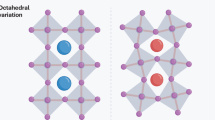
Abstract
Zero-dimensional (0D) metal halides, which feature discrete metal halide octahedra interspersed with large organoammonium cations, are the building blocks of halide perovskites. The optical properties of these materials make them promising candidates in light-emitting devices. However, developing their general design principles remains challenging. Here we report an antisolvent incorporation approach that transforms a broad range of two-dimensional tin iodide perovskites into 0D structures. This approach accommodates diverse organic cations and antisolvent molecules and is extendable to germanium and lead analogues. We show that enhancing the structural rigidity in Sn-based octahedra—modulated by organic packing—increases the radiative recombination rate while decreasing the non-radiative recombination rate, achieving near-unity photoluminescence quantum yield. Ge- and Sn-based structures exhibit large Stokes shifts and microsecond-scale triplet lifetimes due to localized excited states, while their Pb-based counterpart shows faster recombination via triplet–singlet mixing, supported by theoretical calculations. This work offers a versatile synthetic platform for 0D metal halides and deepens our current understanding of excited-state dynamics in halide perovskites.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data that support the finding of this study can be found in the article or its Supplementary Information. Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2370156 (A1), 2370157 (A2), 2370158 (A3), 2370159 (A4), 2370160 (A5), 2370161 (A6), 2370162 (A7), 2370163 (A8), 2370164 (A9), 2370165 (A10), 2370166 (A13), 2370167 (B2), 2370168 (B5), 2370169 (B6), 2370170 (C1), 2370171 [(PEA)6GeI8(Bz)2], 2370172 [(PEA)6PbI8(Bz)2], 2370173 [(PEA)6PbI8(CBz)2], 2370174 [(PEA)6PbI8(DCB)2], 2420403 (A12), 2420404 (A11), 2420405 (B1), 2420406 (B3) and 2420407 (B4). Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
References
Fu, Y. et al. Metal halide perovskite nanostructures for optoelectronic applications and the study of physical properties. Nat. Rev. Mater. 4, 169–188 (2019).
Akkerman, Q. A. & Manna, L. What defines a halide perovskite? ACS Energy Lett. 5, 604–610 (2020).
Lin, H., Zhou, C., Tian, Y., Siegrist, T. & Ma, B. Low-dimensional organometal halide perovskites. ACS Energy Lett. 3, 54–62 (2018).
Yakunin, S. et al. High-resolution remote thermometry and thermography using luminescent low-dimensional tin-halide perovskites. Nat. Mater. 18, 846–852 (2019).
Ju, Y. et al. Water-sensitive mixed-phase PEA6SnI8 perovskite derivative single crystal for humidity detection. Cryst. Growth Des. 22, 4689–4695 (2022).
Zhou, C. et al. Highly efficient broadband yellow phosphor based on zero-dimensional tin mixed-halide perovskite. ACS Appl. Mater. Interfaces 9, 44579–44583 (2017).
Wang, Y. et al. Room temperature ferroelectricity and blue photoluminescence in zero dimensional organic lead iodine perovskites. J. Mater. Chem. C 9, 223–227 (2021).
Li, Z. et al. Precursor tailoring enables alkylammonium tin halide perovskite phosphors for solid‐state lighting. Adv. Funct. Mater. 32, 2111346 (2022).
Xu, L.-J. et al. 0D and 2D: the cases of phenylethylammonium tin bromide hybrids. Chem. Mater. 32, 4692–4698 (2020).
Cui, B.-B. et al. Locally collective hydrogen bonding isolates lead octahedra for white emission improvement. Nat. Commun. 10, 5190 (2019).
Jia, X. et al. Methylpiperazine based 0D chiral hybrid lead halides for second harmonic generation. Dalton Trans. 51, 7248–7254 (2022).
Zhang, L. et al. Organic cation-directed modulation of emissions in zero-dimensional hybrid tin bromides. Inorg. Chem. 61, 14857–14863 (2022).
Fu, P. et al. Organic-inorganic layered and hollow tin bromide perovskite with tunable broadband emission. ACS Appl. Mater. Interfaces 10, 34363–34369 (2018).
Weber, O. J., Marshall, K. L., Dyson, L. M. & Weller, M. T. Structural diversity in hybrid organic-inorganic lead iodide materials. Acta Cryst. 71, 668–678 (2015).
Sun, C. et al. A zero-dimensional hybrid lead perovskite with highly efficient blue-violet light emission. J. Mater. Chem. C 8, 11890–11895 (2020).
Benin, B. M. et al. Highly emissive self-trapped excitons in fully inorganic zero-dimensional tin halides. Angew. Chem. Int. Ed. 57, 11329–11333 (2018).
Zheng, D.-S. et al. Highly emissive 0D Tin(II) hybrid for white LED. J. Lumin. 270, 120530 (2024).
Nikl, M. et al. Photoluminescence of Cs4PbBr6 crystals and thin films. Chem. Phys. Lett. 306, 280–284 (1999).
Huang, X. et al. Understanding electron–phonon interactions in 3D lead halide perovskites from the stereochemical expression of 6s2 lone pairs. J. Am. Chem. Soc. 144, 12247–12260 (2022).
Miyata, K., Atallah, T. L. & Zhu, X.-Y. Lead halide perovskites: crystal–liquid duality, phonon glass electron crystals, and large polaron formation. Sci. Adv. 3, e1701469 (2017).
Ghosh, D., Welch, E., Neukirch, A. J., Zakhidov, A. & Tretiak, S. Polarons in halide perovskites: a perspective. J. Phys. Chem. Lett. 11, 3271–3286 (2020).
Gong, X. et al. Electron–phonon interaction in efficient perovskite blue emitters. Nat. Mater. 17, 550–556 (2018).
Becker, M. A. et al. Bright triplet excitons in caesium lead halide perovskites. Nature 553, 189–193 (2018).
Even, J., Pedesseau, L., Jancu, J.-M. & Katan, C. Importance of spin–orbit coupling in hybrid organic/inorganic perovskites for photovoltaic applications. J. Phys. Chem. Lett. 4, 2999–3005 (2013).
Gu, J. & Fu, Y. Is there an optimal spacer cation for two-dimensional lead iodide perovskites? ACS Mater. Au 5, 24–34 (2025).
Mao, L., Stoumpos, C. C. & Kanatzidis, M. G. Two-dimensional hybrid halide perovskites: principles and promises. J. Am. Chem. Soc. 141, 1171–1190 (2019).
Park, J. Y. et al. Thickness control of organic semiconductor-incorporated perovskites. Nat. Chem. 15, 1745–1753 (2023).
Yaffe, O. et al. Local polar fluctuations in lead halide perovskite crystals. Phys. Rev. Lett. 118, 136001 (2017).
Wang, X. & Yan, Y. in Hybrid Organic Inorganic Perovskites (ed. Vardeny, Z. V.) Ch. 2, 37–63 (World Scientific Publishing, 2022).
Ranfagni, A., Mugnai, D., Bacci, M., Viliani, G. & Fontana, M. P. The optical properties of thallium-like impurities in alkali-halide crystals. Adv. Phys. 32, 823–905 (1983).
Li, X. et al. Optical bandgap anomaly with tuning dimensionality in germanium perovskites: interplay between quantum confinement and lone pair expression. Chem 10, 891–909 (2024).
Li, X., Guan, Y., Li, X. & Fu, Y. Stereochemically active lone pairs and nonlinear optical properties of two-dimensional multilayered tin and germanium iodide perovskites. J. Am. Chem. Soc. 144, 18030–18042 (2022).
Lü, X. et al. Regulating off-centering distortion maximizes photoluminescence in halide perovskites. Natl Sci. Rev. 8, nwaa288 (2020).
Stoumpos, C. C. et al. Hybrid germanium iodide perovskite semiconductors: active lone pairs, structural distortions, direct and indirect energy gaps, and strong nonlinear optical properties. J. Am. Chem. Soc. 137, 6804–6819 (2015).
Wu, X. et al. Light-induced picosecond rotational disordering of the inorganic sublattice in hybrid perovskites. Sci. Adv. 3, e1602388 (2017).
Srimath Kandada, A. R. & Silva, C. Exciton polarons in two-dimensional hybrid metal-halide perovskites. J. Phys. Chem. Lett. 11, 3173–3184 (2020).
Hansen, K. R. et al. Low exciton binding energies and localized exciton–polaron states in 2D tin halide perovskites. Adv. Opt. Mater. 10, 2102698 (2022).
Mahata, A., Meggiolaro, D. & De Angelis, F. From large to small polarons in lead, tin, and mixed lead–tin halide perovskites. J. Phys. Chem. Lett. 10, 1790–1798 (2019).
Ouhbi, H., Ambrosio, F., De Angelis, F. & Wiktor, J. Strong electron localization in tin halide perovskites. J. Phys. Chem. Lett. 12, 5339–5343 (2021).
Shao, W. et al. Molecular templating of layered halide perovskite nanowires. Science 384, 1000–1006 (2024).
Li, Y. et al. Phase-pure 2D tin halide perovskite thin flakes for stable lasing. Sci. Adv. 9, eadh0517 (2023).
Han, D. et al. Tautomeric mixture coordination enables efficient lead-free perovskite LEDs. Nature 622, 493–498 (2023).
Baryshnikov, G., Minaev, B. & Ågren, H. Theory and calculation of the phosphorescence phenomenon. Chem. Rev. 117, 6500–6537 (2017).
Wright, A. D. et al. Electron–phonon coupling in hybrid lead halide perovskites. Nat. Commun. 7, 11755 (2016).
Parrott, E. S. et al. Effect of structural phase transition on charge-carrier lifetimes and defects in CH3NH3SnI3 perovskite. J. Phys. Chem. Lett. 7, 1321–1326 (2016).
Sousa, C., Tosoni, S. & Illas, F. Theoretical approaches to excited-state-related phenomena in oxide surfaces. Chem. Rev. 113, 4456–4495 (2013).
Kolafa, J. & Perram, J. W. Cutoff errors in the Ewald summation formulae for point charge systems. Mol. Simul. 9, 351–368 (1992).
Frisch, M. Gaussian 09, Revision d. 01, Gaussian (Gaussian, 2009).
Schäfer, A., Horn, H. & Ahlrichs, R. Fully optimized contracted Gaussian basis sets for atoms Li to Kr. J. Chem. Phys. 97, 2571–2577 (1992).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Eichkorn, K., Weigend, F., Treutler, O. & Ahlrichs, R. Auxiliary basis sets for main row atoms and transition metals and their use to approximate Coulomb potentials. Theor. Chem. Acta 97, 119–124 (1997).
Binkley, J. S., Pople, J. A. & Hehre, W. J. Self-consistent molecular orbital methods. 21. Small split-valence basis sets for first-row elements. J. Am. Chem. Soc. 102, 939–947 (1980).
Gordon, M. S., Binkley, J. S., Pople, J. A., Pietro, W. J. & Hehre, W. J. Self-consistent molecular-orbital methods. 22. Small split-valence basis sets for second-row elements. J. Am. Chem. Soc. 104, 2797–2803 (1982).
Dobbs, K. D. & Hehre, W. J. Molecular orbital theory of the properties of inorganic and organometallic compounds 4. Extended basis sets for third‐and fourth‐row, main‐group elements. J. Comput. Chem. 7, 359–378 (1986).
Metz, B., Stoll, H. & Dolg, M. Small-core multiconfiguration-Dirac–Hartree–Fock-adjusted pseudopotentials for post-d main group elements: application to PbH and PbO. J. Chem. Phys. 113, 2563–2569 (2000).
Peterson, K. A., Figgen, D., Goll, E., Stoll, H. & Dolg, M. Systematically convergent basis sets with relativistic pseudopotentials. II. Small-core pseudopotentials and correlation consistent basis sets for the post-d group 16–18 elements. J. Chem. Phys. 119, 11113–11123 (2003).
Perdew, J. P., Ernzerhof, M. & Burke, K. Rationale for mixing exact exchange with density functional approximations. J. Chem. Phys. 105, 9982–9985 (1996).
Neese, F. The ORCA program system. WIREs Comput. Mol. Sci. 2, 73–78 (2012).
Neese, F. Software update: the ORCA program system—version 5.0. WIREs Comput. Mol. Sci. 12, e1606 (2022).
Neese, F., Wennmohs, F., Hansen, A. & Becker, U. Efficient, approximate and parallel Hartree–Fock and hybrid DFT calculations. A ‘chain-of-spheres’ algorithm for the Hartree–Fock exchange. Chem. Phys. 356, 98–109 (2009).
Douglas, M. & Kroll, N. M. Quantum electrodynamical corrections to the fine structure of helium. Ann. Phys. 82, 89–155 (1974).
Hess, B. A. Applicability of the no-pair equation with free-particle projection operators to atomic and molecular structure calculations. Phys. Rev. A 32, 756–763 (1985).
Hess, B. A. Relativistic electronic-structure calculations employing a two-component no-pair formalism with external-field projection operators. Phys. Rev. A 33, 3742–3748 (1986).
Neese, F. Efficient and accurate approximations to the molecular spin–orbit coupling operator and their use in molecular g-tensor calculations. J. Chem. Phys. 122, 034107 (2005).
Heß, B. A., Marian, C. M., Wahlgren, U. & Gropen, O. A mean-field spin–orbit method applicable to correlated wavefunctions. Chem. Phys. Lett. 251, 365–371 (1996).
Camiletti, G. G., Machado, S. F. & Jorge, F. E. Gaussian basis set of double zeta quality for atoms K through Kr: application in DFT calculations of molecular properties. J. Comput. Chem. 29, 2434–2444 (2008).
Barros, C. L., De Oliveira, P. J. P., Jorge, F. E., Canal Neto, A. & Campos, M. Gaussian basis set of double zeta quality for atoms Rb through Xe: application in non-relativistic and relativistic calculations of atomic and molecular properties. Mol. Phys. 108, 1965–1972 (2010).
Canal Neto, A. & Jorge, F. E. All-electron double zeta basis sets for the most fifth-row atoms: application in DFT spectroscopic constant calculations. Chem. Phys. Lett. 582, 158–162 (2013).
Canal Neto, A., Muniz, E. P., Centoducatte, R. & Jorge, F. E. Gaussian basis sets for correlated wave functions. Hydrogen, helium, first- and second-row atoms. J. Mol. Struct. THEOCHEM 718, 219–224 (2005).
Acknowledgements
This work is supported by the National Natural Science Foundation of China (grant numbers 22071003 to Z.L., 22271006 to Y.F., 12234001 to H.J., 61935016 to Z.B. and 92156016 to Z.L.), the National Key Research and Development Program of China (grant numbers 2022YFB3503702 to Z.L. and 2023YFB3506901 to Z.L.) and the Li Ge-Zhao Ning Youth Research Foundation (grant number LGZNQN202402 to Y.F.). We acknowledge the high-performance computing (HPC) platform of Peking University and Beijing Super Computing Center (BSCC) for providing HPC resources for the quantum-chemical calculations. The measurements of SCXRD and all the PL spectra in Edinburgh Instruments’ FLS980 PL spectrometer were performed at the Analytical Instrumentation Center of Peking University, with the help of J. Su and M. Chen, respectively.
Author information
Authors and Affiliations
Contributions
N.Z. and Y.F. conceived the idea. N.Z. designed the experiments. N.Z. synthesized and characterized the crystals with the help of C.H., H.L., J.Z., R.G. and W.Y. N.Z., X.J., T.Z., Z.Z. and J.S. solved the single-crystal structures. S.C. and H.J. performed the theoretical calculations. T.Z. performed the low-frequency Raman measurements. P.H. performed the stability tests. Z.L., Y.F., Z.B. and H.J. supervised the project. N.Z. and Y.F. wrote the paper with input of the theoretical part from S.C. All authors have interpreted the findings, revised the paper and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 0D metal halides prepared in this study.
The figure depicts the crystal structure of the compounds along with their symbol and colour coding.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–8, Figs. 1–24, CheckCIF information and Discussion.
Supplementary Data 1 (download ZIP )
Atomic coordinates of optimized computational models.
Source data
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, N., Cao, S., Zhang, T. et al. Synthesis of zero-dimensional octahedral metal halides through solvent incorporation and their photophysical properties. Nat. Chem. 17, 1401–1409 (2025). https://doi.org/10.1038/s41557-025-01869-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01869-x
This article is cited by
-
A universal antisolvent strategy for the synthesis of luminescent 0D metal halide perovskites
Science China Chemistry (2026)