Abstract
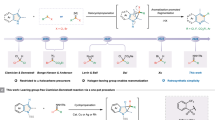
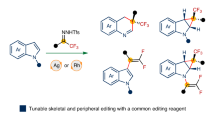
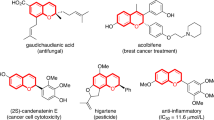
Heterocycle skeletal editing has recently emerged as a powerful tactic for achieving heterocycle-to-heterocycle transmutation without the need for multistep de novo heterocycle synthesis. However, the enantioselective skeletal editing of heteroarenes through single-atom logic remains challenging. Here we report the enantiodivergent dearomative skeletal editing of indoles and pyrroles via an asymmetric carbon-atom insertion, using trifluoromethyl N-triftosylhydrazones as carbene precursors. This strategy provides a straightforward methodology to access enantiomerically enriched six-membered N-heterocycles containing a trifluoromethylated quaternary stereocentre from planar N-heteroarenes. The synthetic utility of this enantiodivergent methodology was demonstrated by a broad evaluation of reaction scope, product derivatization and concise syntheses of drug analogues. Mechanistic studies reveal that the excellent asymmetric induction arises from the initial cyclopropanation step. The asymmetric single-atom insertion strategy is expected to have a broad impact on the field of single-atom skeletal editing and catalytic asymmetric dearomatization of aromatic compounds.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2261312 for (S)-34, 2266339 for (R)-34, 2281736 for (2R,3S)-43, 2281629 for (2R,3S)-44, 2350998 for (R)-64, 2311749 for (5S,6S)-76 and 2350447 for 82. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. DFT-optimized structures, Cartesian coordinates and energies have been deposited online and are freely available at https://doi.org/10.6084/m9.figshare.26820343.
References
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Zhuo, C.-X., Zhang, W. & You, S.-L. Catalytic asymmetric dearomatization reactions. Angew. Chem. Int. Ed. 51, 12662–12686 (2012).
Zheng, C. & You, S.-L. Catalytic asymmetric dearomatization by transition-metal catalysis: a method for transformations of aromatic compounds. Chem 1, 830–857 (2016).
Wertjes, W. C., Southgate, E. H. & Sarlah, D. Recent advances in chemical dearomatization of nonactivated arenes. Chem. Soc. Rev. 47, 7996–8017 (2018).
Huck, C. J. & Sarlah, D. Shaping molecular landscapes: recent advances, opportunities, and challenges in dearomatization. Chem 6, 1589–1603 (2020).
Zhuo, C.-X., Zheng, C. & You, S.-L. Transition-metal-catalyzed asymmetric allylic dearomatization reactions. Acc. Chem. Res. 47, 2558–2573 (2014).
Zheng, C. & You, S.-L. Catalytic asymmetric dearomatization (CADA) reaction-enabled total synthesis of indole-based natural products. Nat. Prod. Rep. 36, 1589–1605 (2019).
Liu, Y.-Z., Song, H., Zheng, C. & You, S.-L. Cascade asymmetric dearomative cyclization reactions via transition-metal-catalysis. Nat. Synth. 1, 203–216 (2022).
Zhu, M., Zhang, X., Zheng, C. & You, S.-L. Energy-transfer-enabled dearomative cycloaddition reactions of indoles/pyrroles via excited-state aromatics. Acc. Chem. Res. 55, 2510–2525 (2022).
Jiang, R., Ding, L., Zheng, C. & You, S.-L. Iridium-catalyzed Z-retentive asymmetric allylic substitution reactions. Science 371, 380–386 (2021).
Tu, H.-F., Zhang, X., Zheng, C., Zhu, M. & You, S.-L. Enantioselective dearomative prenylation of indole derivatives. Nat. Catal. 1, 601–608 (2018).
Zhang, Y.-Q. et al. Asymmetric dearomatization catalysed by chiral Brønsted acids via activation of ynamides. Nat. Chem. 13, 1093–1100 (2021).
Zhang, W.-Y., Wang, H.-C., Wang, Y., Zheng, C. & You, S.-L. Asymmetric dearomatization of indoles with azodicarboxylates via cascade electrophilic amination/aza-Prins cyclization/phenonium-like rearrangement. J. Am. Chem. Soc. 145, 11745–11753 (2023).
Beletskaya, I. P., Nájera, C. & Yus, M. Stereodivergent catalysis. Chem. Rev. 118, 5080–5200 (2018).
Cao, W., Feng, X. & Liu, X. Reversal of enantioselectivity in chiral metal complex-catalyzed asymmetric reactions. Org. Biomol. Chem. 17, 6538–6550 (2019).
Jurczyk, J. et al. Single-atom logic for heterocycle editing. Nat. Synth. 1, 352–364 (2022).
Peplow, M. ‘Almost magical’: chemists can now move single atoms in and out of a molecule’s core. Nature 618, 21–24 (2023).
Liu, Z., Sivaguru, P., Ning, Y., Wu, Y. & Bi, X. Skeletal editing of (hetero) arenes using carbenes. Chem. Eur. J. 29, e202301227 (2023).
Joynson, B. W. & Ball, L. T. Skeletal editing: interconversion of arenes and heteroarenes. Helv. Chim. Acta. 106, e202200182 (2023).
Roque, J. B., Kuroda, Y., Göttemann, L. T. & Sarpong, R. Deconstructive diversification of cyclic amines. Nature 564, 244–248 (2018).
Jurczyk, J. et al. Photomediated ring contraction of saturated heterocycles. Science 373, 1004–1012 (2021).
Bartholomew, G. L., Carpaneto, F. & Sarpong, R. Skeletal editing of pyrimidines to pyrazoles by formal carbon deletion. J. Am. Chem. Soc. 144, 22309–22315 (2022).
Wright, B. A. et al. Skeletal editing approach to bridge-functionalized bicyclo[1.1.1]pentanes from azabicyclo[2.1.1]hexanes. J. Am. Chem. Soc. 145, 10960–10966 (2023).
Kennedy, S. H., Dherange, B. D., Berger, K. J. & Levin, M. D. Skeletal editing through direct nitrogen deletion of secondary amines. Nature 593, 223–227 (2021).
Dherange, B. D., Kelly, P. Q., Liles, J. P., Sigman, M. S. & Levin, M. D. Carbon atom insertion into pyrroles and indoles promoted by chlorodiazirines. J. Am. Chem. Soc. 143, 11337–11344 (2021).
Woo, J. et al. Scaffold hopping by net photochemical carbon deletion of azaarenes. Science 376, 527–532 (2022).
Hyland, E. E., Kelly, P. Q., McKillop, A. M., Dherange, B. D. & Levin, M. D. Unified access to pyrimidines and quinazolines enabled by N–N cleaving carbon atom insertion. J. Am. Chem. Soc. 144, 19258–19264 (2022).
Pearson, T. J. et al. Aromatic nitrogen scanning by ipso-selective nitrene internalization. Science 381, 1474–1479 (2023).
Woo, J., Colin Stein, C., Christian, A. H. & Levin, M. D. Carbon-to-nitrogen single-atom transmutation of azaarenes. Nature 623, 77–82 (2023).
Reisenbauer, J. C., Green, O., Franchino, A., Finkelstein, P. & Morandi, B. Late-stage diversification of indole skeletons through nitrogen atom insertion. Science 377, 1104–1109 (2022).
Zhong, H. et al. Skeletal metalation of lactams through a carbonyl-to-nickel-exchange logic. Nat. Commun. 14, 5273 (2023).
Joynson, B. W., Cumming, G. R. & Ball, L. T. Photochemically mediated ring expansion of indoles and pyrroles with chlorodiazirines: synthetic methodology and thermal hazard assessment. Angew. Chem. Int. Ed. 62, e202305081 (2023).
Guo, H., Qiu, S. & Xu, P. One-carbon ring expansion of indoles and pyrroles: a straightforward access to 3-fluorinated quinolines and pyridines. Angew. Chem. Int. Ed. 63, e202317104 (2024).
Lyu, H., Kevlishvili, I., Yu, X., Liu, P. & Dong, G. Boron insertion into alkyl ether bonds via zinc/nickel tandem catalysis. Science 372, 175–182 (2021).
Wang, H. et al. Dearomative ring expansion of thiophenes by bicyclobutane insertion. Science 381, 75–81 (2023).
Piacentini, P., Bingham, T. W. & Sarlah, D. Dearomative ring expansion of polycyclic arenes. Angew. Chem. Int. Ed. 61, e202208014 (2022).
Mykura, P. et al. Synthesis of polysubstituted azepanes by dearomative ring expansion of nitroarenes. Nat. Chem. 16, 771–779 (2024).
Wu, F.-P. et al. Ring expansion of indene by photoredox-enabled functionalized carbon-atom insertion. Nat. Catal. 7, 242–251 (2024).
Manning, J. R. & Davies, H. M. L. One-pot synthesis of highly functionalized pyridines via a rhodium carbenoid induced ring expansion of isoxazoles. J. Am. Chem. Soc. 130, 8602–8603 (2008).
Liu, S. et al. Tunable molecular editing of indoles with fluoroalkyl carbenes. Nat. Chem. 16, 988–997 (2024).
Huang, M.-Y. & Zhu, S.-F. Catalytic reactions for enantioselective transfers of donor-substituted carbenes. Chem Catal. 2, 3112–3139 (2022).
Davies, H. M. L. & Liao, K. Dirhodium tetracarboxylates as catalysts for selective intermolecular C–H functionalization. Nat. Rev. Chem. 3, 347–360 (2019).
Sattler, A. & Parkin, G. Cleaving carbon–carbon bonds by inserting tungsten into unstrained aromatic rings. Nature 463, 523–526 (2010).
Liu, Z., Sivaguru, P., Zanoni, G. & Bi, X. N-Triftosylhydrazones: a new chapter for diazo-based carbene chemistry. Acc. Chem. Res. 55, 1763–1781 (2022).
Sivaguru, P. & Bi, X. Fluoroalkyl N-sulfonyl hydrazones: an efficient reagent for the synthesis of fluoroalkylated compounds. Sci. China Chem. 64, 1614–1629 (2021).
Nie, J., Guo, H.-C., Cahard, D. & Ma, J.-A. Asymmetric construction of stereogenic carbon centers featuring a trifluoromethyl group from prochiral trifluoromethylated substrates. Chem. Rev. 111, 455–529 (2011).
Yang, X., Wu, T., Phipps, R. J. & Toste, F. D. Advances in catalytic enantioselective fluorination, mono-, di-, and trifluoromethylation, and trifluoromethylthiolation reactions. Chem. Rev. 115, 826–870 (2015).
Delgado‐Rebollo, M., Prieto, A. & Peréz, P. J. Catalytic functionalization of indoles by copper‐mediated carbene transfer. ChemCatChem 6, 2047–2052 (2014).
Yang, Z., Möller, M. & Koenigs, R. M. Synthesis of gem-difluoro olefins through C–H functionalization and β-fluoride elimination reactions. Angew. Chem. Int. Ed. 59, 5572–5576 (2020).
Harada, S. et al. Silver(I)/dirhodium(II) catalytic platform for asymmetric N–H insertion reaction of heteroaromatics. J. Am. Chem. Soc. 146, 733–741 (2024).
Davies, H. M. & Spangler, J. E. Reactions of indoles with metal-bound carbenoids. Adv. Heterocycl. Chem. 110, 43–72 (2013).
Xu, H. et al. Highly enantioselective copper-and iron-catalyzed intramolecular cyclopropanation of indoles. J. Am. Chem. Soc. 139, 7697–7700 (2017).
Zhang, D. et al. Enantioselective palladium(II) phosphate catalyzed three-component reactions of pyrrole, diazoesters, and imines. Angew. Chem. Int. Ed. 52, 13356–13360 (2013).
Chem, Y.-K. et al. A catalyst-controlled enantiodivergent bromolactonization. J. Am. Chem. Soc. 143, 12745–12754 (2021).
Qin, C. & Davies, H. M. L. Role of sterically demanding chiral dirhodium catalysts in site-selective C–H functionalization of activated primary C–H bonds. J. Am. Chem. Soc. 136, 9792–9797 (2014).
Wertz, B., Ren, Z., Bacsa, J., Musaev, D. G. & Davies, H. M. L. Comparison of 1,2-diarylcyclopropanecarboxylates with 1,2,2-triarylcyclopropanecarboxylates as chiral ligands for dirhodium catalyzed cyclopropanation and C–H functionalization. J. Org. Chem. 85, 12199–12211 (2020).
Guo, Y. et al. Chiral phosphoric acid-catalyzed enantioselective phosphinylation of 3,4-dihydroisoquinolines with diarylphosphine oxides. Commun. Chem. 6, 26 (2023).
See, Y. Y., Dang, T. T., Chen, A. & Seayad, A. M. Concise synthesis of vesnarinone and its analogues by using Pd-catalyzed C–N bond-forming reactions. Eur. J. Org. Chem. 2014, 7405–7412 (2014).
Liao, K. et al. Site-selective and stereoselective functionalization of non-activated tertiary C–H bonds. Nature 551, 609–613 (2017).
Liao, K., Negretti, S., Musaev, D. G., Bacsa, J. & Davies, H. M. L. Site-selective and stereoselective functionalization of unactivated C–H bonds. Nature 533, 230–234 (2016).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Acknowledgements
Research reported in this publication was supported by NSFC (22331004 to X.B. and 22371035 to Z.L.), the Department of Science and Technology of Jilin Province (20230508054RC to Z.L. and 20240305092YY to Z.L.) and the Royal Society (Newton Advanced Fellowship NAF\R1\191210 to X.B. and E.A.).
Author information
Authors and Affiliations
Contributions
X.Z., S.L., Y.Y. and Y.N. performed and analysed the experiments. Q.S. carried out the DFT calculations. Z.L. and X.B. conceived the concept, and together with E.A., G.R. and P.S. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Yu-hong Lam, Guang-Jian Mei and the other, anonymous, reviewer for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Materials and methods, Supplementary Figs. 1–289, Tables 1–10 and spectral data for all new compounds.
Supplementary Data 1
Crystallographic data for compound S-34; CCDC reference 2261312.
Supplementary Data 2
Crystallographic data for compound R-34; CCDC reference 2266339.
Supplementary Data 3
Crystallographic data for compound 2R,3S-43; CCDC reference 2281736.
Supplementary Data 4
Crystallographic data for compound 2R,3S-44; CCDC reference 2281629.
Supplementary Data 5
Crystallographic data for compound 5S,6S-76; CCDC reference 2311749.
Supplementary Data 6
Crystallographic data for compound R-64; CCDC reference 2350998.
Supplementary Data 7
Crystallographic data for compound 82; CCDC reference 2350447.
Supplementary Data 8 (download ZIP )
The xyz coordinates of DFT calculations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Song, Q., Liu, S. et al. Asymmetric dearomative single-atom skeletal editing of indoles and pyrroles. Nat. Chem. 17, 215–225 (2025). https://doi.org/10.1038/s41557-024-01680-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-024-01680-0
This article is cited by
-
Photoinduced polarity-mismatched transformations of isoquinolines into naphthalenes
Nature Communications (2026)
-
Switchable skeletal editing of quinolines enabled by cyclizative sequential rearrangements
Nature Chemistry (2025)
-
Chiral hypervalent iodine catalyzed stereoselective skeletal editing of pyrimidine fused heterocycles
Nature Communications (2025)
-
Enantioselective dearomative single-atom skeletal editing of benzofurans
Nature Communications (2025)
-
Skeletal editing of pyridines to aryldialdehydes
Nature Communications (2025)