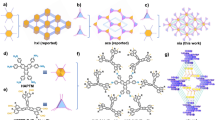
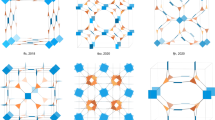
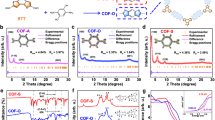
Abstract
Three-dimensional (3D) covalent organic frameworks (COFs) hold significant promise for a variety of applications. However, conventional design approaches using regular building blocks limit the structural diversity of 3D COFs. Here we design and synthesize two 3D COFs, designated as JUC-644 and JUC-645, through a methodology that relies on using eight-connected building blocks with reduced symmetry. Their structures are solved using continuous rotation electron diffraction and high-resolution transmission electron microscopy, which reveal a unique linkage with a double chain structure, a rare phenomenon in COFs. We deconstruct these structures into [4 + 3(+ 2)]-c nets, which leads to six different topologies. Furthermore, JUC-644 demonstrates high adsorption capacity for C3H8 and n-C4H10 (11.28 and 10.45 mmol g−1 at 298 K and 1 bar, respectively), surpassing most known porous materials, with notable selectivity for C3H8/C2H6 and n-C4H10/C2H6. This approach opens avenues for designing intricate architectures and shows the potential of COFs in C2H6 recovery from natural gas liquids.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2296805 (JUC-644) and 2353775 (JUC-645). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information, or from the corresponding authors on reasonable request. Source data are provided with this paper.
References
Cote, A. P. et al. Porous, crystalline, covalent organic frameworks. Science. 310, 1166–1170 (2005).
El-Kaderi, H. M. et al. Designed synthesis of 3D covalent organic frameworks. Science. 316, 268–272 (2007).
Ding, S.-Y. & Wang, W. Covalent organic frameworks (COFs): from design to applications. Chem. Soc. Rev. 42, 548–568 (2013).
Diercks, C. S. & Yaghi, O. M. The atom, the molecule, and the covalent organic framework. Science 355, eaall58 (2017).
Guan, X.-Y., Chen, F.-Q., Fang, Q.-R. & Qiu, S.-L. Design and applications of three dimensional covalent organic frameworks. Chem. Soc. Rev. 49, 1357–1384 (2020).
Colson, J. W. et al. Oriented 2D covalent organic framework thin films on single-layer graphene. Science. 332, 228–231 (2011).
Feng, X., Ding, X. S. & Jiang, D.-L. Covalent organic frameworks. Chem. Soc. Rev. 41, 6010–6022 (2012).
Huang, N., Wang, P. & Jiang, D.-L. Covalent organic frameworks: a materials platform for structural and functional designs. Nat. Rev. Mater. 1, 16068 (2016).
Guan, X.-Y. et al. Chemically stable polyarylether-based covalent organic frameworks. Nat. Chem. 11, 587–594 (2019).
Liu, Y.-Z. et al. A stable luminescent covalent organic framework nanosheet for sensitive molecular recognition. CCS Chem. 5, 2033–2045 (2022).
Wang, M.-C. et al. Exceptionally high charge mobility in phthalocyanine-based poly(benzimidazobenzophenanthroline)-ladder-type two-dimensional conjugated polymers. Nat. Mater. 22, 880–887 (2023).
Kang, X. et al. Reticular synthesis of tbo topology covalent organic frameworks. J. Am. Chem. Soc. 142, 16346–16356 (2020).
Zhang, Y.-B. et al. Single-crystal structure of a covalent organic framework. J. Am. Chem. Soc. 135, 16336–16339 (2013).
Ma, Y.-X. et al. A dynamic three-dimensional covalent organic framework. J. Am. Chem. Soc. 139, 4995–4998 (2017).
Gao, C., Li, J., Yin, S., Sun, J.-L. & Wang, C. Twist building blocks from planar to tetrahedral for the synthesis of covalent organic frameworks. J. Am. Chem. Soc. 142, 3718–3723 (2020).
Guan, X.-Y., Fang, Q.-R., Yan, Y.-S. & Qiu, S.-L. Functional regulation and stability engineering of three-dimensional covalent organic frameworks. Acc. Chem. Res. 55, 1912–1927 (2022).
Uribe-Romo, F. J. et al. A crystalline imine-linked 3-D porous covalent organic framework. J. Am. Chem. Soc. 131, 4570–4571 (2009).
Lin, G.-Q. et al. A pyrene-based, fluorescent three-dimensional covalent organic framework. J. Am. Chem. Soc. 138, 3302–3305 (2016).
Liu, Y.-Z. et al. Design and synthesis of a zeolitic organic framework. Angew. Chem. Int. Ed. 61, e202203584 (2022).
Xie, Y. et al. Tuning the topology of three-dimensional covalent organic frameworks via steric control: from pts to unprecedented Ijh. J. Am. Chem. Soc. 143, 7279–7284 (2021).
Gong, C.-T. et al. Synthesis and visualization of entangled 3D covalent organic frameworks with high-valency stereoscopic molecular nodes for gas separation. Angew. Chem. Int. Ed. 61, e202204899 (2022).
Jin, F.-Z. et al. Bottom-up synthesis of 8-connected three-dimensional covalent organic frameworks for highly efficient ethylene/ethane separation. J. Am. Chem. Soc. 144, 5643–5652 (2022).
Shan, Z. et al. 3D covalent organic frameworks with interpenetrated pcb topology based on 8-connected cubic nodes. J. Am. Chem. Soc. 144, 5728–5733 (2022).
Gropp, C., Ma, T.-Q., Hanikel, N. & Yaghi, O. M. Design of higher valency in covalent organic frameworks. Science 370, eabd6406 (2020).
Jin, F.-Z. et al. Rationally fabricating three-dimensional covalent organic frameworks for propyne/propylene separation. J. Am. Chem. Soc. 144, 23081–23088 (2022).
Das, S. et al. Three-dimensional covalent organic framework with scu-c topology for drug delivery. ACS Appl. Mater. Interfaces 42, 48045–48051 (2022).
Jin, F.-Z. et al. Rationally fabricating 3D porphyrinic covalent organic frameworks with scu topology as highly efficient photocatalysts. Chem 8, 3064–3080 (2022).
Wu, M.-M., Shan, Z., Wang, J.-J., Liu, T.-T. & Zhang, G. Three-dimensional covalent organic framework with tty topology for enhanced photocatalytic hydrogen peroxide production. Chem. Eng. J. 454, 140121 (2023).
Mian, L. et al. Topological analysis of metal–organic frameworks with polytopic linkers and/or multiple building units and the minimal transitivity principle. Chem. Soc. Rev. 114, 1343–1370 (2014).
Yang, S.-Y., Long, L.-S., Huang, R.-B. & Zheng, L.-S. [Zn8(SiO4)(C8H4O4)6]n: the firstborn of a metallosilicate-organic hybrid material family (C8H4O4 = isophthalate). Chem. Commun. 5, 472–473 (2002).
Yang, S.-Y. et al. An exceptionally stable metal-organic framework constructed from the Zn8(SiO4) core. Chem. Mater. 14, 3229–3231 (2002).
Fang, Q.-R. et al. Microporous metal–organic framework constructed from heptanuclear zinc carboxylate secondary building units. Chem. Eur. J. 12, 3754–3758 (2006).
Liu, Q. et al. Mesoporous cages in chemically robust MOFs created by a large number of vertices with reduced connectivity. J. Am. Chem. Soc. 141, 488–496 (2019).
Frisch, M. J. et al. Gaussian 09 (Gaussian Inc., 2009).
Wan, W. et al. Three-dimensional rotation electron diffraction: software RED for automated data collection and data processing. J. Appl. Cryst. 46, 1863–1873 (2013).
Coelho, A. A. TOPAS and TOPAS-Academic: an optimization program integrating computer algebra and crystallographic objects written in C plus. J. Appl. Cryst. 51, 210–218 (2018).
Kabsch, W. Integration, scaling, space-group assignment and post-refinement. Acta. Cryst. D66, 133–144 (2010).
Wang, B., Zou, X.-D. & Smeets, S. Automated serial rotation electron diffraction combined with cluster analysis: an efficient multi-crystal workflow for structure determination. IUCrJ 6, 854–867 (2019).
Sheldrick, G. M. SHELXT—integrated space-group and crystal-structure determination. Acta. Cryst. A71, 3–8 (2015).
Ma, T. Q. et al. Catenated covalent organic frameworks constructed from polyhedra. Nat. Synth. 2, 286–295 (2023).
Yi, L. Z. et al. Structure evolution of 2D covalent organic frameworks unveiled by single-crystal X-ray diffraction. J. Am. Chem. Soc. 146, 19643–19648 (2024).
Materials Studio v.7.0 (Accelrys Inc., 2016).
Hovmöller, S. CRISP: crystallographic image processing on a personal computer. Ultramicroscopy 41, 121–135 (1992).
Liu, Y.-Z. et al. Weaving of organic threads into a crystalline covalent organic framework. Science 351, 365–369 (2016).
Blatov, V. A., Carlucci, L., Ciani, G. & Proserpio, D. M. Interpenetrating metal–organic and inorganic 3D networks: a computer-aided systematic investigation. Part I. Analysis of the Cambridge structural database. Cryst. Eng. Comm. 6, 377–395 (2004).
Ying, Y. et al. Single-crystal three-dimensional covalent organic framework constructed from 6-connected triangular prism node. J. Am. Chem. Soc. 145, 22329–22334 (2023).
Blatov, V. A., Shevchenko, A. P. & Proserpio, D. M. Applied topological analysis of crystal structures with the program package ToposPro. Cryst. Growth Des. 14, 3576–3586 (2014).
Delgado-Friedrichs, O. & O’ Keefee, M. Identification of and symmetry computation for crystal nets. Acta Cryst. A 59, 351–360 (2003).
O’Keeffe, M. et al. The Reticular Chemistry Structure Resource (RCSR) database of, and symbols for, crystal nets. Acc. Chem. Res. 41, 1782–1789 (2008).
Shevchenko, A. P. et al. Topological representations of crystal structures: generation, analysis and implementation in the TopCryst system. Sci. Technol. Adv. Mater. 2, 250–265 (2022).
Osterrieth, J. M. W. et al. How reproducible are surface areas calculated from the BET equation? Adv. Mater. 34, 2201502 (2022).
Willems, T. F. et al. Algorithms and tools for high-throughput geometry-based analysis of crystalline porous materials. Microporous Mesoporous Mater. 149, 134–141 (2012).
Zhang, Y.-B. et al. A microporous metal–organic framework supramolecularly assembled from a CuII dodecaborate cluster complex for selective gas separation. Angew. Chem. Int. Ed. 58, 8145–8150 (2019).
Meng, S. et al. A facile approach to prepare porphyrinic porous aromatic frameworks for small hydrocarbon separation. J. Mater. Chem. A 2, 14536–14541 (2014).
Esteves, I. A. A. C. et al. Adsorption of natural gas and biogas components on activated carbon. Sep. Purif. Technol. 62, 281–296 (2008).
Wang, S. et al. Propane-selective design of zirconium-based MOFs for propylene purification. Chem. Eng. Sci. 219, 115604 (2020).
Kishida, K. et al. Recognition of 1,3-butadiene by a porous coordination polymer. Angew. Chem. Int. Ed. 55, 13784–13788 (2016).
Liao, P.-Q. et al. Controlling guest conformation for efficient purification of butadiene. Science 356, 1193–1196 (2017).
Whittaker, P. B. et al. Predicting the integral heat of adsorption for gas physisorption on microporous and mesoporous adsorbents. J. Phys. Chem. C 118, 8350–8358 (2014).
Saha, B. B. et al. Isotherms and thermodynamics for the adsorption of n-butane on pitch based activated carbon. Int. J. Heat Mass Transf. 51, 1582–1289 (2008).
Li, H. et al. Three-dimensional large-pore covalent organic framework with stp topology. J. Am. Chem. Soc. 142, 13334–13338 (2020).
Pan, H., Ritter, J. A. & Balbuena, P. B. Examination of the approximations used in determining the isosteric heat of adsorption from the Clausius–Clapeyron equation. Langmuir. 14, 6323–6327 (1998).
Myers, A. L. & Prausnitz, J. M. Thermodynamics of mixed-gas adsorption. AIChE J. 11, 121–127 (1965).
Kuhne, T. D. et al. CP2K: an electronic structure and molecular dynamics software package—Quickstep: efficient and accurate electronic structure calculations. J. Chem. Phys. 152, 194103 (2020).
Grimme, S., Bannwarth, C. & Schushkov, P. A robust and accurate tight-binding quantum chemical method for structures, vibrational frequencies, and noncovalent interactions of large molecular systems parametrized for all spd-block elements (Z = 1–86). J. Chem. Theory Comput. 13, 1989–2009 (2017).
Li, L.-B. et al. Ethane/ethylene separation in a metal–organic framework with iron-peroxo sites. Science. 362, 443–446 (2018).
Chen, K.-J. et al. Synergistic sorbent separation for one-step ethylene purification from a four-component mixture. Science 366, 241–246 (2019).
Li, B.-Y., Chrzanowski, M., Zhang, Y. M. & Ma, S. Q. Applications of metal–organic frameworks featuring multi-functional sites. Coord. Chem. Rev. 307, 106–129 (2016).
Zhao, X. et al. Metal–organic frameworks for separation. Adv. Mater. 30, 1705189 (2018).
Bloch, E.-D. et al. Hydrocarbon separations in a metal–organic framework with open iron (II) coordination sites. Science 335, 1606–1610 (2012).
Yang, S.-H. et al. Supramolecular binding and separation of hydrocarbons within a functionalized porous metal–organic framework. Nat. Chem. 7, 121–129 (2015).
Yuan, B.-Q., Wang, X., Zhou, X., Xiao, J. & Li, Z. Novel room-temperature synthesis of MIL-100(Fe) and its excellent adsorption performances for separation of light hydrocarbons. Chem. Eng. J. 355, 679–686 (2019).
Plonka, A. M. et al. Light hydrocarbon adsorption mechanisms in two calcium-based microporous metal organic frameworks. Chem. Mater. 28, 1636–1646 (2016).
Han, G.-P. et al. Enhancing higher hydrocarbons capture for natural gas upgrading by tuning van der Waals interactions in fcu-type Zr-MOFs. Ind. Eng. Chem. Res. 56, 14633–14641 (2017).
Acknowledgements
Q.F., J. Sun and H.L. acknowledge support from the National Key R&D Program of China (2022YFB3704900 and 2021YFF0500504), National Natural Science Foundation of China (22025504, 21621001, 22105082, U21A20285, 22125102, 22475081 and 21527803), Shanghai synchrotron radiation facility (bl14B), the SINOPEC Research Institute of Petroleum Processing, ‘111’ project (BP0719036 and B17020), China Postdoctoral Science Foundation (2020TQ0118 and 2020M681034), Ministry of Science and Technology of the People’s Republic of China (grant no. 2020YFA0210700) and the programme for JLU Science and Technology Innovative Research Team. V.V., Q.F. and S.Q. acknowledge the collaboration in the Sino-French International Research Network ‘Zeolites’.
Author information
Authors and Affiliations
Contributions
Q.F., H.L., J. Sun, V.V. and S.Q. were responsible for the overall project design, direction and supervision. J.C. and H.Z. conducted the synthesis and characterization of all samples. Z.Z., S.Z. and J. Sun collected and analysed the cRED data and topology structure. J.C., Z.Z., H.Z. and H.L. finalized the PXRD refinement and structure analyses. J.C., H.Z. and H.L. conducted the alkane sorption tests and breakthrough experiments. J. Suo and F.C. performed DFT calculations and assisted with the thermogravimetric analysis and FT-IR tests. C.J. assisted with the calculation of Qst and the ideal adsorbed solution theory for alkane sorption. Z.W. captured HRTEM images. All authors contributed to the discussion of results and paper writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Hexiang Deng and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Breakthrough experiments for JUC-644.
Column breakthrough curves for C2H6/C3H8 (a) and C2H6/n-C4H10 (b) mixture (50:50, v/v, Tfr: 2.0 mL min-1) at 298 K. (c) Column breakthrough curves for dry and moist C2H6/C3H8/n-C4H10 mixture (46:34:20, v/v/v, Tfr: 5.0 mL min-1) at 298 K. (d) Cycling breakthrough test of C2H6/C3H8/n-C4H10 mixture (46:34:20, v/v/v, Tfr: 5.0 mL/min) at 298 K.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–81 and Tables 1–21.
Supplementary Video 1 (download GIF )
Animation showing a simulation of the trajectory changes of C2H6 within JUC-644. For details see Supplementary Fig. 70.
Supplementary Video 2 (download GIF )
Animation showing a simulation of the trajectory changes of C3H8 within JUC-644. For details see Supplementary Fig. 70.
Supplementary Video 3 (download GIF )
Animation showing a simulation of the trajectory changes of C4H10 within JUC-644. For details see Supplementary Fig. 70.
Supplementary Data 1
Crystallographic data for JUC-644 (CCDC 2296805).
Supplementary Data 2
Crystallographic data for JUC-645 (CCDC 2353775).
Supplementary Data 3 (download XLSX )
The atomic coordinates of the optimized computational models, Supplementary Fig. 3.
Source data
Source Data Fig. 3 (download XLSX )
Unprocessed PXRD source data.
Source Data Fig. 6 (download ZIP )
Unprocessed source data for N2 adsorption–desorption isotherms at 77 K and C2H6, C3Hs and n-C4H10 adsorption isotherms at 298 K.
Source Data Extended Data Fig. 1 (download XLSX )
Unprocessed source data for column breakthrough curves of C2H6/C3Hs and C2H6/n-C4H10 mixtures at 298 K, column breakthrough curves for dry and humid C2H6/C3Hs/n-C4H10 mixtures, and cycle breakthrough tests for C2H6/C3Hs/n-C4H10 mixtures.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, J., Zhang, Z., Zheng, H. et al. Synthesis of three-dimensional covalent organic frameworks through a symmetry reduction strategy. Nat. Chem. 17, 571–581 (2025). https://doi.org/10.1038/s41557-024-01715-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-024-01715-6
This article is cited by
-
Single-crystal 2D covalent organic frameworks for high-capacity methane storage
Nature Communications (2026)
-
Local Microenvironment Regulation of Covalent Organic Frameworks for Enhanced Photocatalysis
Chemical Research in Chinese Universities (2025)