Abstract
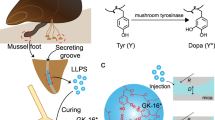
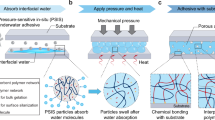
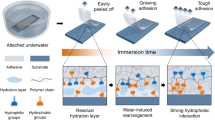
Developing adhesives with robust underwater adhesion to diverse substrates, particularly highly hydrated and lipidic substrates, remains a major challenge. Inspired by the tough adhesion of barnacles on marine creatures, here we report a dilution-resistant and superspreading coacervate to mediate robust underwater adhesion by enabling interfacial infiltration and intertwining. Conjugation of terminal butyl groups to poly(propylene glycol) (PPG) triggers liquid–liquid phase separation of the aqueous solution and formation of PPG coacervate driven by the hydrogen bonding and hydrophobic interactions. The simple coacervate shows distinctive superspreading properties to facilitate interfacial wetting, infiltration and formation of intertwined interfaces on both the hydrated and lipidic substrates and concentrates the loaded hydrophilic curing agents to form a robust underwater adhesive by rapid in situ photocuring. The coacervate-based adhesive demonstrates strong underwater adhesion to diverse substrates, including highly hydrated and lipidic substrates with multiscale porosity and in complex aqueous environments. Potential applications of this adhesive are validated by robust adhesion-mediated hydrogel and/or organogel assemblies and effective sealing of underwater leakages and tissue perforations.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are presented in the paper and Supplementary Information. Source data are provided with this paper.
References
Li, J. et al. Tough adhesives for diverse wet surfaces. Science 357, 378–381 (2017).
Wu, S. J. & Zhao, X. Bioadhesive technology platforms. Chem. Rev. 123, 14084–14118 (2023).
Ahn, B. K. Perspectives on mussel-inspired wet adhesion. J. Am. Chem. Soc. 139, 10166–10171 (2017).
Qin, C. et al. Water-assisted strong underwater adhesion via interfacial water removal and self-adaptive gelation. Proc. Natl Acad. Sci. USA 120, e2301364120 (2023).
Wang, Y. et al. A biorobotic adhesive disc for underwater hitchhiking inspired by the remora suckerfish. Sci. Robot. 2, eaan8072 (2017).
Zhang, L. et al. High-performance organohydrogel artificial muscle with compartmentalized anisotropic actuation under microdomain confinement. Adv. Mater. 35, 2202193 (2023).
Yuk, H. et al. Dry double-sided tape for adhesion of wet tissues and devices. Nature 575, 169–174 (2019).
Maier, G. P., Rapp, M. V., Waite, J. H., Israelachvili, J. N. & Butler, A. Adaptive synergy between catechol and lysine promotes wet adhesion by surface salt displacement. Science 349, 628–632 (2015).
Narayanan, A., Dhinojwala, A. & Joy, A. Design principles for creating synthetic underwater adhesives. Chem. Soc. Rev. 50, 13321–13345 (2021).
Fan, H. & Gong, J. P. Bioinspired underwater adhesives. Adv. Mater. 33, 2102983 (2021).
Yi, B. et al. Surface hydrophobization of hydrogels via interface dynamics-induced network reconfiguration. Nat. Commun. 15, 239 (2024).
Yao, L. et al. Autonomous underwater adhesion driven by water-induced interfacial rearrangement. Nat. Commun. 14, 6563 (2023).
Cui, C. et al. Water-triggered hyperbranched polymer universal adhesives: from strong underwater adhesion to rapid sealing hemostasis. Adv. Mater. 31, 1905761 (2019).
Zhao, Y. et al. Bio-inspired reversible underwater adhesive. Nat. Commun. 8, 2218 (2017).
Zhuo, S. et al. Complex multiphase organohydrogels with programmable mechanics toward adaptive soft-matter machines. Sci. Adv. 6, eaax1464 (2020).
Wan, X. et al. Interfacial instability-induced (3i) adhesives through ‘mediator’ solvent diffusion for robust underoil adhesion. Adv. Mater. 35, 2208413 (2023).
Liu, Y. et al. Synthesis of robust underwater glues from common proteins via unfolding-aggregating strategy. Nat. Commun. 14, 5145 (2023).
Liu, Q., Nian, G., Yang, C., Qu, S. & Suo, Z. Bonding dissimilar polymer networks in various manufacturing processes. Nat. Commun. 9, 846 (2018).
Cintron-Cruz, J. A. et al. Rapid ultratough topological tissue adhesives. Adv. Mater. 34, 2205567 (2022).
Fitzgerald, D. M., Colson, Y. L. & Grinstaff, M. W. Advancing pressure-sensitive adhesives for internal wound closure. Nat. Rev. Mater. 8, 3–5 (2023).
Taboada, G. M. et al. Overcoming the translational barriers of tissue adhesives. Nat. Rev. Mater. 5, 310–329 (2020).
Pal, S. et al. Recyclable surgical, consumer, and industrial adhesives of poly(α-lipoic acid). Science 385, 877–883 (2024).
Harrington, M. J., Mezzenga, R. & Miserez, A. Fluid protein condensates for bio-inspired applications. Nat. Rev. Bioeng. 2, 260–278 (2024).
Zhao, Q. et al. Underwater contact adhesion and microarchitecture in polyelectrolyte complexes actuated by solvent exchange. Nat. Mater. 15, 407 (2016).
Frey, S. T. et al. Octopus-inspired adhesive skins for intelligent and rapidly switchable underwater adhesion. Sci. Adv. 8, eabq1905 (2022).
Guo, Q. et al. Hydrogen-bonds mediate liquid-liquid phase separation of mussel derived adhesive peptides. Nat. Commun. 13, 5771 (2022).
Deepankumar, K. et al. Liquid–liquid phase separation of the green mussel adhesive protein pvfp-5 is regulated by the post-translated dopa amino acid. Adv. Mater. 34, 2103828 (2022).
Liang, C. et al. Biochemistry of barnacle adhesion: an updated review. Front. Mar. Sci. 6, 565 (2019).
Miserez, A., Yu, J. & Mohammadi, P. Protein-based biological materials: molecular design and artificial production. Chem. Rev. 123, 2049–2111 (2023).
Fears, K. P., Orihuela, B., Rittschof, D. & Wahl, K. J. Acorn barnacles secrete phase-separating fluid to clear surfaces ahead of cement deposition. Adv. Sci. 5, 1700762 (2018).
Dompé, M. et al. Thermoresponsive complex coacervate-based underwater adhesive. Adv. Mater. 31, 1808179 (2019).
Sun, J. et al. Genetically engineered polypeptide adhesive coacervates for surgical applications. Angew. Chem. Int. Ed. 60, 23687–23694 (2021).
Hastings, D. E. & Stöver, H. D. H. Exploring the impact of zwitterions in discrete charge arrangements of stimuli-responsive polyelectrolyte complexes. ACS Appl. Polym. Mater. 4, 5035–5046 (2022).
Yewdall, N. A., André, A. A. M., Lu, T. & Spruijt, E. Coacervates as models of membraneless organelles. Curr. Opin. Colloid Interface Sci. 52, 101416 (2021).
Milanović, J., Petrović, L., Sovilj, V. & Katona, J. Complex coacervation in gelatin/sodium caseinate mixtures. Food Hydrocoll. 37, 196–202 (2014).
Owiwe, M. T., Ayyad, A. H. & Takrori, F. M. Surface tension of the oppositely charged sodium poly(styrene sulfonate) /benzyldimethylhexadecylammonium chloride and sodium poly(styrene sulfonate)/polyallylamine hydrochloride mixtures. Colloid Polym. Sci. 298, 1197–1204 (2020).
Wang, S., Liu, K., Yao, X. & Jiang, L. Bioinspired surfaces with superwettability: new insight on theory, design, and applications. Chem. Rev. 115, 8230–8293 (2015).
Cooper, C. B. et al. Autonomous alignment and healing in multilayer soft electronics using immiscible dynamic polymers. Science 380, 935–941 (2023).
Xiao, L. et al. An artificial phase-transitional underwater bioglue with robust and switchable adhesion performance. Angew. Chem. Int. Ed. 60, 12082–12089 (2021).
Vahdati, M., Cedano-Serrano, F. J., Creton, C. & Hourdet, D. Coacervate-based underwater adhesives in physiological conditions. ACS Appl. Polym. Mater. 2, 3397–3410 (2020).
Liang, W. et al. Peritoneum-inspired Janus porous hydrogel with anti-deformation, anti-adhesion and pro-healing characteristics for abdominal wall defect treatment. Adv. Mater. 34, 2108992 (2022).
Yuk, H., Zhang, T., Lin, S., Parada, G. A. & Zhao, X. Tough bonding of hydrogels to diverse non-porous surfaces. Nat. Mater. 15, 190–196 (2016).
Yu, J. et al. Molecular architecture regulation for the design of instant and robust underwater adhesives. Sci. Adv. 9, eadg4031 (2023).
Freedman, B. R. et al. Instant tough adhesion of polymer networks. Proc. Natl Acad. Sci. USA 121, e2304643121 (2024).
Freedman, B. R. et al. Degradable and removable tough adhesive hydrogels. Adv. Mater. 33, 2008553 (2021).
Zhang, J. et al. Wetting ridge assisted programmed magnetic actuation of droplets on ferrofluid-infused surface. Nat. Commun. 12, 7136 (2021).
Daerr, A. & Mogne, A. Pendent_Drop: an ImageJ plugin to measure the surface tension from an image of a pendent drop. J. Open Res. Softw. 4, e3 (2016).
Zhao, Z., Zhang, K., Liu, Y., Zhou, J. & Liu, M. Highly stretchable, shape memory organohydrogels using phase-transition microinclusions. Adv. Mater. 29, 1701695 (2017).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant nos 52433010 to L.B.; 52403181 to B.Y. and 52473129 to P.Z.). This work was also supported by the Guangdong Basic and Applied Basic Research Foundation (grant no. 2025A1515012036 to P.Z.) and the GJYC program of Guangzhou (grant no. 2024D03J0004 to L.B.). This work was also supported by the Fundamental Research Funds for the Central Universities (grant no. 2025ZYGXZR015 to L.B.) and the Open Foundation of Hubei Key Laboratory of Regenerative Medicine and Multi-disciplinary Translational Research (grant no. 2025zsyx01 to P.Z.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the article.
Author information
Authors and Affiliations
Contributions
B.Y. and L.B. conceived the idea. B.Y. conducted the material preparation and characterizations and data analysis. H. Li conducted the cytocompatibility and in vivo studies. S.C., H. Lian, F.T. and H.Y. helped with the data acquisition. X.X., K.Z. and P.Z. helped on the data analysis and discussion. B.Y. wrote the paper. L.B., P.Z., K.Z., B.Y. and H. Li revised the paper. L.B., P.Z. and K.Z. supervised the entire research. All authors approved the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Uttam Manna, Adrivit Mukherjee, Qi Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Notes 1–15, Figs. 1–42, Tables 1–6 and Refs.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yi, B., Li, H., Chen, S. et al. Superspreading and ultra-infiltrative coacervate mediates strong underwater adhesion on hydrated and lipidic substrates. Nat. Chem. (2026). https://doi.org/10.1038/s41557-026-02087-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41557-026-02087-9