Abstract
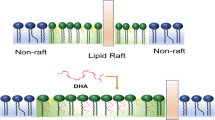
Polyunsaturated fatty acids (PUFAs) represent a fundamental and essential class of lipids that exhibit versatile biofunctions. Lipidomic analysis has identified a growing number of lipid species, including PUFAs with diverse structural variations and biofunctions, yet their structure–function relationships are still largely unknown. In this context, an efficient synthesis of PUFAs would be highly desirable. However, no practical methodology exists for their preparation, in contrast to peptides and nucleic acids, for which diverse molecules are accessible through a well-established solid-phase synthesis. To address this, we have developed an efficient and expedited method to access a wide array of PUFAs by full solid-phase synthesis. The method allows the synthesis of various PUFAs and analogues through rapid and facile operations. Moreover, within our PUFA library, we have discovered an artificial fatty acid, antiefin, that has a high anti-inflammatory effect in vivo. Therefore, our practical synthetic pathway to PUFAs, a crucial class of lipids, is expected to make an important contribution to lipid science.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the Article and its Supplementary Information files. Source data for the assays and measurements are available via the UTokyo Repository at https://repository.dl.itc.u-tokyo.ac.jp/records/2010704 (ref. 44).
References
Dyall, S. C. et al. Polyunsaturated fatty acids and fatty acid-derived lipid mediators: recent advances in the understanding of their biosynthesis, structures, and functions. Prog. Lipid Res. 86, 101165 (2022).
Ulven, T. & Christiansen, E. Dietary fatty acids and their potential for controlling metabolic diseases through activation of FFA4/GPR120. Annu. Rev. Nutr. 35, 239–263 (2015).
Gorjão, R. et al. Comparative effects of DHA and EPA on cell function. Pharmacol. Ther. 122, 56–64 (2009).
Isobe, Y. & Arita, M. Identification of novel omega-3 fatty acid-derived bioactive metabolites based on a targeted lipidomics approach. J. Clin. Biochem. Nutr. 55, 79–84 (2014).
Leuti, A. et al. Bioactive lipids, inflammation and chronic diseases. Adv. Drug Deliv. Rev. 159, 133–169 (2020).
Duvall, M. G. & Levy, B. D. DHA- and EPA-derived resolvins, protectins, and maresins in airway inflammation. Eur. J. Pharmacol. 785, 144–155 (2016).
Calder, P. C. Polyunsaturated fatty acids and inflammation. Prostaglandins Leukot. Essent. Fatty Acids 75, 197–202 (2006).
Ichi, I. et al. Identification of genes and pathways involved in the synthesis of mead acid (20:3n–9), an indicator of essential fatty acid deficiency. Biochim. Biophys. Acta 1841, 204–213 (2014).
Harayama, T. & Riezman, H. Understanding the diversity of membrane lipid composition. Nat. Rev. Mol. Cell Biol. 19, 281–296 (2018).
Kunisawa, J. et al. Dietary ω3 fatty acid exerts anti-allergic effect through the conversion to 17,18-epoxyeicosatetraenoic acid in the gut. Sci. Rep. 5, 9750 (2015).
Nagatake, T. et al. The 17,18-epoxyeicosatetraenoic acid–G protein–coupled receptor 40 axis ameliorates contact hypersensitivity by inhibiting neutrophil mobility in mice and cynomolgus macaques. J. Allergy Clin. Immunol. 142, 470–484.e12 (2018).
Jeffery, T., Gueugnot, S. & Linstrumelle, G. An efficient route to skipped diynes and triynes, (Z,Z) dienes and (Z,Z,Z) trienes. Tetrahedron Lett. 33, 5757–5760 (1992).
Durand, S., Parrain, J. L. & Santelli, M. A large scale and concise synthesis of γ-linolenic acid from 4-chlorobut-2-yn-1-ol. Synthesis 1998, 1015–1018 (1998).
Proteau-Gagne, A. et al. Synthesis and functional pharmacological effects on human bronchi of 20-hydroxyeicosatetraenoic acid. Chem. Nat. Compd 46, 841–847 (2011).
Bräse, S. Combinatorial Chemistry on Solid Supports (Springer, 2007).
Merrifield, R. B. Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 85, 2149–2154 (1963).
Qi, L., Meijler, M. M., Lee, S. H., Sun, C. & Janda, K. D. Solid-phase synthesis of anandamide analogues. Org. Lett. 6, 1673–1675 (2004).
Mant, C. T. & Hodges, R. S. High-Performance Liquid Chromatography of Peptides and Proteins (CRC Press, 1991).
Evano, G. et al. Turning unreactive copper acetylides into remarkably powerful and mild alkyne transfer reagents by oxidative umpolung. Chem. Commun. 50, 10008–10018 (2014).
Asano, Y., Ito, H., Hara, K. & Sawamura, M. Enantioselective addition of terminal alkynes to aromatic aldehydes catalyzed by copper(I) complexes with wide-bite-angle chiral bisphosphine ligands: optimization, scope, and mechanistic studies. Organometallics 27, 5984–5996 (2008).
Pirrung, M. C., Shuey, S. W., Lever, D. C. & Fallon, L. A convenient procedure for the deprotection of silylated nucleosides and nucleotides using triethylamine trihydrofluoride. Bioorg. Med. Chem. Lett. 4, 1345–1346 (1994).
Balas, L., Durand, T., Saha, S., Johnson, I. & Mukhopadhyay, S. Total synthesis of photoactivatable or fluorescent anandamide probes: novel bioactive compounds with angiogenic activity. J. Med. Chem. 52, 1005–1017 (2009).
Whittaker, A. M. & Lalic, G. Monophasic catalytic system for the selective semireduction of alkynes. Org. Lett. 15, 1112–1115 (2013).
Isobe, Y. et al. Comprehensive analysis of the mouse cytochrome P450 family responsible for omega-3 epoxidation of eicosapentaenoic acid. Sci. Rep. 8, 7954 (2018).
Miyata, N. & Roman, R. J. Role of 20-hydroxyeicosatetraenoic acid (20-HETE) in vascular system. J. Smooth Muscle Res. 41, 175–193 (2005).
Tunaru, S. et al. 20-HETE promotes glucose-stimulated insulin secretion in an autocrine manner through FFAR1. Nat. Commun. 9, 177 (2018).
Hwang, S. H. et al. Chemical synthesis and biological evaluation of ω-hydroxy polyunsaturated fatty acids. Bioorg. Med. Chem. Lett. 27, 620–625 (2017).
Xu, L. et al. Furan fatty acids—beneficial or harmful to health? Prog. Lipid Res. 68, 119–137 (2017).
Christiansen, E. et al. Activity of dietary fatty acids on FFA1 and FFA4 and characterisation of pinolenic acid as a dual FFA1/FFA4 agonist with potential effect against metabolic diseases. Br. J. Nutr. 113, 1677–1688 (2015).
Khan, M. Z. & He, L. The role of polyunsaturated fatty acids and GPR40 receptor in brain. Neuropharmacology 113, 639–651 (2017).
Kimura, I., Ichimura, A., Ohue-Kitano, R. & Igarashi, M. Free fatty acid receptors in health and disease. Physiol. Rev. 100, 171–210 (2020).
Defossa, E. & Wagner, M. Recent developments in the discovery of FFA1 receptor agonists as novel oral treatment for type 2 diabetes mellitus. Bioorg. Med. Chem. Lett. 24, 2991–3000 (2014).
Inoue, A. et al. TGFα shedding assay: an accurate and versatile method for detecting GPCR activation. Nat. Methods 9, 1021–1029 (2012).
Houthuijzen, J. M. et al. Fatty acid 16:4(n–3) stimulates a GPR120-induced signaling cascade in splenic macrophages to promote chemotherapy resistance. FASEB J. 31, 2195–2209 (2017).
Morisseau, C. & Hammock, B. D. Impact of soluble epoxide hydrolase and epoxyeicosanoids on human health. Annu. Rev. Pharmacol. Toxicol. 53, 37–58 (2013).
Harris, T. R. & Hammock, B. D. Soluble epoxide hydrolase: gene structure, expression and deletion. Gene 526, 61–74 (2013).
Morisseau, C. et al. Relative importance of soluble and microsomal epoxide hydrolases for the hydrolysis of epoxy-fatty acids in human tissues. Int. J. Mol. Sci. 22, 4993 (2021).
Rose, T. E. et al. 1-Aryl-3-(1-acylpiperidin-4-yl)urea inhibitors of human and murine soluble epoxide hydrolase: structure–activity relationships, pharmacokinetics, and reduction of inflammatory pain. J. Med. Chem. 53, 7067–7075 (2010).
Ostermann, A. I. Oral treatment of rodents with soluble epoxide hydrolase inhibitor 1-(1-propanoylpiperidin-4-yl)-3-[4-(trifluoromethoxy)phenyl]urea (TPPU): resulting drug levels and modulation of oxylipin pattern. Prostaglandins Other Lipid Mediat. 121, 131–137 (2015).
Saika, A. et al. 17(S),18(R)-epoxyeicosatetraenoic acid generated by cytochrome P450 BM-3 from Bacillus megaterium inhibits the development of contact hypersensitivity via G-protein-coupled receptor 40-mediated neutrophil suppression. FASEB Bioadv. 2, 59–71 (2020).
Spector, A. A. & Kim, H. Y. Cytochrome P450 epoxygenase pathway of polyunsaturated fatty acid metabolism. Biochim. Biophys. Acta 1851, 356–365 (2015).
Hay, A. S. Oxidative coupling of acetylenes. II. J. Org. Chem. 27, 3320–3321 (1962).
Thorpe, T. C. et al. BacT/Alert: an automated colorimetric microbial detection system. J. Clin. Microbiol. 28, 1608–1612 (1990).
Saito, Y. et al. Data of assays and measurements on solid-phase synthesis of PUFAs and development of Antiefin. UTokyo Repository https://repository.dl.itc.u-tokyo.ac.jp/records/2010704 (2025).
Acknowledgements
This work was performed in part at the One-stop Sharing Facility Center for Future Drug Discoveries in the Graduate School of Pharmaceutical Sciences, The University of Tokyo. We thank K. Nozaki, T. Iwasaki and X. Jin (The University of Tokyo) for fruitful discussions and support with GC measurements. The atomic absorption measurements were performed at the Common Facilities of the Analytical Chemistry Laboratory, Department of Applied Chemistry, School of Engineering, The University of Tokyo. We thank T. Yamagishi (The University of Tokyo) for fruitful discussions and support with the atomic absorption measurements. This research was supported by JSPS KAKENHI (grant nos. JP22K14780 (to Y. Saito) and JP22KJ1101 (to M.A.)), AMED (grant no. JP233fa727001 (to Y. Saito and J.K.)), the Toyota Riken Scholar Program (to Y. Saito), the Mizuho Foundation for the Promotion of Sciences (to Y. Saito), KONICA MINOLTA Award in Synthetic Organic Chemistry, Japan (to Y. Saito) and JST-CREST (grant no. JPMJCR21N5 (to S.S.)).
Author information
Authors and Affiliations
Contributions
Y. Saito, J.K. and S.S. conceived and designed the project. Y. Saito, M.A. and Y. Sano performed the synthetic experiments with the help of J.M. and S.S. Y. Saito, M.A., A.S., M.H. and T.N. performed the cellular experiments with the help of A.U., J.A., J.K. and S.S. A.S., M.H. and T.N. performed the animal experiments with the help of J.K. Finally, Y. Saito, M.A. and S.S wrote the paper, which was edited by all of the co-authors.
Corresponding authors
Ethics declarations
Competing interests
Y. Saito, M.A., Y. Sano and S.S. have filed a patent application related to the preparation method of polyunsaturated fatty acids on a solid phase (patent applicant: The University of Tokyo; inventors: Y. Saito, S.S., M.A., Y. Sano and Yaohong Shi; international Patent application number: PCT/JP2025/011856; current application status: pending). All other authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Anna Sansone and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Materials and Methods, Figs. 1–4, Tables 1–6, NMR spectra and HPLC, LC–MS and GC chromatograms.
Supplementary Data 1 (download PDF )
Certificate of sterility tests
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saito, Y., Akita, M., Saika, A. et al. Expedited access to polyunsaturated fatty acids and biofunctional analogues by full solid-phase synthesis. Nat. Chem. 17, 1391–1400 (2025). https://doi.org/10.1038/s41557-025-01853-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01853-5
This article is cited by
-
Stereoselective self-assembly of cis- and trans- [Pt2L2] metallacycles via geometrically-asymmetric 90o Pt(II) acceptors
Communications Chemistry (2025)