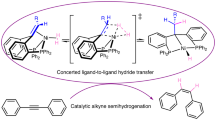
Abstract
Catalytic hydrogenation is essential in synthetic chemistry, with ongoing innovations aimed at enhancing the selectivity and efficiency of these reactions. The hydrogenation of multisubstituted allenes, however, presents a long-standing challenge due to the difficulty of controlling multiple selectivity factors simultaneously. Here we introduce a series of chiral pincer cobalt catalysts featuring multiple metal–ligand cooperative functionalities. These catalysts feature an ‘N–H’ moiety as an outer-sphere binding site and an N-heterocycle group as a hemilabile basic site, enabling the use of structurally diverse tridentate ligands for selective hydrogenation of functionalized allenes. This design liberates a coordination site for H2 activation and enhances selectivity control through the structural tuning of the N-heterocycle group. The catalysts exhibit exceptional chemo-, regio-, enantio- and Z/E-selectivities, along with broad functional group tolerance, enabling access to all possible semihydrogenation products of multisubstituted allenes. Mechanistic studies uncover a distinctive redox-neutral Co(I) catalytic cycle that facilitates heterolytic cleavage of H2, assisted by the basic N-heterocycle on the ligand.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available in this Article and its Supplementary Information. The crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers CCDC 2345206 (11), 2345208 (RL7-CoCl2), 2397603 (RL11-CoCl2), 2345207 (RL12-CoCl2), 2397604 (RL14-CoCl2), 2397605 (RL15-CoCl2) and 2397606 (RL17-CoCl2). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures.
References
de Vries, J. G. & Elsevier, C. J. The Handbook of Homogeneous Hydrogenation (John Wiley & Sons, 2006).
Osborn, J. A., Jardine, F. H., Young, J. F. & Wilkinson, G. The preparation and properties of tris(triphenylphosphine)halogenorhodium(I) and some reactions thereof including catalytic homogeneous hydrogenation of olefins and acetylenes and their derivatives. J. Chem. Soc. A https://doi.org/10.1039/j19660001711 (1966).
Wu, H. et al. Site- and enantioselective iridium-catalyzed desymmetric mono-hydrogenation of 1,4-dienes. Angew. Chem. Int. Ed. 60, 19428–19434 (2021).
Kong, D. et al. Site-selective hydrogenation of electron-poor alkenes and dienes enabled by a rhodium-catalyzed hydride addition/protonolysis mechanism. Angew. Chem. Int. Ed. 61, e202210601 (2022).
Guan, J., Chen, J., Luo, Y., Guo, L. & Zhang, W. Copper-catalyzed chemoselective asymmetric hydrogenation of C=O bonds of exocyclic α,β-unsaturated pentanones. Angew. Chem. Int. Ed. 62, e202306380 (2023).
Hu, Y. et al. Precise synthesis of chiral Z‐allylamides by cobalt‐catalyzed asymmetric sequential hydrogenations. Angew. Chem. Int. Ed. 62, e202217871 (2023).
Chen, Z. & Dong, V. M. Enantioselective semireduction of allenes. Nat. Commun. 8, 784 (2017).
Ma, S. Transition metal-catalyzed/mediated reaction of allenes with a nucleophilic functionality connected to the α-carbon atom. Acc. Chem. Res. 36, 701–712 (2003).
Hashimoto, T., Sakata, K., Tamakuni, F., Dutton, M. J. & Maruoka, K. Phase-transfer-catalysed asymmetric synthesis of tetrasubstituted allenes. Nat. Chem. 5, 240–244 (2013).
Holmes, M., Schwartz, L. A. & Krische, M. J. Intermolecular metal-catalyzed reductive coupling of dienes, allenes, and enynes with carbonyl compounds and imines. Chem. Rev. 118, 6026–6052 (2018).
Ju, T. et al. Dicarboxylation of alkenes, allenes and (hetero)arenes with CO2 via visible-light photoredox catalysis. Nat. Catal. 4, 304–311 (2021).
Wang, W. et al. Stereodefined rhodium-catalysed 1,4-H/D delivery for modular syntheses and deuterium integration. Nat. Catal. 4, 586–594 (2021).
Han, X., Wang, M., Liang, Y., Zhao, Y. & Shi, Z. Regio- and enantioselective nucleophilic addition to gem-difluoroallenes. Nat. Synth. 1, 227–234 (2022).
Tan, T.-D. et al. Kinetically controlled Z-alkene synthesis using iron-catalysed allene dialkylation. Nat. Synth. 4, 116–123 (2025).
Guo, H. et al. [Pd(Ar-BIAN)(alkene)]-catalyzed highly chemo-, regio-, and stereoselective semihydrogenation of 1,2-allenyl phosphonates and related compounds. Angew. Chem. Int. Ed. 45, 4997–5000 (2006).
Long, J., Shi, L., Li, X., Lv, H. & Zhang, X. Rhodium‐catalyzed highly regio‐ and enantioselective hydrogenation of tetrasubstituted allenyl sulfones: an efficient access to chiral allylic sulfones. Angew. Chem. Int. Ed. 57, 13248–13251 (2018).
Liu, G. et al. Challenging task of Ni-catalyzed highly regio-/enantioselective semihydrogenation of racemic tetrasubstituted allenes via a kinetic resolution process. J. Am. Chem. Soc. 146, 7419–7430 (2024).
Burke, M. D. & Schreiber, S. L. A planning strategy for diversity-oriented synthesis. Angew. Chem. Int. Ed. 43, 46–58 (2004).
Negishi, E.-i et al. Recent advances in efficient and selective synthesis of di-, tri-, and tetrasubstituted alkenes via Pd-catalyzed alkenylation-carbonyl olefination synergy. Acc. Chem. Res. 41, 1474–1485 (2008).
Duan, X.-F. Iron catalyzed stereoselective alkene synthesis: a sustainable pathway. Chem. Commun. 56, 14937–14961 (2020).
Chirik, P. J. Iron- and cobalt-catalyzed alkene hydrogenation: catalysis with both redox-active and strong field ligands. Acc. Chem. Res. 48, 1687–1695 (2015).
Filonenko, G. A., van Putten, R., Hensen, E. J. M. & Pidko, E. A. Catalytic (de)hydrogenation promoted by non-precious metals—Co, Fe and Mn: recent advances in an emerging field. Chem. Soc. Rev. 47, 1459–1483 (2018).
Liu, W., Sahoo, B., Junge, K. & Beller, M. Cobalt complexes as an emerging class of catalysts for homogeneous hydrogenations. Acc. Chem. Res. 51, 1858–1869 (2018).
Mukherjee, A. & Milstein, D. Homogeneous catalysis by cobalt and manganese pincer complexes. ACS Catal. 8, 11435–11469 (2018).
Ai, W., Zhong, R., Liu, X. & Liu, Q. Hydride transfer reactions catalyzed by cobalt complexes. Chem. Rev. 119, 2876–2953 (2019).
Chakrabortty, S., de Bruin, B. & de Vries, J. G. Cobalt-catalyzed asymmetric hydrogenation: substrate specificity and mechanistic variability. Angew. Chem. Int. Ed. 63, e202315773 (2024).
Monfette, S., Turner, Z. R., Semproni, S. P. & Chirik, P. J. Enantiopure C1-symmetric bis(imino)pyridine cobalt complexes for asymmetric alkene hydrogenation. J. Am. Chem. Soc. 134, 4561–4564 (2012).
Friedfeld, M. R. S. et al. Cobalt precursors for high-throughput discovery of base metal asymmetric alkene hydrogenation catalysts. Science 342, 1076–1080 (2013).
Friedfeld, M. R., Zhong, H., Ruck, R. T., Shevlin, M. & Chirik, P. J. Cobalt-catalyzed asymmetric hydrogenation of enamides enabled by single-electron reduction. Science 360, 888–893 (2018).
Zhong, H., Friedfeld, M. R. & Chirik, P. J. Syntheses and catalytic hydrogenation performance of cationic bis(phosphine) cobalt(I) diene and arene Compounds. Angew. Chem. Int. Ed. 58, 9194–9198 (2019).
Du, X. et al. Cobalt-catalyzed highly enantioselective hydrogenation of α,β-unsaturated carboxylic acids. Nat. Commun. 11, 3239 (2020).
Viereck, P., Krautwald, S., Pabst, T. P. & Chirik, P. J. A boron activating effect enables cobalt-catalyzed asymmetric hydrogenation of sterically hindered alkenes. J. Am. Chem. Soc. 142, 3923–3930 (2020).
Du, X. et al. Enantioselective hydrogenation of tetrasubstituted α,β‐unsaturated carboxylic acids enabled by cobalt(II) catalysis: scope and mechanistic insights. Angew. Chem. Int. Ed. 60, 11384–11390 (2021).
Hu, Y., Zhang, Z., Liu, Y. & Zhang, W. Cobalt‐catalyzed chemo‐ and enantioselective hydrogenation of conjugated enynes. Angew. Chem. Int. Ed. 60, 16989–16993 (2021).
Lu, P., Wang, H., Mao, Y., Hong, X. & Lu, Z. Cobalt-catalyzed enantioconvergent hydrogenation of minimally functionalized isomeric olefins. J. Am. Chem. Soc. 144, 17359–17364 (2022).
Mendelsohn, L. N. et al. Mechanistic investigations of the asymmetric hydrogenation of enamides with neutral bis(phosphine) cobalt precatalysts. J. Am. Chem. Soc. 144, 15764–15778 (2022).
Chen, T. et al. Cobalt‐catalyzed efficient convergent asymmetric hydrogenation of E/Z‐enamides. Angew. Chem. Int. Ed. 62, e202303488 (2023).
Chakrabortty, S. et al. Cobalt‐catalyzed enantioselective hydrogenation of trisubstituted carbocyclic olefins: an access to chiral cyclic amides. Angew. Chem. Int. Ed. 62, e202301329 (2023).
Li, M., Wang, Z., Chen, H., Huang, Q. & Zuo, W. Metal charge-directed enantiodivergent asymmetric transfer hydrogenation of ketones. Chem 10, 250–264 (2024).
Wang, Z., Li, M. & Zuo, W. Cobalt-catalyzed asymmetric hydrogenation of ketones enabled by the synergism of an N–H functionality and a redox-active ligand. J. Am. Chem. Soc. 146, 26416–26426 (2024).
Hu, Y. et al. Cobalt‐catalyzed asymmetric hydrogenation of C=N bonds enabled by assisted coordination and nonbonding interactions. Angew. Chem. Int. Ed. 58, 15767–15771 (2019).
Junge, K., Papa, V. & Beller, M. Cobalt-pincer complexes in catalysis. Chem. Eur. J. 25, 122–143 (2019).
Wang, H., Wen, J. & Zhang, X. Chiral tridentate ligands in transition metal-catalyzed asymmetric hydrogenation. Chem. Rev. 121, 7530–7567 (2021).
Halpern, J. Mechanism and stereoselectivity of asymmetric hydrogenation. Science 217, 401–407 (1982).
Zhang, G., Vasudevan, K. V., Scott, B. L. & Hanson, S. K. Understanding the mechanisms of cobalt-catalyzed hydrogenation and dehydrogenation reactions. J. Am. Chem. Soc. 135, 8668–8681 (2013).
Zhao, B., Han, Z. & Ding, K. The N–H functional group in organometallic catalysis. Angew. Chem. Int. Ed. 52, 4744–4788 (2013).
Khusnutdinova, J. R. & Milstein, D. Metal-ligand cooperation. Angew. Chem. Int. Ed. 54, 12236–12273 (2015).
Rösler, S., Obenauf, J. & Kempe, R. A highly active and easily accessible cobalt catalyst for selective hydrogenation of C=O bonds. J. Am. Chem. Soc. 137, 7998–8001 (2015).
Srimani, D. et al. Cobalt-catalyzed hydrogenation of esters to alcohols: unexpected reactivity trend indicates ester enolate intermediacy. Angew. Chem. Int. Ed. 54, 12357–12360 (2015).
Dub, P. A. & Gordon, J. C. The role of the metal-bound N–H functionality in Noyori-type molecular catalysts. Nat. Rev. Chem. 2, 396–408 (2018).
Junge, K. et al. Cobalt pincer complexes for catalytic reduction of carboxylic acid esters. Chem. Eur. J. 24, 1046–1052 (2018).
Elsby, M. R. & Baker, R. T. Strategies and mechanisms of metal-ligand cooperativity in first-row transition metal complex catalysts. Chem. Soc. Rev. 49, 8933–8987 (2020).
Jalón, F. A. et al. Facile Ru-H2 heterolytic activation and intramolecular proton transfer assisted by basic N-centers in the ligands. J. Am. Chem. Soc. 127, 15364–15365 (2005).
Fu, S. et al. Ligand-controlled cobalt-catalyzed transfer hydrogenation of alkynes: stereodivergent synthesis of Z- and E-alkenes. J. Am. Chem. Soc. 138, 8588–8594 (2016).
Liu, X., Rong, X., Liu, S., Lan, Y. & Liu, Q. Cobalt-catalyzed desymmetric isomerization of exocyclic olefins. J. Am. Chem. Soc. 143, 20633–20639 (2021).
Rong, X., Yang, J., Liu, S., Lan, Y. & Liu, Q. Remote stereocontrol of all-carbon quaternary centers via cobalt-catalyzed asymmetric olefin isomerization. CCS Chem. 5, 1293–1300 (2023).
Horsman, G. P. & Zechel, D. L. Phosphonate biochemistry. Chem. Rev. 117, 5704–5783 (2017).
Bennett, B. EPR of Co(II) as a structural and mechanistic probe of metalloprotein active sites: a review of studies on aminopeptidase. Physics Faculty Research and Publications https://epublications.marquette.edu/physics_fac/135 (2002).
Fernández, I. & Bickelhaupt, F. M. The activation strain model and molecular orbital theory: understanding and designing chemical reactions. Chem. Soc. Rev. 43, 4953–4967 (2014).
Lu, T. & Chen, Q. Simple, efficient, and universal energy decomposition analysis method based on dispersion-corrected density functional theory. J. Phys. Chem. A 127, 7023–7035 (2023).
Lu, T. & Chen, Q. Independent gradient model based on hirshfeld partition: a new method for visual study of interactions in chemical systems. J. Comput. Chem. 43, 539–555 (2022).
Gant, T. G. Using deuterium in drug discovery: leaving the label in the drug. J. Med. Chem. 57, 3595–3611 (2014).
Cao, H.-Q., Li, J.-K., Zhang, F.-G., Cahard, D. & Ma, J.-A. Asymmetric synthesis of chiral amino carboxylic-phosphonic acid derivatives. Adv. Synth. Catal. 363, 688–729 (2021).
Acknowledgements
We thank H. Jiao from Leibniz Institute for Catalysis and S. Liu from Henan University for helpful discussions. Financial support from the National Natural Science Foundation of China (grant nos. 22225103 (Q.L.) and 22202123 (Z.W.)) and the Tsinghua University Initiative Scientific Research Program are greatly appreciated.
Author information
Authors and Affiliations
Contributions
Conceptualization: X.R., S.M. and Q.L. Methodology: X.R. and Y.R. Investigation: X.R., Y.R., Y.C., C.L., J.W., Z.C. and L.T. Funding acquisition: Z.W. and Q. L. Project administration: L.T., S.M., Z.W. and Q.L. Supervision: L.T., S.M., Z.W. and Q.L. Writing—original draft: X.R., Z.W. and Q.L. Writing—review and editing: X.R. and Q.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Chen Zhu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
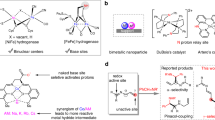
Extended Data Fig. 1 Structure of cobalt catalysts.
a, Solid-state structure of RL7-CoCl2 and RL12-CoCl2 determined by single-crystal X-ray diffraction. Ellipsoids set at 50% probability. Hydrogen atoms are omitted for clarity. Colour code for the atoms in the structures: grey, carbon; light blue, nitrogen; red, oxygen; purple, phosphorus; green, chlorine; royal blue, iron; dark blue, cobalt. b, Dissociation free energy of the N-heterocyclic ligand. T and B denote tridentate and bidentate coordination structures, respectively, and superscript Q indicates quartet spin states. ΔG values (in kcal mol−1) were calculated at the M06L/def2-TZVP/SMD(DMAc)//M06L/def2-SVP level.
Extended Data Fig. 2 Tunable cobalt-catalyzed hydrogenation of racemic trisubstituted allenes.
a, Divergent hydrogenation of racemic trisubstituted allenes. b, Substrate scope of asymmetric 1,2-hydrogenation of racemic allenes. Reaction conditions: a rac-5 (0.25 mmol), Co(BF4)2.6H2O (2 mol%), RL7 or SL7 (2.2 mol%) and zinc dust (20 mol%) in 1 mL of DMAc at 40 °C for 20 h. Isolated yields (%) were given, regioselective ratio (r.r. 6/7) was determined by 31P NMR and enantiomeric excess (% e.e.) was determined by chiral-phase HPLC. b CoCl2 (2 mol%), RL12 (2.2 mol%) and zinc dust (20 mol%) in HFIP (1 mL) at 60 °C for 20 h. E/Z ratio was determined by 31P NMR. c CoCl2 (2 mol%), RL16 (2.2 mol%) and zinc dust (20 mol%) in DMAc (1 mL) at 60 °C for 40 h. Z/E ratio was determined by 31P NMR.
Extended Data Fig. 3 Catalytic cycle.
Proposed mechanism for the stereoselective cobalt-catalyzed hydrogenation of functionalized allenes. DFT calculations were performed at the M06L/Def2-TZVP/SMD(DMAc)//M06L/Def2-SVP level of theory.
Extended Data Fig. 4 Synthetic Applications.
a, Syntheses of deuterated trisubstituted alkene products. b, Gram-scale synthesis with 0.2 mol% catalyst. c, Post functionalisation of reaction products. d, Preparation of intermediate of phosphonomethyl-phenylalanine analogue. i Lawesson’s reagent, toluene, 100 °C. ii TMSBr, DCM, r.t. iii mCPBA, −20 °C. iv NBS, DMSO/H2O (v/v = 7/5), r.t. v NIS, DMSO/H2O (v/v = 7/5), r.t. vi 1) BH3, THF, -20 °C, 2) H2O2, NaOAc, 0 °C. vii 1) Pd(PPh3)4 (2 mol%), CuI (2 mol%), 3-Methyl butynol and Et3N, 80 °C. 2) P(OEt)2Cl, Et3N, DCM, −78 °C - r.t. viii Co(BF4)2·6H2O (2 mol%), RL7 (2.2 mol%) and zinc dust (5 mol%) in 1 mL of DMAc under H2 (30 bar) at 40 °C for 20 h.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–49, Supplementary Tables 1–13, Experimental procedures and characterization data, DFT calculation results, Discussion and References.
Supplementary Data (download ZIP )
x,y,z coordinates for DFT calculations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rong, X., Ren, Y., Chen, Y. et al. Tunable cobalt-catalysed hydrogenation of allenes enabled by multiple metal–ligand cooperative functionalities. Nat. Chem. 17, 1469–1479 (2025). https://doi.org/10.1038/s41557-025-01945-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01945-2
This article is cited by
-
Cooperative ligands enable cobalt-catalysed hydrogenation
Nature Chemistry (2025)