Abstract
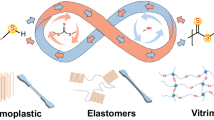
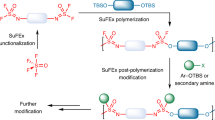
Sulfur–sulfur bonds are ubiquitous across broad classes of natural products, peptides and proteins, drug molecules, and synthetic polymers and materials. The ability to make and break these bonds in a controlled manner is critical for their many scientific and technological applications. Here we report the discovery of an unusual S–S metathesis reaction of organic trisulfides. When exposed to certain polar aprotic solvents, trisulfides were found to undergo spontaneous metathesis, with the reaction equilibrium established in seconds in some cases. No exogenous reagents, heat, light or other stimuli were required to provoke this reaction. Furthermore, the trisulfide metathesis process can occur both inter- and intramolecularly. Understanding the scope and mechanism of this reaction enabled diverse applications of this chemistry in dynamic combinatorial library synthesis, the covalent modification of complex natural products, and S–S metathesis polymerization and depolymerization as a platform for chemically recyclable plastics.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers CCDC 2420468 (Bn2S3), 2479704 (Ph2S2) and 2486578 (18-membered ring). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures. Source data are provided with this paper.
References
Steudel, R. The chemistry of organic polysulfanes R−Sn−R (n > 2). Chem. Rev. 102, 3905–3946 (2002).
Jiang, C.-S., Müller, W. E. G., Schröder, H. C. & Guo, Y.-W. Disulfide- and multisulfide-containing metabolites from marine organisms. Chem. Rev. 112, 2179–2207 (2012).
Wedemeyer, W. J., Welker, E., Narayan, M. & Scheraga, H. A. Disulfide bonds and protein folding. Biochemistry 39, 4207–4216 (2000).
Góngora-Benítez, M., Tulla-Puche, J. & Albericio, F. Multifaceted roles of disulfide bonds. Peptides as therapeutics. Chem. Rev. 114, 901–926 (2014).
Zein, N., Sinha, A. M., McGahren, W. J. & Ellestad, G. A. Calicheamicin γ1I: an antitumor antibiotic that cleaves double-stranded DNA site specifically. Science 240, 1198–1201 (1988).
Zhang, R., Nie, T., Fang, Y., Huang, H. & Wu, J. Poly(disulfide)s: from synthesis to drug delivery. Biomacromolecules 23, 1–19 (2022).
Lee, T., Dirlam, P. T., Njardarson, J. T., Glass, R. S. & Pyun, J. Polymerizations with elemental sulfur: from petroleum refining to polymeric materials. J. Am. Chem. Soc. 144, 5–22 (2022).
Canadell, J., Goossens, H. & Klumperman, B. Self-healing materials based on disulfide links. Macromolecules 44, 2536–2541 (2011).
Houk, J. & Whitesides, G. M. Structure-reactivity relations for thiol-disulfide interchange. J. Am. Chem. Soc. 109, 6825–6836 (1987).
Sarma, R. J., Otto, S. & Nitschke, J. R. Disulfides, imines, and metal coordination within a single system: interplay between three dynamic equilibria. Chem. Eur. J. 13, 9542–9546 (2007).
Caraballo, R., Rahm, M., Vongvilai, P., Brinck, T. & Ramström, O. Phosphine-catalyzed disulfide metathesis. Chem. Commun. 6603-6605 (2008).
Alcock, L. J., Perkins, M. V. & Chalker, J. M. Chemical methods for mapping cysteine oxidation. Chem. Soc. Rev. 47, 231–268 (2018).
Sattler, L. E., Otten, C. J. & Hilt, G. Alternating current electrolysis for the electrocatalytic synthesis of mixed disulfide via sulfur–sulfur bond metathesis towards dynamic disulfide libraries. Chem. Eur. J. 26, 3129–3136 (2020).
Otsuka, H., Nagano, S., Kobashi, Y., Maeda, T. & Takahara, A. A dynamic covalent polymer driven by disulfide metathesis under photoirradiation. Chem. Commun. 46, 1150–1152 (2010).
Belenguer, A. M., Friščić, T., Day, G. M. & Sanders, J. K. M. Solid-state dynamic combinatorial chemistry: reversibility and thermodynamic product selection in covalent mechanosynthesis. Chem. Sci. 2, 696–700 (2011).
Belenguer, A. M., Lampronti, G. I., Wales, D. J. & Sanders, J. K. M. Direct observation of intermediates in a thermodynamically controlled solid-state dynamic covalent reaction. J. Am. Chem. Soc. 136, 16156–16166 (2014).
Sobczak, S. et al. Dynamic covalent chemistry under high-pressure: a new route to disulfide metathesis. Chem. Eur. J. 24, 8769–8773 (2018).
Fritze, U. F. & von Delius, M. Dynamic disulfide metathesis induced by ultrasound. Chem. Commun. 52, 6363–6366 (2016).
Chalker, J. M., Bernardes, G. J. L., Lin, Y. A. & Davis, B. G. Chemical modification of proteins at cysteine: opportunities in chemistry and biology. Chem. Asian J. 4, 630–640 (2009).
Saito, G., Swanson, J. A. & Lee, K.-D. Drug delivery strategy utilizing conjugation via reversible disulfide linkages: role and site of cellular reducing activities. Adv. Drug Deliv. Rev. 55, 199–215 (2003).
Corbett, P. T. et al. Dynamic combinatorial chemistry. Chem. Rev. 106, 3652–3711 (2006).
Yang, Y. & Urban, M. W. Self-healing polymeric materials. Chem. Soc. Rev. 42, 7446–7467 (2013).
Rekondo, A. et al. Catalyst-free room-temperature self-healing elastomers based on aromatic disulfide metathesis. Mater. Horiz. 1, 237–240 (2014).
Block, E. The organosulfur chemistry of the genus Allium—implications for the organic chemistry of sulfur. Angew. Chem., Int. Ed. Engl. 31, 1135–1178 (1992).
Clennan, E. L. & Stensaas, K. L. Recent progress in the synthesis, properties and reactions of trisulfanes and their oxides. Org. Prep. Proced. Int. 30, 551–600 (1998).
Nicolaou, K. C., Smith, A. L. & Yue, E. W. Chemistry and biology of natural and designed enediynes. Proc. Natl Acad. Sci. USA 90, 5881–5888 (1993).
Bertozzi, E. R. Chemistry and technology of elastomeric polysulfide polymers. Rubber Chem. Technol. 41, 114–160 (1968).
Morrison, N. J. & Porter, M. Temperature effects on the stability of intermediates and crosslinks in sulfur vulcanization. Rubber Chem. Technol. 57, 63–85 (1984).
Wu, M. et al. Organotrisulfide: a high capacity cathode material for rechargeable lithium batteries. Angew. Chem. Int. Ed. 55, 10027–10031 (2016).
Trivette, C. D. Jr & Coran, A. Y. Polysulfide exchange reactions. I. Kinetics and mechanism of the thermal exchange between diethyl trisulfide and di-n-propyl trisulfide. J. Org. Chem. 31, 100–104 (1966).
Pickering, T. L., Saunders, K. J. & Tobolsky, A. V. Disproportionation of organic polysulfides. J. Am. Chem. Soc. 89, 2364–2367 (1967).
Tonkin, S. J. et al. Chemically induced repair, adhesion, and recycling of polymers made by inverse vulcanization. Chem. Sci. 11, 5537–5546 (2020).
Yan, P. et al. Stretchable and durable inverse vulcanized polymers with chemical and thermal recycling. Chem. Mater. 34, 1167–1178 (2022).
Lundquist, N. A. et al. Reactive compression molding post-inverse vulcanization: a method to assemble, recycle, and repurpose sulfur polymers and composites. Chem. Eur. J. 26, 10035–10044 (2020).
Kende, I., Pickering, T. L. & Tobolsky, A. V. The dissociation energy of the tetrasulfide linkage. J. Am. Chem. Soc. 87, 5582–5586 (1965).
Chauvin, J.-P. R., Griesser, M. & Pratt, D. A. The antioxidant activity of polysulfides: it’s radical! Chem. Sci. 10, 4999–5010 (2019).
Jabbour, E., Paul, S. & Kantarjian, H. The clinical development of antibody–drug conjugates—lessons from leukaemia. Nat. Rev. Clin. Oncol. 18, 418–433 (2021).
Myers, A. G., Cohen, S. B. & Kwon, B. M. A study of the reaction of calicheamicin γ1 with glutathione in the presence of double-stranded DNA. J. Am. Chem. Soc. 116, 1255–1271 (1994).
Ramström, O. & Lehn, J.-M. Drug discovery by dynamic combinatorial libraries. Nat. Rev. Drug Discov. 1, 26–36 (2002).
Black, S. P., Sanders, J. K. M. & Stefankiewicz, A. R. Disulfide exchange: exposing supramolecular reactivity through dynamic covalent chemistry. Chem. Soc. Rev. 43, 1861–1872 (2014).
Vidal, F. et al. Designing a circular carbon and plastics economy for a sustainable future. Nature 626, 45–57 (2024).
Clark, R. A. & Shaver, M. P. Depolymerization within a circular plastics system. Chem. Rev. 124, 2617–2650 (2024).
Pyun, J. & Norwood, R. A. Infrared plastic optics and photonic devices using chalcogenide hybrid inorganic/organic polymers via inverse vulcanization of elemental sulfur. Prog. Polym. Sci. 156, 101865 (2024).
Baran, T., Duda, A. & Penczek, S. Anionic polymerization of norbornene trisulfide (exo-3,4,5-trithia-tricyclo[5.2.1.02.6]decane). J. Polym. Sci. 22, 1085–1095 (1984).
Baran, T., Duda, A. & Penczek, S. Anionic polymerization of exo-3,4,5-trithiatetracyclo[5.5.1.0.2,608,12]tridec-10-ene (dicyclopentadiene trisulfide). Macromol. Chem. Phys. 185, 2337–2346 (1984).
Pople, J. M. M. et al. Electrochemical synthesis of poly(trisulfides). J. Am. Chem. Soc. 145, 11798–11810 (2023).
Mann, M. et al. Integrated methods for gold leaching and recovery from ore and electronic waste. Nat. Sustain. 8, 947–956 (2025).
Nicholls, T. P. et al. Making and un-making poly(trisulfides) with light: precise regulation of radical concentrations via pulsed LED irradiation. J. Am. Chem. Soc. 147, 46243–46258 (2025).
Chung, W. J. et al. The use of elemental sulfur as an alternative feedstock for polymeric materials. Nat. Chem. 5, 518–524 (2013).
Häußler, M., Eck, M., Rothauer, D. & Mecking, S. Closed-loop recycling of polyethylene-like materials. Nature 590, 423–427 (2021).
Eck, M. & Mecking, S. Closed-loop recyclable and nonpersistent polyethylene-like polyesters. Acc. Chem. Res. 57, 971–980 (2024).
Liu, X. et al. Catalytic closed-loop recycling of polyethylene-like materials produced by acceptorless dehydrogenative polymerization of bio-derived diols. Nat. Chem. 17, 500–506 (2025).
Acknowledgements
We thank the Australian Research Council for funding (DP200100090 to J.M.C., DP230100587 to J.M.C., Z.J. and T.H., DP240100555 to M.L.C., FT220100054 to J.M.C., FT240100330 to W.M.B. and CE230100021 to M.L.C.), generous allocations at the National Facility of the Australian National Computational Infrastructure (M.L.C.) and support from the Flinders University High Impact Collaborative Research Development Fund (M.L.C., J.M.C. and Z.J.). We acknowledge the equipment and technical expertise provided by Flinders Microscopy and Microanalysis (FMMA), Adelaide Microscopy and Microscopy Australia. This research was undertaken in part using the MX1 and MX2 beamlines at the Australian Synchrotron, part of ANSTO, and made use of the Australian Cancer Research Foundation (ACRF) detector. We thank J. Xu, K. Hakobyan, A. Birvé, J. Pople and S. Fraser-Miller for technical assistance. J. Beves is acknowledged for useful suggestions regarding dynamic covalent chemistry. The funders of this research had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
H.D.P. led the metathesis mechanism and substrate scope studies, the calicheamicin modifications, ring-closing and ring-opening metathesis experiments, and S–S metathesis polymerization experiments. A.D.T. contributed to the metathesis mechanism and substrate scope studies, as well as the dynamic combinatorial library synthesis. J.N.S. contributed to the ring-closing and ring-opening metathesis experiments, monomer synthesis and S–S metathesis polymerization experiments. Z.P. contributed to the mechanistic analysis and computational studies. R.S. contributed to the solvent effect studies. S.J.T. and P.Y. discovered the original S–S metathesis phenomenon. S.J.T. carried out mechanical testing of the poly(trisulfide). W.M.B. and M.R.J. contributed to the structural characterization and mechanistic analysis. S.T. and W.M.B. contributed to the X-ray crystallographic analysis, including crystal isolation, data acquisition and refinement. J.R.H. led the EPR studies, supervising H.D.P. and A.D.T. in these experiments. C.T.G. contributed to spectroscopic studies and characterization. M.V.P. and T.H. contributed mechanistic hypotheses, analysis and supervision. M.L.C. led the computational and kinetic studies and mechanistic analysis, and contributed to the polymerization studies and supervision. Z.J. and J.M.C. directed the project and supervised students. The project was conceived by J.M.C., who wrote the paper in close collaboration with Z.J. and M.L.C. All authors contributed to the analysis, critical review and revision of the paper.
Corresponding authors
Ethics declarations
Competing interests
Provisional patent applications have been filed that cover applications of the trisulfide metathesis disclosed in this study, including the dynamic combinatorial libraries, modification of natural products and chemically recyclable polymers (Australia patent application nos. AU2024900381 and AU2025900199). H.D.P., A.D.T., J.N.S., S.J.T., Z.J. and J.M.C. are inventors on these patents. The other authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Courtney Jenkins, Mike Larsen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Discussion, X-ray data and Computational data.
Source data
Source Data Fig. 2 (download ZIP )
GC–MS data and NMR spectra.
Source Data Fig. 3 (download ZIP )
GC–MS data and NMR spectra.
Source Data Fig. 5 (download ZIP )
GC–MS data, LC–MS data and NMR spectra.
Source Data Fig. 6 (download ZIP )
GC–MS data.
Source Data Fig. 7 (download ZIP )
NMR, IR and Raman spectra, and GPC, DSC, TGA, tensile strength and coefficient of friction data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, H.D., Tikoalu, A.D., Smith, J.N. et al. Spontaneous trisulfide metathesis in polar aprotic solvents. Nat. Chem. (2026). https://doi.org/10.1038/s41557-026-02091-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41557-026-02091-z