellular signaling, functions and diseases. Int. J. Mol. Sci. 21, 8342 (2020).
Zhao, Y., Lykov, N. & Tzeng, C. Talin‑1 interaction network in cellular mechanotransduction (review). Int. J. Mol. Med. 49, 60 (2022).
Martel, V. et al. Conformation, localization, and integrin binding of talin depend on its interaction with phosphoinositides. J. Biol. Chem. 276, 21217–21227 (2001).
Ye, X., McLean, M. A. & Sligar, S. G. Phosphatidylinositol 4,5-bisphosphate modulates the affinity of talin-1 for phospholipid bilayers and activates its autoinhibited form. Biochemistry 55, 5038–5048 (2016).
Wang, J. H.Pull and push: talin activation for integrin signaling. Cell Res. 22, 1512–1514 (2012).
Idevall-Hagren, O., Dickson, E. J., Hille, B., Toomre, D. K. & De Camilli, P. Optogenetic control of phosphoinositide metabolism. Proc. Natl Acad. Sci. USA 109, E2316–E2323 (2012).
Thapa, N. et al. Phosphoinositide signaling regulates the exocyst complex and polarized integrin trafficking in directionally migrating cells. Dev. Cell 22, 116–130 (2012).
De Pereda, J. M. et al. Structural basis for phosphatidylinositol phosphate kinase type Iγ binding to talin at focal adhesions. J. Biol. Chem. 280, 8381–8386 (2005).
Calderwood, D. A. et al. The phosphotyrosine binding-like domain of talin activates integrins. J. Biol. Chem. 277, 21749–21758 (2002).
Legate, K. R. et al. Integrin adhesion and force coupling are independently regulated by localized PtdIns(4,5)2 synthesis. EMBO J. 30, 4539–4553 (2011).
Ziegler, W. H., Liddington, R. C. & Critchley, D. R. The structure and regulation of vinculin. Trends Cell Biol. 16, 453–460 (2006).
Fukami, K., Endo, T., Imamura, M. & Takenawa, T. α-Actinin and vinculin are PIP2-binding proteins involved in signaling by tyrosine kinase. J. Biol. Chem. 269, 1518–1522 (1994).
Thompson, P. M. et al. A structural model for vinculin insertion into PIP2-containing membranes and the effect of insertion on vinculin activation and localization. Structure 25, 264–275 (2017).
Chinthalapudi, K. et al. Lipid binding promotes oligomerization and focal adhesion activity of vinculin. J. Cell Biol. 207, 643–656 (2014).
Wehrle-Haller, B. Assembly and disassembly of cell matrix adhesions. Curr. Opin. Cell Biol. 24, 569–581 (2012).
Chao, W. T. et al. Type I phosphatidylinositol phosphate kinase beta regulates focal adhesion disassembly by promoting β1 integrin endocytosis. Mol. Cell. Biol. 30, 4463–4479 (2010).
Kerstein, P. C., Patel, K. M. & Gomez, T. M. Calpain-mediated proteolysis of talin and FAK regulates adhesion dynamics necessary for axon guidance. J. Neurosci. 37, 1568–1580 (2017).
Franco, S. J. et al. Calpain-mediated proteolysis of talin regulates adhesion dynamics. Nat. Cell Biol. 6, 977–983 (2004).
Shao, H. et al. Spatial localization of m-calpain to the plasma membrane by phosphoinositide biphosphate binding during epidermal growth factor receptor-mediated activation. Mol. Cell. Biol. 26, 5481–5496 (2006).
Ezratty, E. J., Bertaux, C., Marcantonio, E. E. & Gundersen, G. G. Clathrin mediates integrin endocytosis for focal adhesion disassembly in migrating cells. J. Cell Biol. 187, 733–747 (2009).
Nader, G. P. F., Ezratty, E. J. & Gundersen, G. G. FAK, talin and PIPKIγ regulate endocytosed integrin activation to polarize focal adhesion assembly. Nat. Cell Biol. 18, 491–503 (2016).
Posor, Y. et al. Local synthesis of the phosphatidylinositol-3,4-bisphosphate lipid drives focal adhesion turnover. Dev. Cell 57, 1694–1711.e7 (2022).
D’Souza, R. S. et al. Calcium-stimulated disassembly of focal adhesions mediated by an ORP3/IQSec1 complex. eLife 9, e54113 (2020).
Pollard, T. D. & Borisy, G. G. Cellular motility driven by assembly and disassembly of actin filaments. Cell 112, 453–465 (2003).
Hilpelä, P., Vartiainen, M. K. & Lappalainen, P. Regulation of the actin cytoskeleton by PI(4,5)P2 and PI(3,4,5)P3. Curr. Top. Microbiol. Immunol. 282, 117–163 (2004).
Welch, H. C. et al. P-Rex1, a PtdIns(3,4,5)P3- and Gβγ-regulated guanine-nucleotide exchange factor for Rac. Cell 108, 809–821 (2002).
Lawson, C. D. & Ridley, A. J. Rho GTPase signaling complexes in cell migration and invasion. J. Cell Biol. 217, 447–457 (2018).
Van Keymeulen, A. et al. To stabilize neutrophil polarity, PIP3 and Cdc42 augment RhoA activity at the back as well as signals at the front. J. Cell Biol. 174, 437–445 (2006).
Srinivasan, S. et al. Rac and Cdc42 play distinct roles in regulating PI(3,4,5)P3 and polarity during neutrophil chemotaxis. J. Cell Biol. 160, 375–385 (2003).
Suetsugu, S., Kurisu, S. & Takenawa, T.Dynamic shaping of cellular membranes by phospholipids and membrane-deforming proteins. Physiol. Rev. 94, 1219–1248 (2014).
Saarikangas, J., Zhao, H. & Lappalainen, P. Regulation of the actin cytoskeleton–plasma membrane interplay by phosphoinositides. Physiol. Rev. 90, 259–289 (2010).
Giraud, Q. & Laporte, J. Amphiphysin-2 (BIN1) functions and defects in cardiac and skeletal muscle. Trends Mol. Med. 30, 579–591 (2024).
Coumans, J. V. F., Davey, R. J. & Moens, P. D. J. Cofilin and profilin: partners in cancer aggressiveness. Biophys. Rev. 10, 1323–1335 (2018).
Janmey, P. A., Bucki, R. & Radhakrishnan, R. Regulation of actin assembly by PI(4,5)P2 and other inositol phospholipids: an update on possible mechanisms. Biochem. Biophys. Res. Commun. 506, 307–314 (2018).
Senju, Y. et al. Mechanistic principles underlying regulation of the actin cytoskeleton by phosphoinositides. Proc. Natl Acad. Sci. USA 114, E8977–E8986 (2017).
Heiss, S. G. & Cooper, J. A. Regulation of CapZ, an actin capping protein of chicken muscle, by anionic phospholipids. Biochemistry 30, 8753–8758 (1991).
Solís, C. & Russell, B. CapZ integrates several signaling pathways in response to mechanical stiffness. J. Gen. Physiol. 151, 660–669 (2019).
Hartman, T. J., Martin, J. L., Solaro, R. J., Samarel, A. M. & Russell, B. CapZ dynamics are altered by endothelin-1 and phenylephrine via PIP2- and PKC-dependent mechanisms. Am. J. Physiol. Cell Physiol. 296, C1034–C1039 (2009).
Bear, J. E., Krause, M. & Gertler, F. B. Regulating cellular actin assembly. Curr. Opin. Cell Biol. 13, 158–166 (2001).
Bilkova, E. et al. Calcium directly regulates phosphatidylinositol 4,5-bisphosphate headgroup conformation and recognition. J. Am. Chem. Soc. 139, 4019–4024 (2017).
Tsai, F. C. & Meyer, T. Ca2+ pulses control local cycles of lamellipodia retraction and adhesion along the front of migrating cells. Curr. Biol. 22, 837–842 (2012).
Herrmann, H. & Aebi, U. Intermediate filaments: molecular structure, assembly mechanism, and integration into functionally distinct intracellular scaffolds. Annu. Rev. Biochem. 73, 749–789 (2004).
Hohmann, T. & Dehghani, F. The cytoskeleton-A complex interacting meshwork. Cells 8, 362 (2019).
Fortier, A. M., Asselin, E. & Cadrin, M. Keratin 8 and 18 loss in epithelial cancer cells increases collective cell migration and cisplatin sensitivity through claudin1 up-regulation. J. Biol. Chem. 288, 11555–11571 (2013).
Ku, N. O., Toivola, D. M., Strnad, P. & Omary, M. B. Cytoskeletal keratin glycosylation protects epithelial tissue from injury. Nat. Cell Biol. 12, 876–885 (2010).
Ostrowska-Podhorodecka, Z., Ding, I., Norouzi, M. & McCulloch, C. A. Impact of vimentin on regulation of cell signaling and matrix remodeling. Front. Cell Dev. Biol. 10, 869069 (2022).
Bargagna-Mohan, P. et al. The tumor inhibitor and antiangiogenic agent withaferin A targets the intermediate filament protein vimentin. Chem. Biol. 14, 623–634 (2007).
Kong, L. et al. Lamin A/C protein is overexpressed in tissue-invading prostate cancer and promotes prostate cancer cell growth, migration and invasion through the PI3K/AKT/PTEN pathway. Carcinogenesis 33, 751–759 (2012).
Escudeiro-Lopes, S., Filimonenko, V. V., Jarolimova, L. & Hozak, P. Lamin A/C and PI(4,5)P2—a novel complex in the cell nucleus. Cells 13, 399 (2024).
Swaffer, M. P. et al. RNA polymerase II dynamics and mRNA stability feedback scale mRNA amounts with cell size. Cell 186, 5254–5268.e26 (2023).
Akhmanova, A. & Kapitein, L. C. Mechanisms of microtubule organization in differentiated animal cells. Nat. Rev. Mol. Cell Biol. 23, 541–558 (2022).
Xue, G. & Hemmings, B. A. PKB/Akt-dependent regulation of cell motility. J. Natl Cancer Inst. 105, 393–404 (2013).
Thapa, N., Chen, M., Cryns, V. L. & Anderson, R. A p85 isoform switch enhances PI3K activation on endosomes by a MAP4- and PI3P-dependent mechanism. Cell Rep. 43, 114119 (2024).
Taniguchi, C. M. et al. The phosphoinositide 3-kinase regulatory subunit p85α can exert tumor suppressor properties through negative regulation of growth factor signaling. Cancer Res. 70, 5305–5315 (2010).
Thorpe, L. M. et al. PI3K–p110α mediates the oncogenic activity induced by loss of the novel tumor suppressor PI3K–p85α. Proc. Natl Acad. Sci. USA 114, 7095–7100 (2017).
Posor, Y. et al. Spatiotemporal control of endocytosis by phosphatidylinositol-3,4-bisphosphate. Nature 499, 233–237 (2013).
Klippel, A., Kavanaugh, W. M., Pot, D. & Williams, L. T. A specific product of phosphatidylinositol 3-kinase directly activates the protein kinase Akt through its pleckstrin homology domain. Mol. Cell. Biol. 17, 338–344 (1997).
Jo, H., Loison, F. & Luo, H. R. Microtubule dynamics regulates Akt signaling via dynactin p150. Cell Signal. 26, 1707–1716 (2014).
Ayloo, S. et al. Dynactin functions as both a dynamic tether and brake during dynein-driven motility. Nat. Commun. 5, 4807 (2014).
Chen, M. et al. Isthmin targets cell-surface GRP78 and triggers apoptosis via induction of mitochondrial dysfunction. Cell Death Differ. 21, 797–810 (2014).
Chen, M. et al. Extracellular anti-angiogenic proteins augment an endosomal protein trafficking pathway to reach mitochondria and execute apoptosis in HUVECs. Cell Death Differ. 25, 1905–1920 (2018).
Eden, E. R., White, I. J., Tsapara, A. & Futter, C. E. Membrane contacts between endosomes and ER provide sites for PTP1B–epidermal growth factor receptor interaction. Nat. Cell Biol. 12, 267–272 (2010).
Chaumet, A. et al. Nuclear envelope-associated endosomes deliver surface proteins to the nucleus. Nat. Commun. 6, 8218 (2015).
Beaty, B. T. et al. β1 integrin regulates Arg to promote invadopodial maturation and matrix degradation. Mol. Biol. Cell 24, 1661–1675 (2013).
Hsu, J. W. et al. The protein kinase Akt acts as a coat adaptor in endocytic recycling. Nat. Cell Biol. 22, 927–933 (2020).
Popova, J. S., Greene, A. K., Wang, J. & Rasenick, M. M. Phosphatidylinositol 4,5-bisphosphate modifies tubulin participation in phospholipase Cβ1 signaling. J. Neurosci. 22, 1668–1678 (2002).
Lin, T. C., Kuo, H. H., Wu, Y. C., Pan, T. S. & Yih, L. H. Phosphatidylinositol-5-phosphate 4-kinase gamma accumulates at the spindle pole and prevents microtubule depolymerization. Cell Div. 14, 9 (2019).
Wang, Y. et al. PLCB1 enhances cell migration and invasion in gastric cancer via regulating actin cytoskeletal remodeling and epithelial–mesenchymal transition. Biochem. Genet. 61, 2618–2632 (2023).
Thines, L., Roushar, F. J., Hedman, A. C. & Sacks, D. B. The IQGAP scaffolds: critical nodes bridging receptor activation to cellular signaling. J. Cell Biol. 222, e202205062 (2023).
Ande, S. R., Gu, Y., Nyomba, B. L. & Mishra, S. Insulin induced phosphorylation of prohibitin at tyrosine 114 recruits Shp1. Biochim. Biophys. Acta 1793, 1372–1378 (2009).
Lemmens, L. J. M., Ottmann, C. & Brunsveld, L. Conjugated protein domains as engineered scaffold proteins. Bioconjug. Chem. 31, 1596–1603 (2020).
Hu, J. et al. Resolution of structure of PIP5K1A reveals molecular mechanism for its regulation by dimerization and dishevelled. Nat. Commun. 6, 8205 (2015).
Nascimbeni, A. C., Codogno, P. & Morel, E. Phosphatidylinositol-3-phosphate in the regulation of autophagy membrane dynamics. FEBS J. 284, 1267–1278 (2017).
Wang, X., Hills, L. B. & Huang, Y. H.Lipid and protein co-regulation of PI3K effectors Akt and Itk in lymphocytes. Front. Immunol. 6, 117 (2015).
Choi, S. et al. Agonist-stimulated phosphatidylinositol-3,4,5-trisphosphate generation by scaffolded phosphoinositide kinases. Nat. Cell Biol. 18, 1324–1335 (2016).
Roy, M., Li, Z. & Sacks, D. B. IQGAP1 is a scaffold for mitogen-activated protein kinase signaling. Mol. Cell. Biol. 25, 7940–7952 (2005).
Choi, S. & Anderson, R. A. And Akt-ion! IQGAP1 in control of signaling pathways. EMBO J. 36, 967–969 (2017).
Chen, M. et al. The specificity of EGF-stimulated IQGAP1 scaffold towards the PI3K–Akt pathway is defined by the IQ3 motif. Sci. Rep. 9, 9126 (2019).
Chen, F. et al. IQGAP1 is overexpressed in hepatocellular carcinoma and promotes cell proliferation by Akt activation. Exp. Mol. Med. 42, 477–483 (2010).
Tekletsadik, Y. K., Sonn, R. & Osman, M. A. A conserved role of IQGAP1 in regulating TOR complex 1. J. Cell Sci. 125, 2041–2052 (2012).
Zhou, H. & Huang, S.Role of mTOR signaling in tumor cell motility, invasion and metastasis. Curr. Protein Pept. Sci. 12, 30–42 (2011).
Choi, S. et al. IQGAP1 is a novel phosphatidylinositol 4,5 bisphosphate effector in regulation of directional cell migration. EMBO J. 32, 2617–2630 (2013).
Watanabe, T. et al. Interaction with IQGAP1 links APC to Rac1, Cdc42, and actin filaments during cell polarization and migration. Dev. Cell 7, 871–883 (2004).
Holz, M. K., Ballif, B. A., Gygi, S. P. & Blenis, J. mTOR and S6K1 mediate assembly of the translation preinitiation complex through dynamic protein interchange and ordered phosphorylation events. Cell 123, 569–580 (2005).
Zhao, Y. et al. mTORC1 and mTORC2 converge on the Arp2/3 complex to promote KrasG12D-induced acinar-to-ductal metaplasia and early pancreatic carcinogenesis. Gastroenterology 160, 1755–1770.e17 (2021).
Baskaran, S. et al. Architecture and dynamics of the autophagic phosphatidylinositol 3-kinase complex. eLife 3, e05115 (2014).
Rong, Y. et al. Clathrin and phosphatidylinositol-4,5-bisphosphate regulate autophagic lysosome reformation. Nat. Cell Biol. 14, 924–934 (2012).
Du, W. et al. Kinesin 1 drives autolysosome tubulation. Dev. Cell 37, 326–336 (2016).
Tan, J. X. & Finkel, T. A phosphoinositide signalling pathway mediates rapid lysosomal repair. Nature 609, 815–821 (2022).
Ebner, M., Lucic, I., Leonard, T. A. & Yudushkin, I. PI(3,4,5)P3 engagement restricts Akt activity to cellular membranes. Mol. Cell 65, 416–431.e6 (2017).
Molina, J. R. et al. PTEN, NHERF1 and PHLPP form a tumor suppressor network that is disabled in glioblastoma. Oncogene 31, 1264–1274 (2012).
Byron, A. et al. Characterisation of a nucleo-adhesome. Nat. Commun. 13, 3053 (2022).
Petrie, R. J., Koo, H. & Yamada, K. M. Generation of compartmentalized pressure by a nuclear piston governs cell motility in a 3D matrix. Science 345, 1062–1065 (2014).
Ungricht, R. & Kutay, U. Mechanisms and functions of nuclear envelope remodelling. Nat. Rev. Mol. Cell Biol. 18, 229–245 (2017).
Chen, M. et al. A p53-phosphoinositide signalosome regulates nuclear AKT activation. Nat. Cell Biol. 24, 1099–1113 (2022).
Graham, D. M. & Burridge, K. Mechanotransduction and nuclear function. Curr. Opin. Cell Biol. 40, 98–105 (2016).
Janota, C. S., Calero-Cuenca, F. J. & Gomes, E. R. The role of the cell nucleus in mechanotransduction. Curr. Opin. Cell Biol. 63, 204–211 (2020).
Krause, M. et al. Cell migration through three-dimensional confining pores: speed accelerations by deformation and recoil of the nucleus. Philos. Trans. R Soc. Lond. B Biol. Sci. 374, 20180225 (2019).
Mendiratta, G. et al. Cancer gene mutation frequencies for the U.S. population. Nat. Commun. 12, 5961 (2021).
Blind, R. D. et al. The signaling phospholipid PIP3 creates a new interaction surface on the nuclear receptor SF-1. Proc. Natl Acad. Sci. USA 111, 15054–15059 (2014).
Chen, C., Chen, M., Wen, T., Anderson, R. A. & Cryns, V. L. Regulation of NRF2 by phosphoinositides and small heat shock proteins. Preprint at bioRxiv https://doi.org/10.1101/2023.10.26.564194 (2024).
Mellman, D. L. et al. A PtdIns4,5P2-regulated nuclear poly(A) polymerase controls expression of select mRNAs. Nature 451, 1013–1017 (2008).
Wang, Y. H. & Sheetz, M. P. When PIP2 meets p53: nuclear phosphoinositide signaling in the DNA damage response. Front. Cell Dev. Biol. 10, 903994 (2022).
Jung, O. et al. Nuclear phosphoinositide signaling promotes YAP/TAZ–TEAD transcriptional activity in breast cancer. EMBO J. 43, 1740–1769 (2024).
Wen, T., Chen, M., Cryns, V. L. & Anderson, R. A. Regulation of the poly(A) polymerase Star-PAP by a nuclear phosphoinositide signalosome. Preprint at bioRxiv https://doi.org/10.1101/2024.07.01.601467 (2024).
Fiume, R. et al. Nuclear phosphoinositides: their regulation and roles in nuclear functions. Int. J. Mol. Sci. 20, 2991 (2019).
Vidalle, M. C. et al. Nuclear phosphoinositides as key determinants of nuclear functions. Biomolecules 13, 1049 (2023).
Choi, S., Chen, M., Cryns, V. L. & Anderson, R. A. A nuclear phosphoinositide kinase complex regulates p53. Nat. Cell Biol. 21, 462–475 (2019).
Carrillo, N. D. et al. Lipid transfer proteins and a PI 4-kinase initiate nuclear phosphoinositide signaling. Preprint at bioRxiv https://doi.org/10.1101/2023.05.08.539894 (2025).
Chen, M., Horn, H. T., Wen, T., Cryns, V. L. & Anderson, R. A.Assessing in situ phosphoinositide–protein interactions through fluorescence proximity ligation assay in cultured cells. Methods Mol. Biol. 2251, 133–142 (2021).
Lemmon, M. A. Pleckstrin homology (PH) domains and phosphoinositides. Biochem. Soc. Symp. 74, 81–93 (2007).
Chen, J. et al. CBX3 promotes clear cell renal carcinoma through PI3K/AKT activation and aberrant immunity. J. Transl. Med. 21, 600 (2023).
Wang, H. et al. Ginsenoside compound K promotes proliferation, migration and differentiation of Schwann cells via the activation of MEK/ERK1/2 and PI3K/AKT pathways. Neurochem. Res. 46, 1400–1409 (2021).
Zhu, J. F. et al. Annexin A1-suppressed autophagy promotes nasopharyngeal carcinoma cell invasion and metastasis by PI3K/AKT signaling activation. Cell Death Dis. 9, 1154 (2018).
Martelli, A. M. et al. The emerging multiple roles of nuclear Akt. Biochim. Biophys. Acta 1823, 2168–2178 (2012).
Freeman, D. J. et al. PTEN tumor suppressor regulates p53 protein levels and activity through phosphatase-dependent and -independent mechanisms. Cancer Cell 3, 117–130 (2003).
Ehm, P., Nalaskowski, M. M., Wundenberg, T. & Jucker, M. The tumor suppressor SHIP1 colocalizes in nucleolar cavities with p53 and components of PML nuclear bodies. Nucleus 6, 154–164 (2015).
Li, W. et al. Star-PAP control of BIK expression and apoptosis is regulated by nuclear PIPKIα and PKCδ signaling. Mol. Cell 45, 25–37 (2012).
Li, W. et al. Distinct regulation of alternative polyadenylation and gene expression by nuclear poly(A) polymerases. Nucleic Acids Res. 45, 8930–8942 (2017).
Duan, A. et al. Star-PAP regulates tumor protein D52 through modulating miR-449a/34a in breast cancer. Biol. Open 8, bio045914 (2019).
Paez, A. V. et al. Heme oxygenase-1 in the forefront of a multi-molecular network that governs cell–cell contacts and filopodia-induced zippering in prostate cancer. Cell Death Dis. 7, e2570 (2016).
Hughes, C. H. K. et al. Steroidogenic factor 1 (SF-1; Nr5a1) regulates the formation of the ovarian reserve. Proc. Natl Acad. Sci. USA 120, e2220849120 (2023).
Blind, R. D., Suzawa, M. & Ingraham, H. A. Direct modification and activation of a nuclear receptor–PIP2 complex by the inositol lipid kinase IPMK. Sci. Signal. 5, ra44 (2012).
Kim, A. C. et al. Targeted disruption of β-catenin in Sf1-expressing cells impairs development and maintenance of the adrenal cortex. Development 135, 2593–2602 (2008).
Yang, C. M. et al. β-Catenin promotes cell proliferation, migration, and invasion but induces apoptosis in renal cell carcinoma. Onco Targets Ther. 10, 711–724 (2017).
De Santa Barbara, P. et al. Direct interaction of SRY-related protein SOX9 and steroidogenic factor 1 regulates transcription of the human anti-Müllerian hormone gene. Mol. Cell. Biol. 18, 6653–6665 (1998).
Capaccione, K. M. et al. Sox9 mediates Notch1-induced mesenchymal features in lung adenocarcinoma. Oncotarget 5, 3636–3650 (2014).
Ma, Q. Role of Nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 53, 401–426 (2013).
Ning, S. et al. The mechanism of NRF2 regulating cell proliferation and mesenchymal transformation in pulmonary hypertension. Int. J. Biol. Macromol. 275, 133514 (2024).
Zanconato, F., Cordenonsi, M. & Piccolo, S. YAP/TAZ at the roots of cancer. Cancer Cell 29, 783–803 (2016).
Zanconato, F., Battilana, G., Cordenonsi, M. & Piccolo, S. YAP/TAZ as therapeutic targets in cancer. Curr. Opin. Pharmacol. 29, 26–33 (2016).
Zanconato, F. et al. Transcriptional addiction in cancer cells is mediated by YAP/TAZ through BRD4. Nat. Med. 24, 1599–1610 (2018).
Naba, A.Mechanisms of assembly and remodelling of the extracellular matrix. Nat. Rev. Mol. Cell Biol. 25, 865–885 (2024).
Malik, A. R., Liszewska, E. & Jaworski, J. Matricellular proteins of the Cyr61/CTGF/NOV (CCN) family and the nervous system. Front. Cell Neurosci. 9, 237 (2015).
Haugh, J. M., Codazzi, F., Teruel, M. & Meyer, T. Spatial sensing in fibroblasts mediated by 3′ phosphoinositides. J. Cell Biol. 151, 1269–1280 (2000).
Hammond, G. R. & Hong, Y. Phosphoinositides and membrane targeting in cell polarity. Cold Spring Harb. Perspect. Biol. 10, a027938 (2018).
Rascio, F. et al. The pathogenic role of PI3K/AKT pathway in cancer onset and drug resistance: an updated review. Cancers 13, 3949 (2021).
Morgan, T. M., Koreckij, T. D. & Corey, E. Targeted therapy for advanced prostate cancer: inhibition of the PI3K/Akt/mTOR pathway. Curr. Cancer Drug Targets 9, 237–249 (2009).
Manfredi, G. I. et al. PI3K/Akt/mTOR signaling in medullary thyroid cancer: a promising molecular target for cancer therapy. Endocrine 48, 363–370 (2015).
Iranpanah, A. et al. The exosome-mediated PI3K/Akt/mTOR signaling pathway in neurological diseases. Pharmaceutics 15, 1006 (2023).
Kakar, R., Ghosh, C. & Sun, Y. Phosphoinositide signaling in immune cell migration. Biomolecules 13, 1705 (2023).
Janku, F., Yap, T. A. & Meric-Bernstam, F. Targeting the PI3K pathway in cancer: are we making headway? Nat. Rev. Clin. Oncol. 15, 273–291 (2018).
Davoodi-Moghaddam, Z., Jafari-Raddani, F., Delshad, M., Pourbagheri-Sigaroodi, A. & Bashash, D. Inhibitors of the PI3K/AKT/mTOR pathway in human malignancies; trend of current clinical trials. J. Cancer Res. Clin. Oncol. 149, 15293–15310 (2023).
Popova, N. V. & Jücker, M. The role of mTOR signaling as a therapeutic target in cancer. Int. J. Mol. Sci. 22, 1743 (2021).
Phillips, T. E. J. & Maguire, E. Phosphoinositides: roles in the development of microglial-mediated neuroinflammation and neurodegeneration. Front. Cell Neurosci. 15, 652593 (2021).
Zhou, P. et al. Phosphatidylinositol 4,5-bisphosphate alters pharmacological selectivity for epilepsy-causing KCNQ potassium channels. Proc. Natl Acad. Sci. USA 110, 8726–8731 (2013).
Kim, E., Beon, J., Lee, S., Park, J. & Kim, S. IPMK: a versatile regulator of nuclear signaling events. Adv. Biol. Regul. 61, 25–32 (2016).
Kalasova, I. et al. Tools for visualization of phosphoinositides in the cell nucleus. Histochem. Cell Biol. 145, 485–496 (2016).
Hammond, G. R. V., Ricci, M. M. C., Weckerly, C. C. & Wills, R. C. An update on genetically encoded lipid biosensors. Mol. Biol. Cell 33, tp2 (2022).
Acknowledgements
M.C. is supported by grants from the National Natural Science Foundation of China (32400577), Guangdong Basic and Applied Basic Research Foundation (2023A1515110237), Shenzhen Medical Research Fund (D2301007), Shenzhen Natural Science Foundation (JCYJ20240813094605008) and Medical Research Innovation Project (G030410001). J.S. is supported by grants from the National Natural Science Foundation of China (82071193), Guangdong Zhujiang Program (0920220233) and Science and Technology Foundation of Shenzhen (RCYX20210706092040044 and JCYJ20220818102611025). This work was supported in part by National Institutes of Health grants (R35GM134955 to R.A.A. and R01CA286492 to R.A.A. and V.L.C.), Department of Defense Breast Cancer Research Program grants (W81XWH-17-1-0258 and HT9425-23-1-0554 to R.A.A. and W81XWH-17-1-0259, W81XWH-21-1-0129 and HT9425-23-1-0553 to V.L.C.) and a grant from the Breast Cancer Research Foundation (to V.L.C.).
Author information
Authors and Affiliations
Contributions
X.H., C.R., J.J., X.L., J.S. and M.C. drafted the manuscript. X.H., Y.C., K.B., N.D.C., R.A.A., V.L.C., J.S. and M.C. edited the manuscript. X.H., C.R., J.J., X.L., Y.C., K.B., N.D.C., J.S. and M.C. visualized the data. R.A.A., V.L.C., J.S. and M.C. acquired funding. J.S. and M.C. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hou, X., Ren, C., Jin, J. et al. Phosphoinositide signalling in cell motility and adhesion. Nat Cell Biol 27, 736–748 (2025). https://doi.org/10.1038/s41556-025-01647-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41556-025-01647-4