Abstract
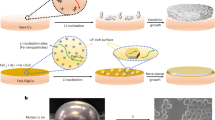
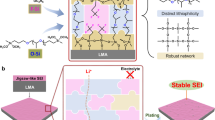
Understanding the lithium nucleation and growth process is crucial for improving lithium metal battery performance. Here we investigate the roles of the lithium–electrolyte and lithium–substrate interfaces during the lithium nucleation process. Using a physics-based model, we identify which of the two interfaces controls lithium nucleation for different electrolytes and substrates. Sluggish lithium transport through the solid–electrolyte interphases (SEIs) and slow charge-transfer kinetics make the nucleation process SEI controlled and substrate independent, while substrate properties control lithium nucleation in a system having fast SEI transport and charge-transfer reactions. For substrate-controlled nucleation, we derive a model that elucidates the need for fast lithium adatom velocity along the substrate that outpaces the critical nuclei formation. We also reveal that lithium nucleation modes have a strong impact on lithium plating/stripping reversibility. Simultaneous fast transport through the SEIs and fast lithium adatom movement on the substrate are essential for achieving dense lithium deposition and long-cycle-life lithium metal batteries.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information. Source data are provided with this paper.
References
Liu, J. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 4, 180–186 (2019).
Cheng, X.-B., Zhang, R., Zhao, C.-Z. & Zhang, Q. Toward safe lithium metal anode in rechargeable batteries: a review. Chem. Rev. 117, 10403–10473 (2017).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 12, 194–206 (2017).
Bai, P., Li, J., R. Brushett, F. & Bazant, Z. M. Transition of lithium growth mechanisms in liquid electrolytes. Energy Environ. Sci. 9, 3221–3229 (2016).
Zhang, W. et al. Recovery of isolated lithium through discharged state calendar ageing. Nature 626, 306–312 (2024).
Thirumalraj, B. et al. Nucleation and growth mechanism of lithium metal electroplating. J. Am. Chem. Soc. 141, 18612–18623 (2019).
Pei, A., Zheng, G., Shi, F., Li, Y. & Cui, Y. Nanoscale nucleation and growth of electrodeposited lithium metal. Nano Lett. 17, 1132–1139 (2017).
Yan, K. et al. Temperature-dependent nucleation and growth of dendrite-free lithium metal anodes. Angew. Chem. Int. Ed. 131, 11486–11490 (2019).
Guan, X. et al. Controlling nucleation in lithium metal anodes. Small 14, 1801423 (2018).
Chen, X.-R., Zhao, B.-C., Yan, C. & Zhang, Q. Review on Li deposition in working batteries: from nucleation to early growth. Adv. Mater. 33, 2004128 (2021).
Yan, C. et al. Nucleation and growth mechanism of anion-derived solid electrolyte interphase in rechargeable batteries. Angew. Chem. Int. Ed. 133, 8602–8606 (2021).
Li, N. et al. Normalized lithium growth from the nucleation stage for dendrite-free lithium metal anodes. Angew. Chem. Int. Ed. 131, 18414–18419 (2019).
Fan, Y. et al. Altering polythiophene derivative substrates to explore the mechanism of heterogeneous lithium nucleation for dendrite-free lithium metal anodes. J. Energy Chem. 59, 63–68 (2021).
Xu, Y. et al. Promoting mechanistic understanding of lithium deposition and solid-electrolyte interphase (SEI) formation using advanced characterization and simulation methods: recent progress, limitations, and future perspectives. Adv. Energy Mater. 12, 2200398 (2022).
Yan, K. et al. Selective deposition and stable encapsulation of lithium through heterogeneous seeded growth. Nat. Energy 1, 16010 (2016).
Fang, Y., Zhang, S. L., Wu, Z.-P. & Luan, D. A highly stable lithium metal anode enabled by Ag nanoparticle–embedded nitrogen-doped carbon macroporous fibers. Sci. Adv. 7, eabg3626 (2021).
Li, S.-Q. et al. A dendrite-free lithium-metal anode enabled by designed ultrathin MgF2 nanosheets encapsulated inside nitrogen-doped graphene-like hollow nanospheres. Adv. Mater. 34, 2201801 (2022).
Huang, G. et al. Lithiophilic 3D nanoporous nitrogen-doped graphene for dendrite-free and ultrahigh-rate lithium-metal anodes. Adv. Mater. 31, 1805334 (2019).
Zhang, R. et al. Lithiophilic sites in doped graphene guide uniform lithium nucleation for dendrite-free lithium metal anodes. Angew. Chem. Int. Ed. 129, 7872–7876 (2017).
He, J. & Manthiram, A. 3D CoSe@C aerogel as a host for dendrite-free lithium-metal anode and efficient sulfur cathode in Li–S full cells. Adv. Energy Mater. 10, 2002654 (2020).
Chi, S.-S. et al. Lithiophilic Zn sites in porous CuZn alloy induced uniform Li nucleation and dendrite-free Li metal deposition. Nano Lett. 20, 2724–2732 (2020).
Park, S., Jin, H.-J. & Yun, Y. S. Advances in the design of 3D-structured electrode materials for lithium-metal anodes. Adv. Mater. 32, 2002193 (2020).
Sun, J. et al. Lithium deposition mechanism on Si and Cu substrates in the carbonate electrolyte. Energy Environ. Sci. https://doi.org/10.1039/D2EE01833K (2022).
Ozhabes, Y., Gunceler, D. & Arias, T. A. Stability and surface diffusion at lithium-electrolyte interphases with connections to dendrite suppression. Preprint at https://arxiv.org/abs/1504.05799v1 (2015).
Wu, Z. et al. Growing single-crystalline seeds on lithiophobic substrates to enable fast-charging lithium-metal batteries. Nat. Energy 8, 340–350 (2023).
Boyle, D. T. et al. Resolving current-dependent regimes of electroplating mechanisms for fast charging lithium metal anodes. Nano Lett. 22, 8224–8232 (2022).
Yuan, X., Liu, B., Mecklenburg, M. & Li, Y. Ultrafast deposition of faceted lithium polyhedra by outpacing SEI formation. Nature 620, 86–91 (2023).
Biswal, P., Stalin, S., Kludze, A., Choudhury, S. & Archer, L. A. Nucleation and early stage growth of Li electrodeposits. Nano Lett. 19, 8191–8200 (2019).
Shi, Y. et al. Interfacial evolution of lithium dendrites and their solid electrolyte interphase shells of quasi-solid-state lithium-metal batteries. Angew. Chem. Int. Ed. 59, 18120–18125 (2020).
Fan, X. et al. Fluorinated solid electrolyte interphase enables highly reversible solid-state Li metal battery. Sci. Adv. 4, eaau9245 (2018).
Liu, Y. et al. Self-assembled monolayers direct a LiF-rich interphase toward long-life lithium metal batteries. Science https://doi.org/10.1126/science.abn1818 (2022).
Zhang, Q. et al. Synergetic effects of inorganic components in solid electrolyte interphase on high cycle efficiency of lithium ion batteries. Nano Lett. 16, 2011–2016 (2016).
Yan, C. et al. Lithium nitrate solvation chemistry in carbonate electrolyte sustains high-voltage lithium metal batteries. Angew. Chem. Int. Ed. 57, 14055–14059 (2018).
Liu, Y. et al. Electro-chemo-mechanical modeling of artificial solid electrolyte interphase to enable uniform electrodeposition of lithium metal anodes. Adv. Energy Mater. 12, 2103589 (2022).
Shen, X. et al. The failure of solid electrolyte interphase on Li metal anode: structural uniformity or mechanical strength? Adv. Energy Mater. 10, 1903645 (2020).
Li, N.-W. et al. A flexible solid electrolyte interphase layer for long-life lithium metal anodes. Angew. Chem. Int. Ed. 130, 1521–1525 (2018).
Yoon, I., Jurng, S., Abraham, D. P., Lucht, B. L. & Guduru, P. R. Measurement of mechanical and fracture properties of solid electrolyte interphase on lithium metal anodes in lithium ion batteries. Energy Storage Mater. 25, 296–304 (2020).
Liu, H. et al. Ultrahigh coulombic efficiency electrolyte enables Li||SPAN batteries with superior cycling performance. Mater. Today 42, 17–28 (2021).
Fan, X. et al. Non-flammable electrolyte enables Li-metal batteries with aggressive cathode chemistries. Nat. Nanotechnol. 13, 715–722 (2018).
Biswal, P. et al. The early-stage growth and reversibility of Li electrodeposition in Br-rich electrolytes. Proc. Natl Acad. Sci. USA 118, e2012071118 (2021).
Danzer, M. A. Generalized distribution of relaxation times analysis for the characterization of impedance spectra. Batteries 5, 53 (2019).
Holoubek, J. et al. Toward a quantitative interfacial description of solvation for Li metal battery operation under extreme conditions. Proc. Natl Acad. Sci. USA 120, e2310714120 (2023).
Zhang, S. S., Xu, K. & Jow, T. R. Electrochemical impedance study on the low temperature of Li-ion batteries. Electrochim. Acta 49, 1057–1061 (2004).
Liao, Y. Practical electron microscopy and database. https://www.globalsino.com/EM/ (2006).
Han, B. et al. Cryo-electron tomography of highly deformable and adherent solid-electrolyte interphase exoskeleton in Li-metal batteries with ether-based electrolyte. Adv. Mater. 34, 2108252 (2022).
Fan, X. et al. All-temperature batteries enabled by fluorinated electrolytes with non-polar solvents. Nat. Energy 4, 882–890 (2019).
Lu, Y., Tu, Z. & Archer, L. A. Stable lithium electrodeposition in liquid and nanoporous solid electrolytes. Nat. Mater. 13, 961–969 (2014).
Ma, X.-X. et al. The origin of fast lithium-ion transport in the inorganic solid electrolyte interphase on lithium metal anodes. Small Structures 3, 2200071 (2022).
Dong, K. et al. Unravelling the mechanism of lithium nucleation and growth and the interaction with the solid electrolyte interface. ACS Energy Lett. 6, 1719–1728 (2021).
Hui, Z. Source data NCHEM-24071878B.zip. figshare https://doi.org/10.6084/m9.figshare.29466917.v1 (2025).
Acknowledgements
The work was supported by the Office of Vehicle Technologies of the US Department of Energy through the Advanced Battery Materials Research (BMR) Program (Battery500 Consortium) under contract number PNNL-595241 (P.L.). Part of the work used the UCSD-MTI Battery Fabrication Facility. We specially acknowledge help from the UCSD cryo-EM centre and from M. Matyszewski with the cryo-S/TEM + EELS characterizations.
Author information
Authors and Affiliations
Contributions
Z.H., S.Y. and P.L. conceived the idea. P.L. directed the project. Z.H. developed the quantitative models and performed the electrochemical experiments and SEM characterizations. S.W. performed the XPS characterization. G.H. helped with the graphical representation of data. J.H. helped with model development and data analysis. K.Z. and M.L. helped with the cryo-TEM experiments. Q.M., V.P. and H.L. helped with SEM characterization. S.T. performed the conductive atomic force microscopy characterization. H.L. helped with electrolyte selection and preparation. K.Z., J.N. and J.Z. provided input on data interpretation and manuscription preparation. Z.H., S.Y., G.H., K.Z, J.N. and P.L. co-wrote and revised the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Tao Gao, Qiang Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1 and 2, Figs. 1–39 and discussion.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hui, Z., Yu, S., Wang, S. et al. Nucleation processes at interfaces with both substrate and electrolyte control lithium growth. Nat. Chem. 18, 33–42 (2026). https://doi.org/10.1038/s41557-025-01911-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01911-y
This article is cited by
-
Two roads to lithium nucleation
Nature Chemistry (2026)