Abstract
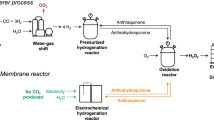
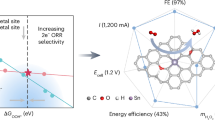
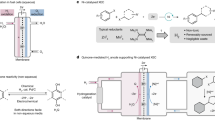
Hydrogen peroxide is manufactured industrially via the anthraquinone autoxidation process—a typical thermocatalytic non-aqueous method. Despite a high interest in using renewable electricity to drive such processes, electrifying non-aqueous syntheses remains a substantial challenge. Here we present a multi-phase electrochemical anthraquinone autoxidation process that leverages an aqueous–non-aqueous interfacial proton-coupled electron transfer method facilitated by heterogeneous molecular mediation. This design enables the reduction of aqueous anthraquinones with high efficiency at high current densities, using only carbon electrodes. The method operates with high selectivity through a quinhydrone intermediate and prevents the over-reduction of aromatics during thermocatalytic hydrogenation. This approach combines the benefits of aqueous electrochemistry with those of the traditional non-aqueous process to achieve high current density electrochemistry with rapid kinetics and mass transport, while avoiding unwanted electrolyte in the hydrogen peroxide product. This strategy bridges aqueous electrochemistry with non-aqueous chemistry and establishes a framework for the electrification and decentralization of other non-aqueous chemical processes.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available in the Article and Supplementary Information, and are available via figshare at https://doi.org/10.6084/m9.figshare.29817470 (ref. 43).
References
Goor, G., Glenneberg, J., Jacobi, S., Dadabhoy, J. & Candido, E. Hydrogen peroxide. In Ullmann’s Encyclopedia of Industrial Chemistry 1–40 (Wiley, 2019).
Wang, H.-X., Toh, W. L., Tang, B. Y. & Surendranath, Y. Metal surfaces catalyse polarization-dependent hydride transfer from H2. Nat. Catal. 6, 351–362 (2023).
Twilton, J. et al. Quinone-mediated hydrogen anode for non-aqueous reductive electrosynthesis. Nature 623, 71–76 (2023).
Minteer, S. D. & Baran, P. Electrifying synthesis: recent advances in the methods, materials, and techniques for organic electrosynthesis. Acc. Chem. Res. 53, 545–546 (2020).
Kawamata, Y. & Baran, P. S. Electrosynthesis: sustainability is not enough. Joule 4, 701–704 (2020).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemistry: calling all engineers. Angew. Chem. Int. Ed. Engl. 57, 4149–4159 (2017).
Jing, Y. et al. In situ electrochemical recomposition of decomposed redox-active species in aqueous organic flow batteries. Nat. Chem. 14, 1103–1109 (2022).
Huskinson, B. et al. A metal-free organic–inorganic aqueous flow battery. Nature 505, 195–198 (2014).
Elgazzar, A. & Wang, H. Beyond molecular transformations in electrochemical porous solid electrolyte reactors. Nat. Chem. Eng. 2, 3–7 (2025).
Mendez, M. A. et al. Molecular electrocatalysis at soft interfaces. Phys. Chem. Chem. Phys. 12, 15163–15171 (2010).
Marken, F. & Wadhawan, J. D. Multiphase methods in organic electrosynthesis. Acc. Chem. Res. 52, 3325–3338 (2019).
Perry, S. C. et al. Electrochemical synthesis of hydrogen peroxide from water and oxygen. Nat. Rev. Chem. 3, 442–458 (2019).
Tang, J. et al. Selective hydrogen peroxide conversion tailored by surface, interface, and device engineering. Joule 5, 1432–1461 (2021).
Zhang, X., Xia, Y., Xia, C. & Wang, H. Insights into practical-scale electrochemical H2O2 synthesis. Trends Chem. 2, 942–953 (2020).
Yamanaka, I. & Murayama, T. Neutral H2O2 synthesis by electrolysis of water and O2. Angew. Chem. Int. Ed. Engl. 120, 1926–1928 (2008).
Huang, A. et al. Direct H2O2 synthesis, without H2 gas. J. Am. Chem. Soc. 144, 14548–14554 (2022).
Xia, C., Xia, Y., Zhu, P., Fan, L. & Wang, H. Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte. Science 366, 226–231 (2019).
Xia, Y. et al. Highly active and selective oxygen reduction to H2O2 on boron-doped carbon for high production rates. Nat. Commun. 12, 4225 (2021).
Zhang, X. et al. Electrochemical oxygen reduction to hydrogen peroxide at practical rates in strong acidic media. Nat. Commun. 13, 2880 (2022).
Preger, Y. et al. Quinone-mediated electrochemical O2 reduction accessing high power density with an off-electrode Co-N/C catalyst. Joule 2, 2722–2731 (2018).
Murray, A. T., Voskian, S., Schreier, M., Hatton, T. A. & Surendranath, Y. Electrosynthesis of hydrogen peroxide by phase-transfer catalysis. Joule 3, 2942–2954 (2019).
Huissoud, A. & Tissot, P. Electrochemical reduction of 2-ethyl-9,10-anthraquinone (EAQ) and mediated formation of hydrogen peroxide in a two-phase medium part II: production of alkaline hydrogen peroxide by the intermediate electroreduction of EAQ in a flow-by porous electrode in two-phase liquid–liquid flow. J. Appl. Electrochem. 29, 17–25 (1999).
Fink, A. G. et al. Indirect H2O2 synthesis without H2. Nat. Commun. 15, 766 (2024).
Ji, Y. et al. A phosphonate functionalized quinone redox flow battery at near‐neutral pH with record capacity retention rate. Adv. Energy Mater. 9, 1900039 (2019).
Chanda, A. & Fokin, V. V. Organic synthesis “on water”. Chem. Rev. 109, 725–748 (2009).
Fajt, V., Kurc, L. & Červený, L. The effect of solvents on the rate of catalytic hydrogenation of 6‐ethyl‐1,2,3,4‐tetrahydroanthracene-9,10-dione. Int. J. Chem. Kinet. 40, 240–252 (2008).
Patil, A., Curtin, D. & Paul, I. Interconversion by hydrogen transfer of unsymmetrically substituted quinhydrones in the solid state. Crystal structure of the 1:2 complex of 2, 5-dimethylquinone with hydroquinone. J. Am. Chem. Soc. 106, 4010–4015 (1984).
Patil, A., Curtin, D. & Paul, I. Solid-state formation of quinhydrones from their components. Use of solid-solid reactions to prepare compounds not accessible from solution. J. Am. Chem. Soc. 106, 348–353 (1984).
Mercado, F. V., Fernández, R. A., Iglesias, R. A. & Dassie, S. A. Facilitated proton transfer reactions via water autoprotolysis across oil| water interfaces. Spectroelectrochemical analysis. Electrochim. Acta 299, 430–440 (2019).
Goulet, M. A. et al. Extending the lifetime of organic flow batteries via redox state management. J. Am. Chem. Soc. 141, 8014–8019 (2019).
Shang, H., Zhou, H., Zhang, W. & Zhang, K. Regeneration of working solution in the hydrogen peroxide production process. Can. J. Chem. Eng. 89, 1296–1302 (2011).
Lu, S., Wang, L., Wang, Y. & Mi, Z. Kinetic model of gas‐liquid‐liquid reactive extraction for production of hydrogen peroxide. Chem. Eng. Technol. 34, 823–830 (2011).
Oener, S. Z., Foster, M. J. & Boettcher, S. W. Accelerating water dissociation in bipolar membranes and for electrocatalysis. Science 369, 1099–1103 (2020).
Kerr, E. F. et al. High energy density aqueous flow battery utilizing extremely stable, branching-induced high-solubility anthraquinone near neutral pH. ACS Energy Lett. 8, 600–607 (2022).
Kurc, L., Páter, M. & Červený, L. Activity of basic catalysts in oxidation of 2-ethyl-5,6,7,8-tetrahydro-9,10-anthrahydroquinone. J. Mol. Catal. A 202, 327–332 (2003).
Osakai, T., Okamoto, M., Sugihara, T. & Nakatani, K. Bimolecular-reaction effect on the rate constant of electron transfer at the oil/water interface as studied by scanning electrochemical microscopy. J. Electroanal. Chem. 628, 27–34 (2009).
Cukier, R. I. & Nocera, D. G. Proton-coupled electron transfer. Annu. Rev. Phys. Chem. 49, 337–369 (1998).
Xia, C. et al. Continuous production of pure liquid fuel solutions via electrocatalytic CO2 reduction using solid-electrolyte devices. Nat. Energy 4, 776–785 (2019).
Lewis, R. J. & Hutchings, G. J. Selective oxidation using in situ-generated hydrogen peroxide. Acc. Chem. Res. 57, 106–119 (2023).
Bazant, M. Z. Unified quantum theory of electrochemical kinetics by coupled ion–electron transfer. Faraday Discuss. 246, 60–124 (2023).
Shang, H. et al. Scalable and selective gold recovery from end-of-life electronics. Nat. Chem. Eng. 1, 170–179 (2024).
Han, C. et al. Electrocatalytic hydrogenation of alkenes with Pd/carbon nanotubes at an oil–water interface. Nat. Catal. 5, 1110–1119 (2022).
Xi, D. et al. Electrifying industrial hydrogen peroxide production via soft interfacial molecular mediation. figshare https://doi.org/10.6084/m9.figshare.29817470.v2 (2025).
Acknowledgements
This research was supported by The Harvard Climate and Sustainability Translational Fund.
Author information
Authors and Affiliations
Contributions
D.X. conceived the idea. D.X. and Y.W. designed and conducted hardware design, tests and electrochemical experiments. Z.Y. studied the hydrogenation and performance of non-aqueous anthraquinones using hydrogen gas. Y.L. did the DFT calculation. R.Y.L. supervised the reaction mechanism study. M.J.A. supervised the project. D.X., Y.W. and M.J.A. drafted the manuscript. All authors edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Harvard University has filed a patent application (PCT/US25/10441) based on this work with D.X., Y.W. and M.J.A. as inventors for ‘Electrochemically reducing an aqueous-soluble, non-aqueous-insoluble molecule in an aqueous electrochemical cell to provide ANIHAT to a molecule from which H2O2 is produced upon exposure to O2’. M.J.A. is co-founder of Adiabatic Materials, a startup company that has licensed this intellectual property from Harvard. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Joaquim Henrique Teles and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–27, Tables 1–4 and Data 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xi, D., Wu, Y., Li, Y. et al. Electrifying industrial hydrogen peroxide production via soft interfacial molecular mediation. Nat. Chem. 17, 1883–1890 (2025). https://doi.org/10.1038/s41557-025-01940-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01940-7