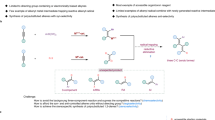
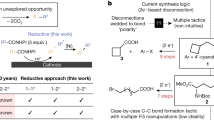
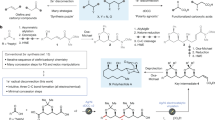
Abstract
The pursuit of increasingly complex, three-dimensional molecules is pushing the boundaries of modern organic synthesis, particularly in drug discovery where rigid, saturated scaffolds such as cyclobutanes, azetidines and oxetanes are in high demand. Here we outline a modular, scalable, chemoselective approach to solve this problem using simple α-bromoacids and aryl halides as intuitive starting materials. As demonstrated herein, a sequential series of nickel-electrocatalytic cross-couplings can be enlisted to enable rapid access to such structures, many of which have been nearly impossible to access before without recourse to time-consuming polar bond disconnections that are inherently limiting in terms of accessible chemical space. The scalability of this new reaction sequence is demonstrated, alongside direct applications to known patented structures. A simple user guide is also presented to accelerate adoption of this strategy in medicinal chemistry and related fields.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main text or Supplementary Information. Experimental procedures in graphical form, additional experimental results, NMR characterization data and X-ray crystallographic details for all new compounds prepared in this study are provided in Supplementary Information. The X-ray crystallographic coordinates for compound Ni(L10)Cl2 have been deposited at the Cambridge Crystallographic Data Centre with the accession code 2388490. Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures/.
References
Bauer, M. R. et al. Put a ring on it: application of small aliphatic rings in medicinal chemistry. RSC Med. Chem. 12, 448–471 (2021).
van der Kolk, M. R., Janssen, M. A. C. H., Rutjes, F. P. J. T. & Blanco-Ania, D. Cyclobutanes in small-molecule drug candidates. ChemMedChem 17, e202200020 (2022).
Wrobleski, M. L. et al. Cyclobutane derivatives as potent NK1 selective antagonists. Bioorg. Med. Chem. Lett. 16, 3859–3863 (2006).
Meanwell, N. A. Improving drug design: an update on recent applications of efficiency metrics, strategies for replacing problematic elements, and compounds in nontraditional drug space. Chem. Res. Toxicol. 29, 564–616 (2016).
Burkhard, J. A., Wuitschik, G., Rogers-Evans, M., Müller, K. & Carreira, E. M. Oxetanes as versatile elements in drug discovery and synthesis. Angew. Chem. Int. Ed. 49, 9052–9067 (2010).
Subbaiah, M. A. M. & Meanwell, N. A. Bioisosteres of the phenyl ring: recent strategic applications in lead optimization and drug design. J. Med. Chem. 64, 14046–14128 (2021).
Mughal, H. & Szostak, M. Recent advances in the synthesis and reactivity of azetidines: strain-driven character of the four-membered heterocycle. Org. Biomol. Chem. 19, 3274–3286 (2021).
Hsu, C.-M. et al. Azetidines with all-carbon quaternary centers: merging relay catalysis with strain release functionalization. J. Am. Chem. Soc. 145, 19049–19059 (2023).
Bull, J. A., Croft, R. A., Davis, O. A., Doran, R. & Morgan, K. F. Oxetanes: recent advances in synthesis, reactivity, and medicinal chemistry. Chem. Rev. 116, 12150–12233 (2016).
Denis, C. et al. Synthesis of 3,3-diarylazetidines by calcium(II)-catalyzed Friedel–Crafts reaction of azetidinols with unexpected Cbz enhanced reactivity. Org. Lett. 21, 300–304 (2019).
Tian, D., Chen, G., Wang, X. & Zhang, H.-J. Modular access to functionalized oxetanes as benzoyl bioisosteres. J. Am. Chem. Soc. 146, 18011–18018 (2024).
Nicolaou, K. C. et al. Synthesis and biopharmaceutical evaluation of imatinib analogues featuring unusual structural motifs. ChemMedChem 11, 31–37 (2016).
Altenbach Robert, J., Cowart Marlon, D., Liu, H. & Pu, Y.-M. Bicyclic-substituted amines having cyclic-substituted monocyclic substituents. US patent 2005/0272728 A1 (2005).
Baumann, K. et al. Modulators for amyloid beta. WO patent 2009/103652 A1 (2009).
Baumann, K. et al. Gamma secretase modulators. US patent 2011/0190269 A1 (2011).
Zhang, Y., Hodous Brian, L., Kim Joseph, L., Wilson Kevin, J. & Wilson, D. Compositions useful for treating disorders related to KIT. WO patent 2015/057873 A1 (2015).
Huestis, M. et al. Lactams as CBL-B inhibitors selective over C-CBL. WO patent 2023/081853 A1 (2023).
Bock William, J. et al. Substituted bicyclic pyridone derivatives. WO patent 2024/105563 A1 (2024).
Croft, R. A. et al. Catalytic Friedel–Crafts reactions on saturated heterocycles and small rings for sp3–sp2 coupling of medicinally relevant fragments. Eur. J. Org. Chem. 2019, 5385–5395 (2019).
Li, X.-S. et al. Lewis-acid-catalyzed tandem cyclization by ring expansion of tertiary cycloalkanols with propargyl alcohols. Org. Lett. 23, 9457–9462 (2021).
Wang, J. et al. Ag-catalyzed ring-opening of tertiary cycloalkanols for C–H functionalization of cyclic aldimines. Chem. Commun. 57, 1506–1509 (2021).
Wang, X. et al. Brønsted acid-catalyzed Markovnikov hydroarylation of arylcyclobutene: a route to gem-diaryl-substituted cyclobutanes. Adv. Synth. Catal. 366, 2705–2709 (2024).
Arseniyadis, S., Kyler, K. S. & Watt, D. S. in Organic Reactions Ch. 6 (Wiley, 2005).
He, Z.-T. & Hartwig, J. F. Palladium-catalyzed α-arylation for the addition of small rings to aromatic compounds. Nat. Commun. 10, 4083 (2019).
Wright, B. A. & Ardolino, M. J. Surprising reactivity in NiXantphos/palladium-catalyzed α-arylation of substituted cyclopropyl nitriles. J. Org. Chem. 84, 4670–4679 (2019).
McCabe Dunn, J. M., Kuethe, J. T., Orr, R. K., Tudge, M. & Campeau, L.-C. Development of a palladium-catalyzed α-arylation of cyclopropyl nitriles. Org. Lett. 16, 6314–6317 (2014).
Ariki, Z. T., Maekawa, Y., Nambo, M. & Crudden, C. M. Preparation of quaternary centers via nickel-catalyzed Suzuki–Miyaura cross-coupling of tertiary sulfones. J. Am. Chem. Soc. 140, 78–81 (2018).
Gao, L. et al. Lewis acid-catalyzed selective reductive decarboxylative pyridylation of N-hydroxyphthalimide esters: synthesis of congested pyridine-substituted quaternary carbons. ACS Catal. 9, 10142–10151 (2019).
Polites, V. C., Badir, S. O., Keess, S., Jolit, A. & Molander, G. A. Nickel-catalyzed decarboxylative cross-coupling of bicyclo[1.1.1]pentyl radicals enabled by electron donor–acceptor complex photoactivation. Org. Lett. 23, 4828–4833 (2021).
Xue, W. et al. Nickel-catalyzed formation of quaternary carbon centers using tertiary alkyl electrophiles. Chem. Soc. Rev. 50, 4162–4184 (2021).
Mills, L. R., Monteith, J. J., dos Passos Gomes, G., Aspuru-Guzik, A. & Rousseaux, S. A. L. The cyclopropane ring as a reporter of radical leaving-group reactivity for Ni-catalyzed C(sp3)–O arylation. J. Am. Chem. Soc. 142, 13246–13254 (2020).
Liu, W., Lavagnino, M. N., Gould, C. A., Alcázar, J. & MacMillan, D. W. C. A biomimetic SH2 cross-coupling mechanism for quaternary sp3-carbon formation. Science 374, 1258–1263 (2021).
Salgueiro, D. C., Chi, B. K., Guzei, I. A., García-Reynaga, P. & Weix, D. J. Control of redox-active ester reactivity enables a general cross-electrophile approach to access arylated strained rings. Angew. Chem. Int. Ed. 61, e202205673 (2022).
Laudadio, G. et al. Nickel-electrocatalytic decarboxylative arylation to access quaternary centers. Angew. Chem. Int. Ed. 63, e202314617 (2024).
Gan, X.-c. et al. Carbon quaternization of redox active esters and olefins by decarboxylative coupling. Science 384, 113–118 (2024).
Chen, R. et al. Alcohol–alcohol cross-coupling enabled by SH2 radical sorting. Science 383, 1350–1357 (2024).
Gütz, C., Bänziger, M., Bucher, C., Galvão, T. R. & Waldvogel, S. R. Development and scale-up of the electrochemical dehalogenation for the synthesis of a key intermediate for NS5A inhibitors. Org. Process Res. Dev. 19, 1428–1433 (2015).
Palkowitz, M. D. et al. Overcoming limitations in decarboxylative arylation via Ag–Ni electrocatalysis. J. Am. Chem. Soc. 144, 17709–17720 (2022).
Harwood, S. J. et al. Modular terpene synthesis enabled by mild electrochemical couplings. Science 375, 745–752 (2022).
Huang, H., Alvarez-Hernandez, J. L., Hazari, N., Mercado, B. Q. & Uehling, M. R. Effect of 6,6′-substituents on bipyridine-ligated Ni catalysts for cross-electrophile coupling. ACS Catal. 14, 6897–6914 (2024).
Erlanson, D. et al. Modulators of G-protein coupled receptors. WO patent 2022/241287 A3 (2022).
Deng, Y. et al. Novel substituted tetrahydroquinolin compounds as indoleamine 2,3-dioxygenase (IDO) inhibitors. WO patent 2019/089412 A1 (2019).
Salituro Francesco, G., Saunders Jeffrey, O. & Yan, S. Therapeutic compounds and compositions. WO patent 2012/092442 A1 (2012).
Bartolozzi, A. et al. Heterocyclic compounds as inhibitors of leukotriene production. US patent 2013/0196967 A1 (2013).
Dai, X., Jiang, Y. & Liu, Y. Novel salts of indoleamine 2,3-dioxygenase inhibitors. WO patent 2021/102618 A1 (2021).
Ma, L. I., Guo, J. & Zhou, Y. Synthetic method of aryl cyclobutane compound. CN patent 113861071 A (2021).
He, W. et al. Identification of a novel small-molecule binding site of the fat mass and obesity associated protein (FTO). J. Med. Chem. 58, 7341–7348 (2015).
Huxley, P., Heal Jonathan, R. & Todd Richard, S. Cycloalkyl amine compounds. WO patent 2015/027058 A2 (2015).
Dubois, M. A. J. et al. Short synthesis of oxetane and azetidine 3-aryl-3-carboxylic acid derivatives by selective furan oxidative cleavage. Org. Lett. 22, 5279–5283 (2020).
Guo, J. et al. Unlocking tertiary acids for metallaphotoredox C(sp2)–C(sp3) decarboxylative cross-couplings. ACS Catal. 13, 11910–11918 (2023).
Everson, D. A., Jones, B. A. & Weix, D. J. Replacing conventional carbon nucleophiles with electrophiles: nickel-catalyzed reductive alkylation of aryl bromides and chlorides. J. Am. Chem. Soc. 134, 6146–6159 (2012).
Zhang, P., Le, C. C. & MacMillan, D. W. C. Silyl radical activation of alkyl halides in metallaphotoredox catalysis: a unique pathway for cross-electrophile coupling. J. Am. Chem. Soc. 138, 8084–8087 (2016).
Franke, M. C. et al. Zinc-free, scalable reductive cross-electrophile coupling driven by electrochemistry in an undivided cell. ACS Catal. 12, 12617–12626 (2022).
Neigenfind, P. et al. Simplifying access to targeted protein degraders via nickel electrocatalytic cross-coupling. Angew. Chem. Int. Ed. 63, e202319856 (2024).
Hioki, Y. et al. Overcoming the limitations of Kolbe coupling with waveform-controlled electrosynthesis. Science 380, 81–87 (2023).
Garrido-Castro, A. F. et al. Scalable electrochemical decarboxylative olefination driven by alternating polarity. Angew. Chem. Int. Ed. 62, e202309157 (2023).
Acknowledgements
Financial support for this work was provided by the NSF Center for Synthetic Organic Electrochemistry (CHE-2002158). L.M. thanks the Swedish Research Council (Vetenskapsrådet, VR-2023-00499) and the Stiftelsen Bengt Lundqvists Minne for postdoctoral fellowships. P.N. thanks the Kellogg Family for a graduate research fellowship. We thank L. Pasternack and G. J. Kroon for assistance with NMR spectroscopy; J. B. Bailey for X-ray crystallographic analysis; J. Chen, B. Sanchez, Q. N. Wong and J. Lee for assistance with high-resolution mass spectrometry; and M. Costantini, Á. Péter, M. Nassir, A. Pollatos, G. Laudadio, T. El-Hayek Ewing and G. Leoni for helpful discussions.
Author information
Authors and Affiliations
Contributions
L.M., P.N. and P.S.B. conceived the project. All authors were involved in designing, performing and analysing the experiments and writing the paper. P.S.B. directed the project.
Corresponding author
Ethics declarations
Competing interests
P.S.B. is a paid consultant at Bristol Myers Squibb. The other authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Hao Wei and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
General procedures with visual guides, Supplementary discussion on reaction optimization, Tables 1–22, Figs. 1 and 2, Schemes 1 and 2, Discussion on limitations, and Characterization data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Massaro, L., Neigenfind, P., Feng, A. et al. Triply convergent Ni-electrocatalytic assembly of 1,1-diaryl cyclobutanes, azetidines and oxetanes. Nat. Chem. 18, 326–334 (2026). https://doi.org/10.1038/s41557-025-01990-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-025-01990-x