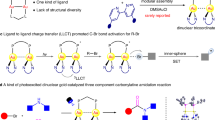
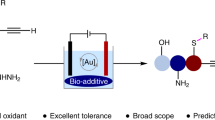
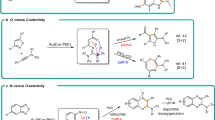
Abstract
Gold redox catalysis, which exploits the ability of strong π-acid activation in combination with redox reactions, has emerged as an attractive synthetic method with unique reactivities compared to other transition metals. However, gold redox chemistry bears the challenge to overcome the high redox potential of Au(I)/Au(III) (1.41 V). The classical strategy of gold redox catalysis applies strong external chemical oxidants which inevitably results in low atom economy and substrate limitations due to incompatibility with functional groups. Here we report a bidentate N-ligand (for example, Phen, Bpy) assisted gold redox catalysis using H2O2 as oxidant, which proved to be generally applicable for many forms of coupling reactions. In addition, C(sp2)–C(sp2) bicyclization coupling (cross-coupling of two cyclized substrates) is accessible under our conditions. Mechanistic studies reveal a redox elimination process in which a bidentate N-ligand is crucial for the catalytic cycle. The formation of alkynyl-AuIII–OH and vinyl-AuIII–OH species is the key process for the synergistic π-bond activation and AuI oxidation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this work are provided in the Supplementary Information, including experimental procedures and characterization of new compounds. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2372959 (3au), CCDC 2180908 (9al), CCDC 2218248 (11m), CCDC 2218295 (11u), CCDC 2372960 (12h) and CCDC 2372961 (12l). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Hashmi, A. S. K. & Hutchings, G. J. Gold catalysis. Angew. Chem. Int. Ed. 45, 7896–7936 (2006).
Gorin, D. J. & Toste, F. D. Relativistic effects in homogeneous gold catalysis. Nature 446, 395–403 (2007).
Krause, N. & Winter, C. Gold-catalyzed nucleophilic cyclization of functionalized allenes: a powerful access to carbo- and heterocycles. Chem. Rev. 111, 1994–2009 (2011).
Campeau, D., León Rayo, D. F., Mansour, A., Muratov, K. & Gagosz, F. Gold-catalyzed reactions of specially activated alkynes, allenes, and alkenes. Chem. Rev. 121, 8756–8867 (2020).
Zhou, T., Gao, P., Lalancette, R., Szostak, R. & Szostak, M. Gold-catalysed amine synthesis by reductive hydroamination of alkynes with nitroarenes. Nat. Chem. 16, 2025–2035 (2024).
Miro, J. & Del Pozo, C. Fluorine and gold: a fruitful partnership. Chem. Rev. 116, 11924–11966 (2016).
Bhoyare, V. W., Tathe, A. G., Das, A., Chintawar, C. C. & Patil, N. T. The interplay of carbophilic activation and Au(I)/Au(III) catalysis: an emerging technique for 1,2-difunctionalization of C–C multiple bonds. Chem. Soc. Rev. 50, 10422–10450 (2021).
Fricke, C., Reid, W. B. & Schoenebeck, F. A review on oxidative gold‐catalyzed C–H arylation of arenes—challenges and opportunities. Eur. J. Org. Chem. 2020, 7119–7130 (2020).
Font, P., Valdes, H. & Ribas, X. Consolidation of the oxidant-free Au(I)/Au(III) catalysis enabled by the hemilabile ligand strategy. Angew. Chem. Int. Ed. 63, e202405824 (2024).
Nijamudheen, A. & Datta, A. Gold‐catalyzed cross‐coupling reactions: an overview of design strategies, mechanistic studies, and applications. Chem. Eur. J. 26, 1442–1487 (2019).
Leyva-Pérez, A., Doménech, A., Al-Resayes, S. I. & Corma, A. Gold redox catalytic cycles for the oxidative coupling of alkynes. ACS Catal. 2, 121–126 (2011).
Cai, R. et al. Ligand‐assisted gold‐catalyzed cross‐coupling with aryldiazonium salts: redox gold catalysis without an external oxidant. Angew. Chem. Int. Ed. 54, 8772–8776 (2015).
Kumar, A., Shukla, K., Ahsan, S., Paul, A. & Patil, N. T. Electrochemical gold-catalyzed 1,2-difunctionalization of C–C multiple bonds. Angew. Chem. Int. Ed. 62, e202308636 (2023).
Li, X., Xie, X., Sun, N. & Liu, Y. Gold‐catalyzed Cadiot–Chodkiewicz‐type cross‐coupling of terminal alkynes with alkynyl hypervalent iodine reagents: highly selective synthesis of unsymmetrical 1,3‐diynes. Angew. Chem. Int. Ed. 56, 6994–6998 (2017).
Li, Y., Brand, J. P. & Waser, J. Gold‐catalyzed regioselective synthesis of 2‐ and 3‐alkynyl furans. Angew. Chem. Int. Ed. 52, 6743–6747 (2013).
Yang, Y. et al. Dual gold/silver catalysis involving alkynylgold(III) intermediates formed by oxidative addition and silver‐catalyzed C–H activation for the direct alkynylation of cyclopropenes. Angew. Chem. Int. Ed. 58, 5129–5133 (2019).
Ye, X. et al. Facilitating gold redox catalysis with electrochemistry: an efficient chemical‐oxidant‐free approach. Angew. Chem. Int. Ed. 58, 17226–17230 (2019).
Liu, K., Li, N., Ning, Y., Zhu, C. & Xie, J. Gold-catalyzed oxidative biaryl cross-coupling of organometallics. Chem 5, 2718–2730 (2019).
Ye, X. et al. Gold-catalyzed oxidative coupling of alkynes toward the synthesis of cyclic conjugated diynes. Chem 4, 1983–1993 (2018).
Ball, L. T., Lloyd-Jones, G. C. & Russell, C. A. Gold-catalyzed oxidative coupling of arylsilanes and arenes: origin of selectivity and improved precatalyst. J. Am. Chem. Soc. 136, 254–264 (2013).
de Haro, T. & Nevado, C. Gold-catalyzed ethynylation of arenes. J. Am. Chem. Soc. 132, 1512–1513 (2010).
Peng, H. et al. Gold-catalyzed oxidative cross-coupling of terminal alkynes: selective synthesis of unsymmetrical 1,3-diynes. J. Am. Chem. Soc. 136, 13174–13177 (2014).
Sahoo, B., Hopkinson, M. N. & Glorius, F. Combining gold and photoredox catalysis: visible light-mediated oxy- and aminoarylation of alkenes. J. Am. Chem. Soc. 135, 5505–5508 (2013).
Shu, X.-z, Zhang, M., He, Y., Frei, H. & Toste, F. D. Dual visible light photoredox and gold-catalyzed arylative ring expansion. J. Am. Chem. Soc. 136, 5844–5847 (2014).
Huang, B., Hu, M. & Toste, F. D. Homogeneous gold redox chemistry: organometallics, catalysis, and beyond. Trends Chem. 2, 707–720 (2020).
Wu, C.-Y., Horibe, T., Jacobsen, C. B. & Toste, F. D. Stable gold(III) catalysts by oxidative addition of a carbon–carbon bond. Nature 517, 449–454 (2015).
Wolf, W. J., Winston, M. S. & Toste, F. D. Exceptionally fast carbon–carbon bond reductive elimination from gold(III). Nat. Chem. 6, 159–164 (2013).
Rocchigiani, L. & Bochmann, M. Recent advances in gold(III) chemistry: structure, bonding, reactivity, and role in homogeneous catalysis. Chem. Rev. 121, 8364–8451 (2021).
Hashmi, A. S. K. Gold-catalyzed organic reactions. Chem. Rev. 107, 3180–3211 (2007).
Joost, M., Estévez, L., Miqueu, K., Amgoune, A. & Bourissou, D. Oxidative addition of carbon–carbon bonds to gold. Angew. Chem. Int. Ed. 54, 5236–5240 (2015).
Joost, M. et al. Facile oxidative addition of aryl iodides to gold(I) by ligand design: bending turns on reactivity. J. Am. Chem. Soc. 136, 14654–14657 (2014).
Harper, M. J. et al. Oxidative addition, transmetalation, and reductive elimination at a 2,2′-bipyridyl-ligated gold center. J. Am. Chem. Soc. 140, 4440–4445 (2018).
Zeineddine, A. et al. Rational development of catalytic Au(I)/Au(III) arylation involving mild oxidative addition of aryl halides. Nat. Commun. 8, 565 (2017).
Xia, Z. et al. Photosensitized oxidative addition to gold(I) enables alkynylative cyclization of o-alkylnylphenols with iodoalkynes. Nat. Chem. 11, 797–805 (2019).
Zhao, F. et al. Reactant-induced photoactivation of in situ generated organogold intermediates leading to alkynylated indoles via Csp2–Csp cross-coupling. Nat. Commun. 13, 2295 (2022).
Ball, L. T., Lloyd-Jones, G. C. & Russell, C. A. Gold-catalyzed direct arylation. Science 337, 1644–1648 (2012).
Cambeiro, X. C., Ahlsten, N. & Larrosa, I. Au-catalyzed cross-coupling of arenes via double C–H activation. J. Am. Chem. Soc. 137, 15636–15639 (2015).
Brand, J. P. & Waser, J. Direct alkynylation of thiophenes: cooperative activation of TIPS–EBX with gold and Brønsted acids. Angew. Chem. Int. Ed. 49, 7304–7307 (2010).
Hu, L. et al. Au–Ag bimetallic catalysis: 3‐alkynyl benzofurans from phenols via tandem C–H alkynylation/oxy‐alkynylation. Angew. Chem. Int. Ed. 60, 10637–10642 (2021).
Banerjee, S., Bhoyare, V. W. & Patil, N. T. Gold and hypervalent iodine(III): liaisons over a decade for electrophilic functional group transfer reactions. Chem. Commun. 56, 2677–2690 (2020).
Han, C., Tian, X., Zhang, H., Rominger, F. & Hashmi, A. S. K. Tetrasubstituted 1,3-enynes by gold-catalyzed direct C(sp2)–H alkynylation of acceptor-substituted enamines. Org. Lett. 23, 4764–4768 (2021).
Liu, Y. et al. Synthesis of amide enol 2-iodobenzoates by the regio- and stereoselective gold-catalyzed acyloxyalkynylation of ynamides with hypervalent iodine reagents. Org. Lett. 24, 7101–7106 (2022).
Li, X., Wodrich, M. D. & Waser, J. Accessing elusive σ-type cyclopropenium cation equivalents through redox gold catalysis. Nat. Chem. 16, 901–912 (2024).
Zhang, G., Peng, Y., Cui, L. & Zhang, L. Gold‐catalyzed homogeneous oxidative cross‐coupling reactions. Angew. Chem. Int. Ed. 48, 3112–3115 (2009).
Brenzovich, W. E. et al. Gold‐catalyzed intramolecular aminoarylation of alkenes: C–C bond formation through bimolecular reductive elimination. Angew. Chem. Int. Ed. 49, 5519–5522 (2010).
Hopkinson, M. N., Tlahuext-Aca, A. & Glorius, F. Merging visible light photoredox and gold catalysis. Acc. Chem. Res. 49, 2261–2272 (2016).
Witzel, S., Hashmi, A. S. K. & Xie, J. Light in gold catalysis. Chem. Rev. 121, 8868–8925 (2021).
Liang, H., Julaiti, Y., Zhao, C.-G. & Xie, J. Electrochemical gold-catalysed biocompatible C(sp2)–C(sp) coupling. Nat. Synth. 2, 338–347 (2023).
Zhang, S., Wei, J., Ye, X., Perez, A. & Shi, X. Accessing gold p-acid reactivity under electrochemical anode oxidation (EAO) through oxidation relay. Nat. Commun. 14, 8265 (2023).
Kumar, A., Bhattacharya, N. & Patil, N. T. Electrochemical gold redox catalysis. ChemCatChem 16, e202401112 (2024).
Li, W. et al. Cooperative Au/Ag dual-catalyzed cross-dehydrogenative biaryl coupling: reaction development and mechanistic insight. J. Am. Chem. Soc. 141, 3187–3197 (2019).
Boogaerts, I. I. F. & Nolan, S. P. Carboxylation of C–H bonds using N-heterocyclic carbene gold(I) complexes. J. Am. Chem. Soc. 132, 8858–8859 (2010).
Rosca, D. A., Wright, J. A. & Bochmann, M. An element through the looking glass: exploring the Au–C, Au–H and Au–O energy landscape. Dalton Trans. 44, 20785–20807 (2015).
Roşca, D.-A., Wright, J. A., Hughes, D. L. & Bochmann, M. Gold peroxide complexes and the conversion of hydroperoxides into gold hydrides by successive oxygen-transfer reactions. Nat. Commun. 4, 2167 (2013).
Bockris, J. O’M. & Oldfield, L. F. The oxidation–reduction reactions of hydrogen peroxide at inert metal electrodes and mercury cathodes. Trans. Faraday Soc. 51, 249–259 (1955).
Wegner, H. A., Ahles, S. & Neuburger, M. A new gold‐catalyzed domino cyclization and oxidative coupling reaction. Chem. Eur. J. 14, 11310–11313 (2008).
Hashmi, A. S. et al. Gold catalysis: in situ EXAFS study of homogeneous oxidative esterification. Chem. Eur. J. 16, 8012–8019 (2010).
Zhang, Y., Peng, H., Zhang, M., Cheng, Y. & Zhu, C. Gold-complexes catalyzed oxidative α-cyanation of tertiary amines. Chem. Commun. 47, 2354–2356 (2011).
Lu, Z., Li, T., Mudshinge, S. R., Xu, B. & Hammond, G. B. Optimization of catalysts and conditions in gold(I) catalysis—counterion and additive effects. Chem. Rev. 121, 8452–8477 (2021).
Yang, Y. et al. Trans influence of ligands on the oxidation of gold(I) complexes. J. Am. Chem. Soc. 141, 17414–17420 (2019).
Shi, W. & Lei, A. 1,3-Diyne chemistry: synthesis and derivations. Tetrahedron Lett. 55, 2763–2772 (2014).
Shi Shun, A. L. K. & Tykwinski, R. R. Synthesis of naturally occurring polyynes. Angew. Chem. Int. Ed. 45, 1034–1057 (2006).
Siemsen, P., Livingston, R. C. & Diederich, F. Acetylenic coupling: a powerful tool in molecular construction. Angew. Chem. Int. Ed. 39, 2632–2657 (2000).
Rudolph, M. & Hashmi, A. S. Gold catalysis in total synthesis—an update. Chem. Soc. Rev. 41, 2448–2462 (2012).
Zheng, Z. et al. Homogeneous gold-catalyzed oxidation reactions. Chem. Rev. 121, 8979–9038 (2021).
Hopkinson, M. N. et al. Gold‐catalyzed intramolecular oxidative cross‐coupling of nonactivated arenes. Chem. Eur. J. 16, 4739–4743 (2010).
Taylor, R. D., MacCoss, M. & Lawson, A. D. G. Rings in drugs. J. Med. Chem. 57, 5845–5859 (2014).
Nevagi, R. J., Dighe, S. N. & Dighe, S. N. Biological and medicinal significance of benzofuran. Eur. J. Med. Chem. 97, 561–581 (2015).
Leyva-Pérez, A., Doménech-Carbó, A. & Corma, A. Unique distal size selectivity with a digold catalyst during alkyne homocoupling. Nat. Commun. 6, 6703 (2015).
Acknowledgements
H.S., Y.T., Y.L. and T.W. are grateful for PhD fellowships from the China Scholarship Council (CSC). H.S. gratefully acknowledges the CPSF (number GZC20230165) and the NSFC (number 22401010). J.L. gratefully acknowledges the Alexander von Humboldt Foundation for a postdoctoral fellowship. H.H., P.M.S. and A.S.K.H. gratefully acknowledge the Hector Fellow Academy for the generous provision of funding. H.S. and N.J. acknowledge the NSFC (numbers 22293014, 22131002, 22161142019), the National Key R&D Program of China (number 2021YFA1501700), Changping Laboratory, the New Cornerstone Science Foundation through the New Cornerstone Investigator Program and the XPLORER PRIZE for financial support. We thank X. Zhang for his help with the ESI-MS mechanism experiments.
Author information
Authors and Affiliations
Contributions
H.S. conceived and designed the experiments. A.S.K.H. and N.J. directed the research. H.S. carried out most of the experiments. H.S., M.R., J.L., Y.T., M.C.D., H.A., Y.L., T.W., H.H., P.M.S., N.J. and A.S.K.H. participated in discussion. P.K. analysed IR data. F.R. and T.O. conducted X-ray crystal structure analysis. H.S., M.R., N.J. and A.S.K.H. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Nitin Patil, Xiaodong Shi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary data, figures, tables and discussion.
Supplementary Data 1
Crystallographic data for compound 3au; CCDC reference 2372959
Supplementary Data 2
Crystallographic data for compound 9al; CCDC reference 2180908
Supplementary Data 3
Crystallographic data for compound 11m; CCDC reference 2218248
Supplementary Data 4
Crystallographic data for compound 11u; CCDC reference 2218295
Supplementary Data 5
Crystallographic data for compound 12h; CCDC reference 2372960
Supplementary Data 6
Crystallographic data for compound 12l; CCDC reference 2372961
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, H., Rudolph, M., Li, J. et al. Bidentate N-ligand-assisted gold redox catalysis with hydrogen peroxide. Nat. Chem. 17, 822–834 (2025). https://doi.org/10.1038/s41557-025-01835-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01835-7
This article is cited by
-
Precise post-modification of polystyrene with gold-catalyzed C(sp2)–H arylation
Science China Chemistry (2026)
-
Bidentate mixed-ligand strategy in dinuclear gold photoredox catalysis
Nature Communications (2025)
-
Gold redox catalysis with hydrogen peroxide
Nature Chemistry (2025)