Abstract
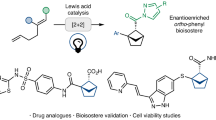
Replacing benzene rings with C(sp3)-rich bioisosteres to yield compounds with improved drug-like properties is an attractive strategy in medicinal chemistry. While many caged hydrocarbons have been validated as bioisosteres of ortho- and meta-disubstituted benzenes, 3D analogues of 1,2,4-trisubstituted benzenes—the second most prevalent benzenoid pattern in drugs—remain elusive because vector fidelity and enantioselective access are still formidable challenges. Here we report a practical route to (enantiomerically pure) 2-thiabicyclo[3.1.1]heptanes (thia-BCHeps) by cycloadditions of bicyclo[1.1.0]butanes with 1,4-dithiane-2,5-diol. This method produces cycloadducts with two and three exit vectors, which serve as promising bioisosteres for ortho-substituted and 1,2,4-trisubstituted benzenes, respectively. Moreover, the cycloadducts can be transformed into a diverse chemical space, including 1,5-disubstituted thiabicyclo[3.1.1]heptenes. Crystallographic analysis and a comparison of the pharmacokinetic properties, along with an evaluation of the biological activity of diflunisal, salicylanilide and the anticancer drug sonidegib, in relation to their 3D thia-BCHep analogues, demonstrate that the thia-BCHeps obtained can provide new surrogates for 1,2,4-trisubstituted, meta- and ortho-substituted benzene rings in drug discovery programmes.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2470121 (for (S)-3k), 2412757 (for (R)-7a), 2412760 (for 24), 2470119 (for S9), 2412872 (for 27) and 2412761 (for 29). Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within the Article and its Supplementary Information, which include synthetic procedures, characterization data, nuclear magnetic resonance spectra, HPLC traces, computational details, biological studies, and evaluations of physicochemical and pharmacokinetic properties.
References
Meanwell, N. A. Improving drug design: an update on recent applications of efficiency metrics, strategies for replacing problematic elements, and compounds in nontraditional drug space. Chem. Res. Toxicol. 29, 564–616 (2016).
Shearer, J., Castro, J. L., Lawson, A. D. G., MacCoss, M. & Taylor, R. D. Rings in clinical trials and drugs: present and future. J. Med. Chem. 65, 8699–8712 (2022).
Ritchie, T. J. & Macdonald, S. J. F. The impact of aromatic ring count on compound developability—are too many aromatic rings a liability in drug design?. Drug Discov. Today 14, 1011–1020 (2009).
Tsien, J., Hu, C., Merchant, R. R. & Qin, T. Three-dimensional saturated C(sp3)-rich bioisosteres for benzene. Nat. Rev. Chem. 8, 605–627 (2024).
Mykhailiuk, P. K. Saturated bioisosteres of benzene: where to go next?. Org. Biomol. Chem. 17, 2839–2849 (2019).
Subbaiah, M. A. M. & Meanwell, N. A. Bioisosteres of the phenyl ring: recent strategic applications in lead optimization and drug design. J. Med. Chem. 64, 14046–14128 (2021).
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Stepan, A. F. et al. Application of the bicyclo[1.1.1]pentane motif as a nonclassical phenyl ring bioisostere in the design of a potent and orally active γ-secretase inhibitor. J. Med. Chem. 55, 3414–3424 (2012).
Pellicciari, R. et al. S)-(+)-2-(3′-Carboxybicyclo[1.1.1]pentyl)-glycine, a structurally new group I metabotropic glutamate receptor antagonist. J. Med. Chem. 39, 2874–2876 (1996).
Dilmaç, A. M., Spuling, E., de Meijere, A. & Bräse, S. Propellanes-from a chemical curiosity to ‘explosive’ materials and natural products. Angew. Chem. Int. Ed. 56, 5684–5718 (2017).
Ma, X. & Pham, L. N. Selective topics in the syntheses of bicyclo[1.1.1]pentane (BCP) analogues. Asian J. Org. Chem. 9, 8–22 (2020).
Shire, B. R. & Anderson, E. A. Conquering the synthesis and functionalization of bicyclo[1.1.1]pentanes. JACS Au 3, 1539–1553 (2023).
Gianatassio, R. et al. Strain-release amination. Science 351, 241–246 (2016).
Ripenko, V. et al. Light-enabled scalable synthesis of bicyclo[1.1.1]pentane halides and their functionalizations. Nat. Synth. 3, 1538–1549 (2024).
Chalmers, B. A. et al. Validating Eaton’s hypothesis: cubane as a benzene bioisostere. Angew. Chem. Int. Ed. 55, 3580–3585 (2016).
Reekie, T. A., Williams, C. M., Rendina, L. M. & Kassiou, M. Cubanes in medicinal chemistry. J. Med. Chem. 62, 1078–1095 (2019).
Auberson, Y. P. et al. Improving nonspecific binding and solubility: bicycloalkyl groups and cubanes as para-phenyl bioisosteres. ChemMedChem 12, 590–598 (2017).
Levterov, V. V. et al. 2-Oxabicyclo[2.2.2]octane as a new bioisostere of the phenyl ring. Nat. Commun. 14, 5608 (2023).
Zhao, J.-X. et al. 1,2-Difunctionalized bicyclo[1.1.1]pentanes: long-sought-after mimetics for ortho/meta-substituted aenes. Proc. Natl Acad. Sci. USA 118, e2108881118 (2021).
Garry, O. L. et al. Rapid access to 2-substituted bicyclo[1.1.1]pentanes. J. Am. Chem. Soc. 145, 3092–3100 (2023).
Denisenko, A. et al. 2-Oxabicyclo[2.1.1]hexanes as saturated bioisosteres of the ortho-substituted phenyl ring. Nat. Chem. 15, 1155–1163 (2023).
Denisenko, A., Garbuz, P., Shishkina, S. V., Voloshchuk, N. M. & Mykhailiuk, P. K. Saturated bioisosteres of ortho-substituted benzenes. Angew. Chem. Int. Ed. 59, 20515–20521 (2020).
Agasti, S. et al. A catalytic alkene insertion approach to bicyclo[2.1.1]hexane bioisosteres. Nat. Chem. 15, 535–541 (2023).
Levterov, V. V. et al. 2-Oxabicyclo[2.1.1]hexanes: synthesis, properties, and validation as bioisosteres of ortho- and meta-benzenes. Angew. Chem. Int. Ed. 63, e202319831 (2024).
Levterov, V. V., Panasyuk, Y., Pivnytska, V. O. & Mykhailiuk, P. K. Water-soluble non-classical benzene mimetics. Angew. Chem. Int. Ed. 59, 7161–7167 (2020).
Frank, N. et al. Synthesis of meta-substituted arene bioisosteres from [3.1.1]propellane. Nature 611, 721–726 (2022).
Iida, T. et al. Practical and facile access to bicyclo[3.1.1]heptanes: potent bioisosteres of meta-substituted benzenes. J. Am. Chem. Soc. 144, 21848–21852 (2022).
Wiesenfeldt, M. P. et al. General access to cubanes as benzene bioisosteres. Nature 618, 513–518 (2023).
Smith, E. et al. Silver(I)-catalyzed synthesis of cuneanes from cubanes and their investigation as isosteres. J. Am. Chem. Soc. 145, 16365–16373 (2023).
Son, J.-Y. et al. Exploring cuneanes as potential benzene isosteres and energetic materials: scope and mechanistic investigations into regioselective rearrangements from cubanes. J. Am. Chem. Soc. 145, 16355–16364 (2023).
Fujiwara, K. et al. Biological evaluation of isosteric applicability of 1,3-substituted cuneanes as m-substituted benzenes enabled by selective isomerization of 1,4-substituted cubanes. Chem. Eur. J. 30, e202303548 (2023).
Epplin, R. C. et al. 2]-Ladderanes as isosteres for meta-substituted aromatic rings and rigidified cyclohexanes. Nat. Commun. 13, 6056 (2022).
Zhang, M. et al. Catalytic asymmetric synthesis of meta benzene isosteres. Nature 633, 90–95 (2024).
Yang, Y. et al. An intramolecular coupling approach to alkyl bioisosteres for the synthesis of multisubstituted bicycloalkyl boronates. Nat. Chem. 13, 950–955 (2021).
Yang, Y. et al. Programmable late-stage functionalization of bridge-substituted bicyclo[1.1.1]pentane bis-boronates. Nat. Chem. 16, 285–293 (2024).
Bychek, R. & Mykhailiuk, P. K. A practical and scalable approach to fluoro-substituted bicyclo[1.1.1]pentanes. Angew. Chem. Int. Ed. 61, e202205103 (2022).
Reinhold, M. et al. Synthesis of polysubstituted bicyclo[2.1.1]hexanes enabling access to new chemical space. Chem. Sci. 14, 9885–9891 (2023).
Harmata, A. S., Spiller, T. E., Sowden, M. J. & Stephenson, C. R. J. Photochemical formal (4+2)-cycloaddition of imine-substituted bicyclo[1.1.1]pentanes and alkenes. J. Am. Chem. Soc. 143, 21223–21228 (2021).
Li, Y.-J. et al. Catalytic intermolecular asymmetric [2π + 2σ] cycloadditions of bicyclo[1.1.0]butanes: practical synthesis of enantioenriched highly substituted bicyclo[2.1.1]hexanes. J. Am. Chem. Soc. 146, 34427–34441 (2024).
Nilova, A., Campeau, L.-C., Sherer, E. C. & Stuart, D. R. Analysis of benzenoid substitution patterns in small molecule active pharmaceutical ingredients. J. Med. Chem. 63, 13389–13396 (2020).
Zhang, Y. et al. Photochemical intermolecular [3σ+2σ]-cycloaddition for the construction of aminobicyclo[3.1.1]heptanes. J. Am. Chem. Soc. 144, 23685–23690 (2022).
Yu, T. et al. Selective [2σ+2σ] cycloaddition enabled by boronyl radical catalysis: synthesis of highly substituted bicyclo[3.1.1]heptanes. J. Am. Chem. Soc. 145, 4304–4310 (2023).
Nguyen, T. V. T., Bossonnet, A., Wodrich, M. D. & Waser, J. Photocatalyzed [2σ+2σ] and [2σ+2π] cycloadditions for the synthesis of bicyclo[3.1.1]heptanes and 5- or 6-membered carbocycles. J. Am. Chem. Soc. 145, 25411–25421 (2023).
Lin, Z. et al. Synthesis of azabicyclo[3.1.1]heptenes enabled by catalyst-controlled annulations of bicyclo[1.1.0]butanes with vinyl azides. J. Am. Chem. Soc. 146, 18565–18575 (2024).
Liu, Y. et al. Pyridine-boryl radical-catalyzed [3π+2σ] cycloaddition for the synthesis of pyridine isosteres. Chem 10, 1–10 (2024).
Zhou, J.-L. et al. Palladium-catalyzed ligand-controlled switchable hetero-(5+3)/enantioselective [2σ+2σ] cycloadditions of bicyclobutanes with vinyl oxiranes. J. Am. Chem. Soc. 146, 19621–19628 (2024).
Wang, X., Gao, R. & Li, X. Catalytic asymmetric construction of chiral polysubstituted 3‑azabicyclo[3.1.1]heptanes by copper-catalyzed stereoselective formal [4π+2σ] cycloaddition. J. Am. Chem. Soc. 146, 21069–21077 (2024).
Dutta, S. et al. Formal [2σ+2σ]-cycloaddition of aziridines with bicyclo[1.1.0]butanes: access to enantiopure 2‑azabicyclo[3.1.1]heptane derivatives. J. Am. Chem. Soc. 146, 27204–27212 (2024).
Zhang, J. et al. Eu(OTf)3-catalyzed formal dipolar [4π+2σ] cycloaddition of bicyclo[1.1.0]butanes with nitrones: access to polysubstituted 2-oxa-3-azabicyclo[3.1.1]heptanes. Angew. Chem. Int. Ed. 63, e202318476 (2024).
Wu, W.-B. et al. Enantioselective formal (3+3) cycloaddition of bicyclobutanes with nitrones enabled by asymmetric Lewis acid catalysis. Nat. Commun. 15, 8005 (2024).
Zhang, X.-G. et al. Copper-catalyzed enantioselective [4π+2σ] cycloaddition of bicyclobutanes with nitrones. J. Am. Chem. Soc. 146, 27274–27281 (2024).
George, M. et al. Double strain-release (3+3)-cycloaddition: Lewis Acid catalyzed reaction of bicyclobutane carboxylates and aziridines. Chem. Eur. J. 31, e202404099 (2024).
Chintawar, C.-C. et al. Photoredox-catalysed amidyl radical insertion to bicyclo[1.1.0]butanes. Nat. Catal. 7, 1232–1242 (2024).
Golfmann, M. & Walker, J. C. L. Bicyclobutanes as unusual building blocks for complexity generation in organic synthesis. Commun. Chem. 6, 9 (2023).
Sujansky, S. J. & Ma, X. Reaction paradigms that leverage cycloaddition and ring strain to construction bicyclic aryl bioisosteres from bicyclo[1.1.0]butanes. Asian J. Org. Chem. 13, e202400045 (2024).
Kleinmans, R. et al. Intermolecular [2π+2σ]-photocycloaddition enabled by triplet energy transfer. Nature 605, 477–482 (2022).
Guo, R. et al. Strain-release [2π+2σ] cycloadditions for the synthesis of bicyclo[2.1.1]hexanes initiated by energy transfer. J. Am. Chem. Soc. 144, 7988–7994 (2022).
Wang, H. et al. Dearomative ring expansion of thiophenes by bicyclobutane insertion. Science 381, 75–81 (2023).
Dhake, K. et al. Beyond bioisosteres: divergent synthesis of azabicyclohexanes and cyclobutenyl amines from bicyclobutanes. Angew. Chem. Int. Ed. 61, e202204719 (2022).
Wipf, P. et al. Pericyclic cascade reactions of (bicyclo[1.1.0]butylmethyl)amines. Angew. Chem. Int. Ed. 45, 4172–4175 (2006).
Ni, D. et al. Intermolecular formal cycloaddition of indoles with bicyclo[1.1.0]butanes by Lewis acid catalysis. Angew. Chem. Int. Ed. 62, e202308606 (2023).
Yang, L. et al. B(C6F5)3‑catalyzed formal (n + 3) (n = 5 and 6) cycloaddition of bicyclo[1.1.0]butanes to medium bicyclo[n.1.1]alkanes. Org. Lett. 26, 4104–4110 (2024).
Hu, Q.-Q. et al. Lewis acid catalyzed cycloaddition of bicyclobutanes with ynamides for the synthesis of polysubstituted 2-aminobicyclo[2.1.1]hexenes. Angew. Chem. Int. Ed. 63, e202405781 (2024).
Liu, Y. et al. Titanium catalyzed [2σ+2π] cycloaddition of bicyclo[1.1.0]-butanes with 1,3-dienes for efficient synthesis of stilbene bioisosteres. Nat. Commun. 15, 4374 (2024).
de Robichon, M. et al. Enantioselective, intermolecular [π2+σ2] photocycloaddition reactions of 2(1H)-quinolones and bicyclo[1.1.0]butanes. J. Am. Chem. Soc. 145, 24466–24470 (2023).
Fu, Q. et al. Enantioselective [2π+2σ] cycloadditions of bicyclo[1.1.0]butanes with vinylazaarenes through asymmetric photoredox catalysis. J. Am. Chem. Soc. 146, 8372–8380 (2024).
Jeong, J. et al. Divergent enantioselective access to diverse chiral compounds from bicyclo[1.1.0]butanes and α,β-unsaturated ketones under catalyst control. J. Am. Chem. Soc. 146, 27830–27842 (2024).
Reissig, H.-U. & Zimmer, R. Donor-acceptor-substituted cyclopropane derivatives and their application in organic synthesis. Chem. Rev. 103, 1151–1196 (2003).
Schneider, T. F. et al. A new golden age for donor–acceptor cyclopropanes. Angew. Chem. Int. Ed. 53, 5504–5523 (2014).
Oku, A., Abe, M. & Iwamoto, M. Electron transfer profile of cyclopropanone acetals in the nonirradiated reaction with tetracyanoethylene, chloranil, and dicy anodichlorobenzoquinone. J. Org. Chem. 59, 7445–7452 (1994).
Scott, K. A. & Njardarson, J. T. Analysis of US FDA‑approved drugs containing sulfur atoms. Top Curr. Chem (Z). 376, 5 (2018).
Mustafa, M. & Winum, J.-Y. The importance of sulfur-containing motifs in drug design and discovery. Expert Opin. Drug Dis. 17, 501–512 (2022).
Feng, M. et al. Sulfur containing scaffolds in drugs: synthesis and application in medicinal chemistry. Curr. Top. Med. Chem. 16, 1200–1216 (2016).
Pathania, S. et al. Role of sulphur-heterocycles in medicinal chemistry: An update. Eur. J. Med. Chem. 180, 486–508 (2019).
Regueiro-Ren, A. Cyclic sulfoxides and sulfones in drug design. Adv. Heterocycl. Chem. 134, 1–30 (2021).
Beno, B. R. et al. A survey of the role of noncovalent sulfur interactions in drug design. J. Med. Chem. 58, 4383–4438 (2015).
Dibchak, D. et al. General synthesis of 3-azabicyclo[3.1.1]heptanes and evaluation of their properties as saturated isosteres. Angew. Chem. Int. Ed. 62, e202304246 (2023).
Wang, M. & Jiang, X. Prospects and challenges in organosulfur chemistry. ACS Sustain. Chem. Eng. 10, 671–677 (2022).
Kosuge, S., Hamanaka, N. & Hayashi, M. Synthesis of thromboxane A2 analog DL-(9,11), (11,12)-dideoxa-(9,11)-methylene-(11,12)-epithio-thromboxane A2 methyl ester. Tetrahedron Lett. 22, 1345–1348 (1981).
Garrido-García, P. et al. Enantioselective photocatalytic synthesis of bicyclo[2.1.1]hexanes as ortho-disubstituted benzene bioisosteres with improved biological activity. Nat. Chem. 17, 734–745 (2025).
Pitzer, L., Schäfers, F. & Glorius, F. Rapid assessment of the reaction-condition-based sensitivity of chemical transformations. Angew. Chem. Int. Ed. 58, 8572–8576 (2019).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Legault, C. Y. CYLview, 1.0b (Université de Sherbrooke, 2009).
Wu, F. et al. Zinc-catalyzed enantioselective formal (3+2) cycloadditions of bicyclobutanes with imines: catalytic asymmetric synthesis of azabicyclo[2.1.1]hexanes. Angew. Chem. Int. Ed. 63, e202406548 (2024).
Bickelhaupt, F. M. & Houk, K. N. Analyzing reaction rates with the distortion/interaction-activation strain model. Angew. Chem. Int. Ed. 56, 10070–10086 (2017).
Lee, C.-C. et al. Discovery of 5-(2′,4′-difluorophenyl)-salicylanilides as new inhibitors of receptor activator of NF-κB ligand (RANKL)-induced osteoclastogenesis. Eur. J. Med. Chem. 98, 115–126 (2015).
Li, Q.-R. et al. Novel-smoothened inhibitors for therapeutic targeting of naïve and drug-resistant hedgehog pathway-driven cancers. Acta Pharmacol. Sin. 40, 257–267 (2019).
Yang, L., Xie, G., Fan, Q. & Xie, J. Activation of the hedgehog-signaling pathway in human cancer and the clinical implications. Oncogene 29, 469–481 (2010).
Wang, F., Stappenbeck, F. & Parhami, F. Inhibition of hedgehog signaling in fibroblasts, pancreatic, and lung tumor cells by oxy186, an oxysterol analogue with drug-like properties. Cells 8, 509 (2019).
Ma, C. et al. Molecular mechanisms involving the Sonic Hedgehog pathway in lung cancer therapy: recent advances. Front. Oncol. 12, 729088 (2022).
Della Corte, C. M. et al. SMO Gene amplification and activation of the hedgehog pathway as novel mechanisms of resistance to anti-epidermal growth factor receptor drugs in human lung cancer. Clin. Cancer Res. 21, 4686–4697 (2015).
Kenny, P. W. & Montanari, C. A. Inflation of correlation in the pursuit of drug-likeness. J. Comput. Aided Mol. Des. 27, 1–13 (2013).
Acknowledgements
We are grateful for financial support from the National Natural Science Foundation of China (grant nos. 22471068 to J.-J.F. and 82371812 to W.Z.), Guangdong Provincial Key Laboratory of Chiral Molecule and Drug Discovery (grant no. 2023B1212060022 to Y.Q.) and Key-Area Research and Development Program of Guangdong Province (grant no. 2022B1111050003 to Y.Q.). We acknowledge the robotic AI-Scientist platform of Chinese Academy of Sciences for its assistance with the theoretical calculations.
Author information
Authors and Affiliations
Contributions
J.-J.F. conceived and directed the project. F.W., J.-J.W. and Y.X. conducted the synthetic investigation. Q.P. and Y.-J.L. helped with the collection of some new compounds and data analysis. K.P. and Y.Q. conducted the osteoclast-inhibitory and antitumour activity studies. Q.H. and W.Z. conducted the COX-2 inhibitory activity assays, preliminary safety evaluations and molecular docking analyses. M.W. and G.W. performed the density functional theory calculations. J.-J.F., Y.Q., W.Z. and G.W. wrote the paper. All authors discussed the results and commented on the manuscript. F.W., J.-J.W. and Y.X. contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare the following competing financial interests: J.-J.F. and J.-J.W. (Hunan University) are listed as inventors on Chinese patent application number CN202411347274.8, which covers the synthesis of ‘sp3-analogues of the antiresorptive agent 40 via (3 + 3) cycloadditions of bicyclobutanes with 1,4-dithiane-2,5-diol and medical uses thereof’ reported in this Article. The other authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Synthetic procedures, characterization data, nuclear magnetic resonance (NMR) spectra, HPLC traces, computational details, X-ray crystallographic data, determination of aqueous solubility, determination of lipophilicity (logD), determination of metabolic stability and biological studies, Supplementary Tables 1–21 and Supplementary Figs. 1–19.
Supplementary Data 1 (download ZIP )
Contains the Cartesian coordinates and energetic details used to generate potential-energy surfaces and dihedral-torsion energy profiles. A README.txt file with detailed descriptions of the folder contents is included in the zipped archive.
Supplementary Data 2 (download XLSX )
Physicochemical and pharmacokinetic property data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, F., Wang, JJ., Xiao, Y. et al. Collective synthesis of 1,2,4-trisubstituted, meta- and ortho-substituted arene bioisosteres from bicyclobutanes. Nat. Chem. (2026). https://doi.org/10.1038/s41557-026-02097-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41557-026-02097-7