Abstract
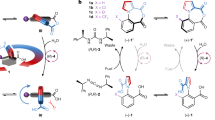
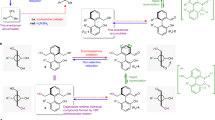
The structural anisotropy necessary to distinguish clockwise from counterclockwise motions in motor-molecules continuously rotating about a covalent single bond has previously been supplied by chiral fuelling systems or by enzymes. Here we report a class of rotary motors in which, like motor proteins, structural asymmetry in the motor itself causes directional rotary catalysis. A single stereogenic centre in azaindole–phenylethanoic acid motors is sufficient to produce diastereomeric intermediates of atropisomeric conformations in the catalytic cycle, generating 8:1 clockwise:counterclockwise directional bias in the motor’s rotary catalysis of diisopropylcarbodiimide hydration (motor substituent PhCH2–). One enantiomer of a chiral hydrolysis promoter increases the directionality to 30:1 for clockwise rotation (motor substituent CH3–), while the other enantiomer reverses the direction to 1:2 clockwise:counterclockwise. The experimental demonstration that a chiral molecular motor can be powered by a chemical fuel to rotate either with, or counter to, the motor’s dominant power stroke informs the understanding of how chemical energy is transduced through catalysis, the fundamental process that powers biology.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2338856 (2-IV), 2338857 (S12-I), 2338858 (2-I), 2338859 (S8-I) and 2338860 (S8-IV). These data can be obtained free of charge via www.ccdc.cam.ac.uk/conts/retrieving.html. The data that support the findings of this study are available within the paper and its Supplementary Information, or are available from the Mendeley data repository (https://data.mendeley.com/) via https://doi.org/10.17632/kby9y39k2h.1 (ref. 55).
References
Wilson, M. R. et al. An autonomous chemically fuelled small-molecule motor. Nature 534, 235–240 (2016).
Amano, S., Fielden, S. D. P. & Leigh, D. A. A catalysis-driven artificial molecular pump. Nature 594, 529–534 (2021).
Borsley, S., Kreidt, E., Leigh, D. A. & Roberts, B. M. W. Autonomous fuelled directional rotation about a covalent single bond. Nature 604, 80–85 (2022).
Liu, E. et al. A molecular information ratchet using a cone-shaped macrocycle. Chem 9, 1147–1163 (2023).
Berreur, J. et al. Redox-powered autonomous directional C–C bond rotation under enzyme control. Nature 644, 96–101 (2025).
Gallagher, J. M., Roberts, B. M. W., Borsley, S. & Leigh, D. A. Conformational selection accelerates catalysis by an organocatalytic molecular motor. Chem 10, 855–866 (2024).
Wang, P.-L. et al. Transducing chemical energy through catalysis by an artificial molecular motor. Nature 637, 594–600 (2025).
Liu, H.-K. et al. Structural influence of the chemical fueling system on a catalysis-driven rotary molecular motor. J. Am. Chem. Soc. 147, 8785–8795 (2025).
Wang, P.-L. et al. A catalysis-driven dual molecular motor. J. Am. Chem. Soc. 147, 10690–10697 (2025).
Liu, H.-K. et al. In situ quantification of directional rotation by a catalysis-driven azaindole‑N‑oxide−phenoic acid molecular motor. J. Am. Chem. Soc. 147, 29534–29541 (2025).
Liu, E., Daou, D., Hasenknopf, B., Vives, G. & Sollogoub, M. Not going back: unidirectional movement by intramolecular one-way ratcheting of functionalized cyclodextrin. Chem 11, 102623 (2025).
Schliwa, M. & Woehlke, G. Molecular motors. Nature 422, 759–765 (2003).
Biagini, C. & Di Stefano, S. Abiotic chemical fuels for the operation of molecular machines. Angew. Chem. Int. Ed. 59, 8344–8354 (2020).
Borsley, S., Leigh, D. A. & Roberts, B. M. W. Chemical fuels for molecular machinery. Nat. Chem. 14, 728–738 (2022).
Sangchai, T., Al Shehimy, S., Penocchio, E. & Ragazzon, G. Artificial molecular ratchets: tools enabling endergonic processes. Angew. Chem. Int. Ed. 62, e202309501 (2023).
Borsley, S., Leigh, D. A. & Roberts, B. M. W. Molecular ratchets and kinetic asymmetry: giving chemistry direction. Angew. Chem. Int. Ed. 63, e202400495 (2024).
Kelly, T. R., De Silva, H. & Silva, R. A. Unidirectional rotary motion in a molecular system. Nature 401, 150–152 (1999).
Kelly, T. R. et al. Progress toward a rationally designed, chemically powered rotary molecular motor. J. Am. Chem. Soc. 129, 376–386 (2007).
Lin, Y., Dahl, B. J. & Branchaud, B. P. Net directed 180° aryl-aryl bond rotation in a prototypical achiral biaryl lactone synthetic molecular motor. Tetrahedron Lett. 46, 8359–8362 (2005).
Fletcher, S. P., Dumur, F., Pollard, M. M. & Feringa, B. L. A reversible, unidirectional molecular rotary motor driven by chemical energy. Science 310, 80–82 (2005).
Mondal, A., Toyoda, R., Costil, R. & Feringa, B. L. Chemically driven rotatory molecular machines. Angew. Chem. Int. Ed. 61, e202206631 (2022).
Han, T.-J., Ke, X.-Y., Wang, M.-C., Ni, S.-F. & Mei, G.-J. A chemically powered rotary molecular motor based on reversible oxazepine formation. Angew. Chem. Int. Ed. 64, e202418933 (2025).
Kay, E. R., Leigh, D. A. & Zerbetto, F. Synthetic molecular motors and mechanical machines. Angew. Chem. Int. Ed. 46, 72–191 (2007).
Astumian, R. D. Design principles for Brownian molecular machines: how to swim in molasses and walk in a hurricane. Phys. Chem. Chem. Phys. 9, 5067–5083 (2007).
Abendroth, J. M., Bushuyev, O. S., Weiss, P. S. & Barrett, C. J. Controlling motion at the nanoscale: rise of the molecular machines. ACS Nano 9, 7746–7768 (2015).
Lancia, F., Ryabchun, A. & Katsonis, N. Life-like motion driven by artificial molecular machines. Nat. Rev. Chem. 3, 536–551 (2019).
Amano, S., Borsley, S., Leigh, D. A. & Sun, Z. Chemical engines: driving systems away from equilibrium through catalyst reaction cycles. Nat. Nanotechnol. 16, 1057–1067 (2021).
Amano, S. & Hermans, T. M. Repurposing a catalytic cycle for transient self-assembly. J. Am. Chem. Soc. 146, 23289–23296 (2024).
Astumian, R. D. Kinetic asymmetry allows macromolecular catalysts to drive an information ratchet. Nat. Commun. 10, 3837 (2019).
Serreli, V., Lee, C.-F., Kay, E. R. & Leigh, D. A. A molecular information ratchet. Nature 445, 523–527 (2007).
Astumian, R. D. Kinetic asymmetry and directionality of nonequilibrium molecular systems. Angew. Chem. Int. Ed. 63, e202306569 (2024).
Koumura, N., Zijlstra, R. W. J., van Delden, R. A., Harada, N. & Feringa, B. L. Light-driven monodirectional molecular rotor. Nature 401, 152–155 (1999).
Sheng, J. et al. Formylation boosts the performance of light-driven overcrowded alkene-derived rotary molecular motors. Nat. Chem. 16, 1330–1338 (2024).
Walsh, C. T., Tu, B. P. & Tang, Y. Eight kinetically stable but thermodynamically activated molecules that power cell metabolism. Chem. Rev. 118, 1460–1494 (2018).
Dahl, B. J. & Branchaud, B. P. Synthesis and characterization of a functionalized chiral biaryl capable of exhibiting unidirectional bond rotation. Tetrahedron Lett. 45, 9599–9602 (2004).
Dahl, B. J. & Branchaud, B. P. 180° unidirectional bond rotation in a biaryl lactone artificial molecular motor prototype. Org. Lett. 8, 5841–5844 (2006).
Collins, B. S. L., Kistemaker, J. C. M., Otten, E. & Feringa, B. L. A chemically powered unidirectional rotary molecular motor based on a palladium redox cycle. Nat. Chem. 8, 860–866 (2016).
Zhang, Y. et al. A chemically driven rotary molecular motor based on reversible lactone formation with perfect unidirectionality. Chem 6, 2420–2429 (2020).
Leigh, D. A. Reaction: of myths, misconceptions and motors—a matter of equilibrium. Chem 11, 102745 (2025).
Astumian, R. D. Trajectory and cycle-based thermodynamics and kinetics of molecular machines: the importance of microscopic reversibility. Acc. Chem. Res. 51, 2653–2661 (2018).
Astumian, R. D. Irrelevance of the power stroke for the directionality, stopping force, and optimal efficiency of chemically driven molecular machines. Biophys. J. 108, 291–303 (2015).
Amano, S. et al. Using catalysis to drive chemistry away from equilibrium: relating kinetic asymmetry, power strokes, and the Curtin–Hammett principle in Brownian ratchets. J. Am. Chem. Soc. 144, 20153–20164 (2022).
Penocchio, E., Gu, G., Albaugh, A. & Gingrich, T. R. Power strokes in molecular motors: predictive, irrelevant, or somewhere in between?. J. Am. Chem. Soc. 147, 1063–1073 (2025).
Kariyawasam, L. S. & Hartley, C. S. Dissipative assembly of aqueous carboxylic acid anhydrides fueled by carbodiimides. J. Am. Chem. Soc. 139, 11949–11955 (2017).
Schwarz, P. S., Tena-Solsona, M., Daia, K. & Boekhoven, J. Carbodiimide-fueled catalytic reaction cycles to regulate supramolecular processes. Chem. Commun. 58, 1284–1297 (2022).
Binks, L. et al. The role of kinetic asymmetry and power strokes in an information ratchet. Chem 9, 2902–2917 (2023).
Kariyawasam, L. S., Hossain, M. M. & Hartley, C. S. The transient covalent bond in abiotic nonequilibrium systems. Angew. Chem. Int. Ed. 60, 12648–12658 (2021).
Griehl, C., Kolbe, A. & Merkel, S. Quantitative description of epimerization pathways using the carbodiimide method in the synthesis of peptides. J. Chem. Soc. Perkin Trans. 2, 2525–2529 (1996).
Seeman, J. I. Effect of conformational change on reactivity in organic chemistry. Evaluations, applications, and extensions of Curtin–Hammett Winstein–Holness kinetics. Chem. Rev. 83, 83–134 (1983).
Feringa, B. L. The art of building small: from molecular switches to motors (Nobel Lecture). Angew. Chem. Int. Ed. 56, 11060–11078 (2017).
Greb, L. & Lehn, J.-M. Light-driven molecular motors: imines as four-step or two-step unidirectional rotors. J. Am. Chem. Soc. 136, 13114–13117 (2014).
Uhl, E., Mayer, P. & Dube, H. Active and unidirectional acceleration of biaryl rotation by a molecular motor. Angew. Chem. Int. Ed. 59, 5730–5737 (2020).
Richards, C. J. & Arthurs, R. A. Catalyst optimisation for asymmetric synthesis by ligand chirality element addition: a perspective on stereochemical cooperativity. Chem. Eur. J. 23, 11460–11478 (2017).
Xie, M.-S. et al. Rational design of 2-substituted DMAP-N-oxides as acyl transfer catalysts: dynamic kinetic resolution of azlactones. J. Am. Chem. Soc. 142, 19226–19238 (2020).
Liu, H.-K. et al. Chiral catalysis-driven rotary molecular motors. Mendeley Data https://doi.org/10.17632/kby9y39k2h.1 (2026).
Acknowledgements
We thank S. Amano for initial discussions regarding the concept of a chiral rotary motor, and S. Amano, T. W. Mrad and L. Binks for work on early prototypes of chiral catalysis-driven rotary molecular motors. This work was funded by the Engineering and Physical Sciences Research Council (EP/P027067/1, D.A.L.; EP/V007580/1, R.W.A.) and the European Research Council (advanced grant number 786630; D.A.L.). S.B. is a Royal Society University Research Fellow. D.A.L. is a Royal Society Research Professor. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
H.-K.L. performed the synthesis and motor operations. B.M.W.R. and H.-K.L. performed kinetic analysis of the data. R.W.A. obtained deconstructed NMR spectra of the individual species in the catalytic cycle. G.F.S.W. and A.H. carried out the X-ray crystallography. S.B. and D.A.L. directed the research. The manuscript was prepared with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–46, Discussion, Tables 1–12 and Spectra 1–55.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, HK., Roberts, B.M.W., Borsley, S. et al. Chiral catalysis-driven rotary molecular motors. Nat. Chem. (2026). https://doi.org/10.1038/s41557-025-02050-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41557-025-02050-0