Abstract
Climate velocity—the speed and direction species must move to track climate change—is often estimated without accounting for vegetation-driven microclimatic variation. Using mechanistic microclimate models parameterized with three-dimensional maps of topography and vegetation structure, here we show that microclimate heterogeneity reduces the magnitude and alters the direction of climate velocity for maximum and minimum temperatures. For understory-dwelling organisms, the magnitude of maximum temperature velocity was halved and generally oriented towards areas with dense vegetation. For canopy-dwelling organisms, the magnitude of maximum temperature velocity was nearly zero, with vectors oriented vertically downward. These results demonstrate that vegetation complexity produces localized microrefugia, enabling short-term persistence of species under warming conditions. Our findings emphasize the need to integrate fine-scale habitat heterogeneity into predictions of climate resilience and highlight the value of structurally complex forests in providing microclimatic refugia.
Similar content being viewed by others
Main
Climate change is causing the redistribution of species globally, with range shifts generally occurring towards higher latitudes and elevations1,2,3. Climate velocity indicates the speed and direction in which suitable climatic conditions for species are shifting locally and estimates the distance per year a species occurring at any given location would have to move to keep pace with climate change4,5,6,7. However, range shifts often lag behind rates of temperature velocity or occur in directions opposing dominant thermal gradients across latitude and elevation, suggesting that species are unable to migrate fast enough to keep pace with the effects of global warming2,8,9,10.
Local climate velocity is calculated as the temporal rate of climate change divided by the spatial gradient of climate change, with climate variables typically extracted from global databases of free-air conditions at relatively coarse spatial scales4,5,11. However, these data overlook the role of climatic buffering by forest canopies, which may reduce velocities and alter their direction by providing local refugia that allow species to persist in increasingly inhospitable landscapes12,13,14,15. A better understanding of local climate velocities that account for microclimate variability is therefore urgently needed to provide insight into the impacts of climate change on range shifts16,17.
Forests are three-dimensional (3D) ecosystems where complex vegetative structures produce microclimatic variability, both horizontally along the forest floor and vertically within the canopy14,18,19. By reducing solar radiation and airflow, vegetation reduces temperature extremes, which can produce highly heterogeneous microclimates beneath structurally complex canopies that influence the distribution of terrestrial and arboreal species14,19,20,21. As the climate warms, species may move along microclimate gradients produced by vegetation to maintain their thermal niche22,23. Building upon previous research addressing free-air climate velocities4,5,11,24, we model microclimates to examine how vegetation impacts the speed and direction of microclimate velocities along forest floors and within the 3D structure of forest canopies at three spatial grains, as climate change may impact species at the scale of a few centimetres or hundreds of metres, depending on organism size25,26.
Tropical forests are threatened by high temperatures, which are contributing to redistribution of species27,28. Our study focused on tropical montane forests in northern Trinidad, where an airborne light detection and ranging (LiDAR) scan allowed us to generate a digital elevation model (DEM), canopy height model (CHM) and map of the vertical distribution of plant area density across the 1,300 km2 mountain range, spanning 900 m in elevation (Supplementary Fig. 1). We integrate these maps with ERA5 macroclimate data in a mechanistic microclimate model to predict maximum temperature of the warmest month and minimum temperature of the coldest month across the land surface (2 m above ground) and within the canopy at 20-m, 100-m and 1-km resolutions within forests for 1960 and 2015 (Fig. 1 and Supplementary Figs. 2 and 3). We then calculate microclimate velocities across spatial scales over the land surface and advance microclimate research by extending the climate velocity algorithm to 3D within the canopy. These velocities represent the distance and direction that a ground-dwelling or arboreal species at any given location would need to move locally to track warming temperatures (Methods).
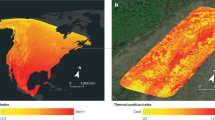
a, Plant area index (PAI) in the northern mountain range of Trinidad derived from airborne LiDAR collected in June 2014. b, Three representations of climate and climate velocity at a 100-m spatial resolution. Free-air climates at 100 m were mechanistically modelled and represent conditions accounting for impacts of topography but not vegetation. Land-surface microclimates represent conditions 2 m above ground and were mechanistically modelled accounting for impacts of topography and vegetation. The 3D within-canopy microclimates were modelled at 5-m intervals from the ground to the top of the canopy. Climate velocity is represented by the red arrows. The length of the arrow represents the speed of climate velocity. For 2D climate velocities, which were calculated using free-air and land-surface climate maps, the angle of the arrow from north (θ) represents the direction of climate velocity. For 3D microclimate velocities calculated from within-canopy climate maps, the angle of the arrow from north represents the horizontal direction of velocity and the angle of the arrow from horizontal represents the vertical direction of velocity. Basemaps in a from Esri (left) and GADm (right; https://gadm.org/data.html).
Here we (1) examine impacts of vegetative buffering on microclimate velocities by comparing their speed to macroclimate velocities at 100-m and 1-km resolutions; (2) evaluate the impact of spatial scale on microclimate velocities across the land surface and within the canopy; and (3) determine the impact of vegetative buffering and spatial scale on the direction of 2D and 3D velocities. By decoupling microclimate from macroclimate conditions and/or increasing spatial microclimate heterogeneity, we expect that vegetative buffering will reduce the speed of microclimate velocities and alter their direction.
Local forest structure variation reduces climate velocity
In montane ecosystems, including in the tropics, species are lagging behind rates of macroclimate change8,29. Accounting for impacts of vegetation on microclimates reduced the magnitude of climate velocities for maximum temperatures across spatial scales (Fig. 2, Extended Data Table 1 and Extended Data Fig. 1). These reductions could have arisen from decreases in the temporal rate or increases in the spatial gradient of climate change, recalling that climate velocity is calculated as the temporal rate divided by the spatial gradient. We found that changes in temporal rates were not responsible, as they were similar to, or exceeded, free-air warming rates (Fig. 2 and Supplementary Fig. 4). Given that understories are experiencing new temperatures across the tropics27, it is unsurprising that the temporal rate does not substantially contribute to climate velocity declines.
Climate velocity (m yr−1), the temporal rate of climate change (°C yr−1) and the spatial gradient of climate change (°C m−1) in the northern range of Trinidad calculated for free-air, land-surface and within-canopy climate conditions at 1-km and 100-m spatial resolutions. Free-air velocities represent climatic conditions accounting for impacts of topography but not vegetation, while land-surface and within-canopy velocities represent climatic conditions accounting for impacts of topography and vegetation on microclimate variability. Free-air and land-surface velocities are calculated in 2D and within-canopy velocities are calculated in 3D. Boxplots display median and 25th and 75th percentiles, with upper and lower whiskers corresponding to 1.5× interquartile range (IQR) from the 25th or 75th percentiles. Note that y axes differ between maximum and minimum temperatures and are truncated at the upper end to aid visualization. Sample sizes (n grid cells) used to produce boxplots represent a 15% random sampling of each category. Maximum temperature: nfree-air, 1km = 455; nland-surface, 1km = 371; nwithin-canopy, 1km = 814; nfree-air, 100m = 43,816; nland-surface, 100m = 42,906; nwithin-canopy, 100m = 83,087. Minimum temperature: nfree-air, 1km = 458; nland-surface, 1km = 373; nwithin-canopy, 1km = 814; nfree-air, 100m = 43,815; nland-surface, 100m = 43,105; nwithin-canopy, 100m = 83,083.
Instead, strong increases in the spatial gradient of climate change generated by local variation in canopy structure and therefore buffering capacity were responsible for reducing microclimate velocities30,31 (Fig. 2 and Supplementary Fig. 5). Across the land surface, median maximum temperature velocities were 1.6× slower than free-air velocities at a 1-km resolution and 2× slower than free-air velocities at a 100-m resolution (Fig. 2 and Extended Data Table 1). Over 55 years, these reduced velocities shorten the distance that maximum temperature isotherms shift from 4.2 km to 2.7 km at a 1-km resolution and from 1.1 km to 540 m at a 100-m resolution. Differences were greater between free-air and 3D microclimate velocities as a result of the additional vertical microclimatic heterogeneity. Relative to free-air velocities, median 3D microclimate velocities were 161.3× slower at a 1-km resolution and 52× slower at a 100-m resolution. These declines translated into shifts of only 15 m and 11 m over 55 years. Similar patterns were observed for minimum temperatures (Fig. 2 and Extended Data Fig. 2). Increases in the spatial gradient of climate change reduced land-surface and within-canopy velocities relative to free-air velocities across spatial resolutions, although the temporal rate of microclimate change was also slower than free-air conditions (Extended Data Table 1 and Supplementary Figs. 6 and 7).
Slower microclimate velocities suggest that the ranges of species may not have to shift as quickly as previously thought to keep pace with rates of climate change, because high microclimate heterogeneity produced by variation in vegetation density shortens the distance organisms must move to reach cooler climates. Examining range shifts in the context of free-air velocities may therefore overestimate climatic lags in redistribution. Overestimation may be particularly prevalent in tropical lowland forests, where shallow free-air spatial gradients (low spatial thermal variation) produce high climate velocities4. Accounting for variation in vegetation structure and understory microclimate may improve our understanding of redistribution patterns of species in these regions.
Granularity of climate estimates
Within forests, species respond to climatic conditions at a variety of spatial scales from centimetres to kilometres depending on the size of the organism26,32. We examined the impact of spatial grain on microclimate velocities by comparing velocities calculated at 1-km, 100-m and 20-m resolutions.
Maximum temperature velocities mirrored patterns previously observed for macroclimate velocities, increasing at coarser spatial grains because of the inverse relationship between spatial grain and climatic heterogeneity11,24,32. At fine spatial grains, the capacity to detect variation in the structural complexity of vegetation increases microclimate heterogeneity14. Microclimate velocities at a 20-m resolution across the land surface were therefore low, at a median rate of only 3.4 m yr−1. At coarser spatial grains, microclimate variability declined as pixels were averaged over space, while the temporal rate of climate change was less affected (Fig. 3). Lower spatial gradients of climate change therefore increased microclimate velocities to median rates of 9.8 m yr−1 at a 100-m resolution and 48.4 m yr−1 at a 1-km resolution. However, the impact of spatial grain largely disappeared in 3D, with over 99% of within-canopy velocities under 1 m yr−1, owing to high microclimatic heterogeneity imposed by the vertical thermal gradient (Fig. 3 and Extended Data Table 1). Minimum temperature velocities exhibited a similar pattern (Fig. 3 and Extended Data Table 1).
Microclimate velocity (m yr−1), the temporal rate of climate change (°C yr−1) and the spatial gradient of climate change (°C m−1) in the northern range of Trinidad calculated at 1-km, 100-m and 20-m spatial resolutions within the forest. Microclimate velocity calculations account for impacts of topography and vegetation on climatic variability. Land-surface velocities are calculated in 2D at 2 m above ground and within-canopy velocities are calculated in 3D at 5-m vertical intervals within the upper half of the canopy. Boxplots display median and 25th and 75th percentiles, with upper and lower whiskers corresponding to 1.5× IQR from the 25th or 75th percentiles. Note that y axes differ between maximum and minimum temperatures and are truncated at the upper end to aid visualization. Sample size (n grid cells) used to produce boxplots represent a 15% random sampling of each category. Maximum temperature: nland-surface, 1km = 371; nland-surface, 100m = 42,906; nland-surface, 20m = 1,027,724; nwithin-canopy, 1km = 814; nwithin-canopy, 100m = 83,087; nwithin-canopy, 20m = 1,944,640. Minimum temperature: nland-surface, 1km = 373; nland-surface, 100m = 43,105; nland-surface, 20m = 1,034,544; nwithin-canopy, 1km = 814; nwithin-canopy, 100m = 83,083; nwithin-canopy, 20m = 1,944,548.
The rate at which species are expected to move thus depends on the spatial grain at which they perceive microclimate. Species that respond to microclimate at larger spatial grains will need to shift ranges more quickly to keep pace with climate change, because fine-grained microclimate heterogeneity may not provide thermal refuge. In contrast, species responding to climate conditions at finer spatial scales or in 3D may be able to move shorter distances to remain in suitable climate conditions. For example, heavily shaded understory environments, as well as structural microhabitats, including tree holes and leaf litter, provide cool microclimates that reduce exposure to extreme temperatures33,34. These slower velocities at fine spatial grains may represent opportunities for thermoregulatory behaviour rather than range shifts to reduce exposure to temperature extremes within thermally variable local environments23.
Local forest structure alters climate velocity direction
Traditional views of species range shifts assume movement towards higher latitudes and elevations as species track their preferred thermal niche16. However, empirical evidence challenges this notion2,9,10. For example, a recent study by Rubenstein et al.10 found that only 47% of documented range shifts align with these expectations, which may be attributed to numerous factors, including persistence in local microclimates14 or range shifts along environmental gradients that oppose thermal gradients across latitudes or elevations11,15,35,36. To explore the impact of vegetation on the direction of climate velocity, we examined whether climate velocities were directed upslope or towards areas with denser vegetation using circular correlations37,38 between the angle of climate velocity in the latitude–longitude plane and the angle a species would need to move to reach higher elevations or denser vegetation. We then graphed the distribution of differences between the angle of climate velocity and the direction of higher elevation or denser vegetation to visualize these correlations (Fig. 4).
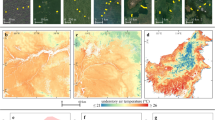
The proportion of grid cells in the northern mountain range of Trinidad with climate velocity directed towards a higher elevation or towards denser vegetation. The x axis represents the angular difference between the direction of maximum or minimum temperature velocity and the direction a species would need to move to reach a higher elevation or denser vegetation. An angular difference of zero indicates that the direction of climate velocity is pointed towards a higher elevation (upslope) or towards denser vegetation. An angular difference of 180 indicates that the direction of climate velocity is pointed downslope or away from denser vegetation. The y axis represents the proportion of grid cells exhibiting a given angular difference. Proportions were calculated on the basis of 15° intervals. Land-surface velocities are 2 m above the ground and within-canopy velocities are 3D velocities in the top half of the forest structure measured from the ground to the canopy. Credit: tree icons, OpenClipart under a Creative Commons license CC0 1.0.
The direction of free-air velocities for maximum and minimum temperatures at 1-km and 100-m spatial resolutions were directed upslope, exhibiting strong positive correlations with the direction needed to reach higher elevations and not with the direction needed to reach denser vegetation (Fig. 4 and Extended Data Table 1). Small differences between the direction of free-air climate velocities and the direction of higher elevations support the pervasive view that species will shift upslope in the tropics39.
At the same spatial resolutions, land-surface velocities for maximum temperatures exhibited positive correlations with both the direction of higher elevation and the direction of denser vegetation (Fig. 4 and Extended Data Table 1). Reducing the spatial resolution to 20 m produced greater variability in velocity directions, but maintained positive correlations with denser vegetation, while exhibiting negative correlations with the direction of higher elevation. Dense vegetation may therefore reverse the direction of range shifts by altering local climate gradients.
When dispersal capacity, biotic interactions or life history traits prevent upslope range shifts at a pace matching that of climate change1,8,9, species may find refuge from increasing maximum temperatures by moving to locally denser forest patches. Forest density is a strong predictor of microclimatic decoupling, reducing diurnal temperature ranges and increasing maximum temperature offsets relative to more sparsely vegetated areas21,40. By stabilizing temperature fluctuations, these vegetatively dense areas may provide refugia from high temperature extremes. However, movement to denser vegetation may not protect populations at warm range edges, which may already be restricted to the coolest microhabitats in the local landscape15. These populations are unable to respond to slow microclimate velocities directed towards dense vegetation and must instead shift upslope to reach cooler environments. Furthermore, populations at warm range edges that are increasingly restricted to denser forest patches may face density declines as the extent of suitable habitat shrinks. Therefore, while dense forest patches may increase short-term persistence in the landscape, populations may face extirpation as the geographic extent of locally suitable microclimates shrink with increasing temperatures. Species responding to fine-scale climate gradients will thus depend on the conservation and restoration of forests with complex vegetative structure where taller and denser patches offset maximum temperatures that may otherwise exceed the narrow critical thermal limits of tropical understory species41,42.
Land-surface velocities for minimum temperatures were also directed upslope at a 100-m resolution, but in contrast to maximum temperatures, exhibited negative correlations with the direction of denser vegetation at 100-m and 20-m resolutions (Extended Data Table 1), because understory thermal minima are generally warmer than macroclimate conditions43,44. Minimum temperatures have strong impacts on species distribution limits at cold range edges, particularly at higher latitudes45. While potentially having a lower impact on range dynamics than maximum temperatures in the tropics, the direction of minimum temperature velocities may be especially applicable to expansion dynamics at cold range edges in temperate and boreal forests, and reflect local reductions in minimum temperature constraints that could broaden microhabitat use for peripheral populations.
Three-dimensional velocities demonstrate refugia for arboreal species
In response to climate change, species may move across multidimensional climate gradients22. In addition to elevational gradients, arboreal species can move across vertical thermal gradients, which can exhibit temperature increases of up to 2.2 °C over just 20 m from the ground to the canopy, compared with temperature changes of just 1.4 °C over 200 m in elevation46. To examine how the spatial dimensionality of climate influences velocities, we calculated the direction of 3D temperature velocities, which represent directions in which arboreal species would need to move to keep pace with climate change. Rather than being directed towards higher elevations or denser vegetation (Fig. 4), over 88% of maximum temperature velocities across spatial scales were directed vertically downward (Extended Data Fig. 3). However, downward shifts in temperature isotherms were not ubiquitous for either maximum or minimum temperatures. Maximum temperature velocities directed vertically upward occurred more frequently in areas with sparser vegetation (Extended Data Fig. 4), where vertical temperature gradients are reversed, such that the understory is warmer than the canopy18 (Extended Data Fig. 5). Furthermore, only 52.4%, 66.9% and 77.8% of minimum temperature velocity vectors exhibited downward movement at 20-m, 100-m and 1-km spatial scales, respectively, owing to weaker vertical gradients in minimum temperatures (Extended Data Fig. 5).
For tropical arboreal species whose ranges will be most impacted by increasing maximum temperatures, slow downward-directed velocities indicate opportunities for organisms to dwell further down forest canopies without the need to migrate over the land surface. Indeed, vertical shifts in habitat use have been documented across short spatial and temporal gradients for arboreal frogs, which shift towards the ground at lower elevations and during the dry season46,47. Whether these thermoregulatory behaviours persist over longer time spans in response to warming climates remains unknown.
However, the full 3D forest environment is not available to all species. Resource distributions, including food and light, limit vertical habitat availability for arboreal plants and animals. For example, low light in the lower canopy may prevent colonization by epiphytes and predator–prey, mutualistic and competitive interactions may prevent vertical reorganization of animal communities despite changing climates48. Furthermore, arboreal species have evolved mobility traits, such as flying and gliding locomotion and adhesive toe pads48, which may compromise their success in lower canopy or terrestrial environments where vegetation structure differs. If species are unable to extend their vertical habitat use, ranges could become vertically compressed into narrower canopy strata49. After reaching the lower limit of suitable vertical habitat, arboreal species would be expected to move in the speed and direction of 2D velocities within the canopy, which exceed velocities across the land surface (Supplementary Methods, Supplementary Table 1 and Supplementary Fig. 8). Although georeferenced occurrence records for numerous taxa are now readily accessible through platforms such as GBIF50, these records rarely contain information about height above ground. Combining our models with empirical data on shifts in vertical habitat use will be critical to evaluate the extent to which arboreal species track 3D climate velocities.
Towards a general understanding of microclimate velocity
Overall, we found that accounting for impacts of vegetation on climate variability reduces velocities and alters their direction. Notably, microclimate velocities revealed an additional dimension to isotherm shifts determined by the density of vegetation and height above ground. In addition to shifting across elevations, maximum temperature velocities were often directed towards denser vegetation or towards the ground, while minimum temperature velocities were often directed towards sparser vegetation at fine spatial resolutions. Species may therefore reduce exposure to warming maximum temperatures by increasing their use of, or becoming restricted to, understory habitats beneath dense vegetation, reflecting the multidimensionality of range shift dynamics that are increasingly recognized as critical for understanding redistribution of species in a changing climate22.
While our study is restricted to temperature velocities within a tropical montane system, the mechanistic nature of the microclimate model and climate velocity calculations allows our conclusions to be generalized to other biomes, such as temperate and boreal forests where minimum temperatures strongly impact cold range limits45. Our approach could also be used to evaluate the velocity of microclimate variables associated with water stress, such as vapour pressure deficit. At macroclimate scales, diverging precipitation and temperature velocity may prevent species from maintaining their historical climatic niche and cause reshuffling of ecological communities11,36. However, at microclimatic scales, the forest understory is both cooler and more humid than macroclimate conditions18,19,21. Moving under dense vegetation to seek refuge from high maximum temperatures would therefore simultaneously reduce hydric stress, preventing substantial mismatches between the direction of microclimate velocity vectors representing thermal and hydric conditions.
The capacity to escape high temperatures by exploiting thermally complex landscapes will be critical for species with limited dispersal capacity and species living in landscapes with homogeneous macroclimate gradients, such as lowland tropical rainforests4,39. However, predicted increases in thermal offsets that provide refuge are contingent upon forests maintaining constant buffering capacity44. Deforestation combined with tree mortality due to increasing disturbances from droughts, wildfires and insect outbreaks are reducing canopy cover globally51,52, yet our models assume constant vegetation cover owing to the lack of repeat LiDAR surveys. Vegetation declines could increase land-surface and within-canopy climate velocities by increasing rates of microclimate warming53 and homogenizing microclimate variability. Forest understory communities would thus be expected to exhibit faster rates of change relative to predictions made assuming constant vegetation structure54. Maintaining and restoring structurally complex forests will therefore be critical to reduce microclimate velocities and provide microclimatic refugia beneath dense vegetation that offer alternative routes to prolonging maintenance of climatic niches under global warming.
Methods
All analyses took place in the northern range of Trinidad, a Caribbean island that lies off the coast of Venezuela, because of the availability of a wall-to-wall LiDAR survey of the island.
Climate grids
We mechanistically modelled maximum temperature of the warmest month and minimum temperature of the coldest month for 1960 and 2015, as climate extremes have a greater impact on species recruitment and survival than climate means55,56,57. The microclimate models were initially produced at a 20-m resolution using the R package microclimf v.0.1.058,59, which uses the physical laws of thermodynamics to connect macroclimate data to local microclimate conditions based on the impacts of topography and vegetation on solar radiation and windspeed and allows approximation of microclimate conditions in regions of the world lacking in situ microclimate sensor networks60. We chose the years 1960 and 2015 because they best represent average temperature during the decades 1951–1960 and 2011–2020 and a 20-m resolution based on a sensitivity analysis to determine a cell size that captured fine-scale variation in vegetation structure while minimizing outliers (Supplementary Methods). These models were produced at 2 m above the ground for land-surface climate estimates and then from 5 m to 40 m above the ground at 5-m intervals (2 m, 5 m, 10 m and so on) to estimate within-canopy conditions (Supplementary Methods). We then coarsened these microclimate models to 100-m and 1-km resolutions by aggregating and averaging grid cells. Regardless of spatial resolution, we refer to these as microclimate models, as they represent climate conditions experienced by terrestrial or arboreal organisms.
We obtained free-air temperatures at a 100-m spatial resolution, by mechanistically modelling climate conditions, accounting for impacts of topography, but not vegetation, using the R package microclima v.0.1.061. We obtained free-air climate conditions at an ~1-km resolution from CHELSA v.2.162,63 to represent a readily accessible and frequently used macroclimate data source. Because CHELSA data were not available for 1960, we estimated the 1960 climate based on offsets between CHELSA and ERA5 data in 1980. We do not model free-air conditions at a finer resolution because the processes mediating the relationship between topography and climate act at scales from hundreds of metres to kilometres64. However, in doing so, we may miss identifying localized thermal extremes in extremely heterogeneous terrain65.
The climate models are based on first principles of energy conservation58. They first apply a topographic correction for adiabatic lapse rate and then estimate microclimate parameters by solving the Penmen–Monteith equation assuming the relationships between sensible heat fluxes and latent heat fluxes remain in balance. Microclimf has been validated against over 400 in situ temperature loggers spanning four continents in different land-cover types, including 70 loggers in tropical rainforests, yielding more accurate predictions than other global climate models (for example, Worldclim and ERA5)27,66.
Model inputs included spatially gridded data describing macroclimate, topography, vegetation structure and characteristics, soil type and habitat type. Gridded climate data were obtained from ERA567,68 using the mcera5 R package69 at an ~25-km resolution and at hourly time intervals for 1960 and 2015. Topography and vegetation layers were derived from discrete return LiDAR data, which were collected in June 2014. The consultant provided classifications for last return ground points and non-ground points, which were then kriged in ArcMap 10.5 to develop a DEM of ground points and a digital surface model of non-ground points at a 1-m resolution. A CHM was developed by subtracting the DEM from the first-return non-ground points. The height of each point above the ground was computed in LAStools using the lasheight function70. The DEM and CHM were then aggregated and averaged to a 20-m resolution. PAI (m2 per m2) and plant area density (PAD; m2 per m3) were calculated at a 20-m horizontal resolution and 1-m vertical resolution based on the Beer–Lambert law for light transmittance through a turbid medium and assuming an extinction coefficient of 0.5 (ref. 71). We estimated PAI and PAD seasonality by modelling monthly fluctuations in MODIS LAI with a generalized additive mixed model and applying a standard offset across all months based on the difference in MODIS LAI and LiDAR PAI (Supplementary Methods).
We mapped soil type according to the US Department of Agriculture soil classification triangle with sand, silt and clay content obtained from the SoilGrids database at a 250-m resolution72,73. We obtained habitat type data from a classification of Trinidadian vegetation74 and reclassified habitat types to those specified in the microclimf R package to estimate other vegetation parameters, including the ratio of vertical to horizontal leaf foliage, maximum stomatal conductance, leaf reflectance, canopy clumsiness and leaf diameter58 (Supplementary Methods).
To model microclimates in the northern range from remotely sensed data, we had to make assumptions that compromised model accuracy. First, the lack of repeat LiDAR surveys required that we assume constant vegetation over time. We also assume that soil and vegetation properties are constant within broad categories and concur with average parameters identified by the model. Furthermore, we note that our models are limited to a small area relative to the global tropics. This is due to computing limitations, as our models at a 20-m resolution include over 2 million climate velocity estimates across the land surface and over 4.5 million estimates in 3D within the canopy spanning the northern mountain range of Trinidad for both maximum and minimum temperatures. However, the mechanistic nature of the climate models and climate velocity calculations allows our conclusions to be generalized to other regions.
Climate velocity
Climate velocity is calculated as the temporal rate of climate change divided by the spatial gradient of climate change4,5
where x is distance, C is the climate variable of interest and t is time. We calculated 2D (land surface) and 3D (within-canopy) microclimate velocities for maximum temperature of the warmest month (°C) and minimum temperature of the coldest month (°C) at three spatial scales—1-km, 100-m and 20-m resolutions. The vertical resolution of all 3D velocities was 5 m. We restrict 3D microclimate velocities to the upper half of the forest as measured from the ground to the canopy, because species occupying the lower canopy can only move a few metres downwards in response to warming. We compared 100-m and 1-km microclimate velocities to 2D free-air velocities at a 100-m resolution (calculated from free-air climate models) and at an ~1-km resolution (calculated from CHELSA climate data).
Climate velocity calculations were conducted in the R programming language75 adapting code from ref. 76 (Supplementary Methods). We calculated the temporal rate of climate change as the slope of temperature change between 1960 and 2015. The spatial gradient of climate change represents the average temperature change (°C m−1) between neighbouring grid cells. For each grid cell, the spatial gradient in 2D is defined on the basis of a 3 × 3 grid around the central cell. For each pair of adjacent cells, the temperature differences are calculated and divided by the distance between cell centres. Differences between cells that neighbour each other to the west and east were averaged to produce the x dimension of the spatial gradient and differences between cells that neighbour each other to the north and south were averaged to produce the y dimension of the spatial gradient. When calculating averages, differences that did not include the focal cell were weighted by \(1/\sqrt{2}\). The 2D spatial gradient for each grid cell, i, is then calculated as \({{\rm{spatial}}\,{\rm{gradient}}}_{i}=\sqrt{{x}_{i}^{2}+{y}_{i}^{2}}\), where xi represents the east–west dimension of the spatial gradient for grid cell i and yi represents the north–south dimension of the spatial gradient for grid cell i (Supplementary Methods).
To calculate the 3D spatial gradient of climate change, we took a similar approach, but made calculations based on adjacent voxels in a 3 × 3 × 3 cube (that is, the central voxel and the six voxels that share a surface with the central one in the cube). We similarly calculated mean temperature differences in the x and y dimensions, but additionally calculated the differences between the central voxel and the voxel below it and the central voxel and the voxel above it. Vertical differences were divided by the height of each voxel (5 m) to obtain the amount (°C m−1) that temperature changes vertically. These vertical differences were averaged to produce the z dimension of the spatial gradient of climate change. The 3D spatial gradient of climate change for each voxel, i, is then calculated as \({{\rm{spatial}}\,{\rm{gradient}}}_{i}=\sqrt{{x}_{i}^{2}+{y}_{i}^{2}+{z}_{i}^{2}}\). For 2D and 3D calculations at a 20-m resolution, we applied an elevational correction to account for the increase in distance that must be travelled if moving parallel to a slope (Supplementary Methods).
To calculate climate velocity (m yr−1), we took the absolute value of the temporal rate of climate change divided by the spatial gradient of climate change. For 3D velocities, we only considered vectors that fell within the canopy, which we defined as falling between 50% and 100% of the relative height of the forest (where relative height is calculated as height of the climate velocity vector divided by canopy height). Additionally, we excluded velocities occurring in non-forested grid cells, as identified by a habitat classification for Trinidad74, as well as those that exceeded the 99th quantile. These high values occur when the spatial gradient of climate change is extremely small and do not accurately represent projected range shifts, particularly when temporal rates of climate change are relatively small.
The direction of climate velocity is the direction of the 2D or 3D vector describing the spatial gradient of climate change. We calculated the direction of climate velocity in the latitude/longitude plane as the angle from north (that is, 0° is north, 180° is south). For 3D velocities, we additionally calculated the vertical angle of movement from horizontal (where horizontal is parallel to the ground). The vertical angle ranges from −90° to 90°, where −90° indicates that the velocity vector is pointed directly towards the ground with no horizontal movement and 90° indicates that the velocity vector is pointed directly upward with no horizontal movement.
To determine whether climate velocities were directed upslope, we calculated the angular difference between the direction opposite to the aspect and the direction of climate velocity. To determine whether climate velocities were directed towards denser vegetation, we calculated the average direction of denser vegetation using the same method that we used to calculate the spatial gradient of climate velocity. We then took the angular difference between the average direction of denser vegetation and the direction of climate velocity. We plotted angular differences using proportional histograms to show the proportion of grid cells where climate velocity is directed towards higher elevations or denser vegetation. An angular difference of 0° indicates that climate velocities are directed upslope or towards denser vegetation and a difference of 180° indicates that climate velocities are directed downslope or towards sparser vegetation. Finally, we calculated the circular correlation between the direction of climate velocity and the direction of higher elevation or denser vegetation37,38. All data and code required for analysis are available via Zenodo at https://zenodo.org/records/17068852 (ref. 77).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Data used to produce microclimate models (except LiDAR data) are publicly available: ERA5 (https://doi.org/10.24381/cds.adbb2d47), MODIS LAI MCD15A3H v.6 (https://doi.org/10.5067/MODIS/MCD15A3H.006), SoilGrids (https://soilgrids.org/), Trinidad land-cover classification (https://doi.org/10.1016/j.foreco.2012.05.016). CHELSA climate data are publicly available (https://chelsa-climate.org/). Microclimate models and climate velocity maps required to reproduce results are available via Zenodo at https://zenodo.org/records/17068852 (ref. 77).
Code availability
Code is available via Zenodo at https://zenodo.org/records/17068852 (ref. 77).
References
Chen, I.-C., Hill, J. K., Ohlemüller, R., Roy, D. B. & Thomas, C. D. Rapid range shifts of species associated with high levels of climate warming. Science 333, 1024–1026 (2011).
Lawlor, J. A. et al. Mechanisms, detection and impacts of species redistributions under climate change. Nat. Rev. Earth Environ. 5, 351–368 (2024).
Parmesan, C. & Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 421, 37–42 (2003).
Loarie, S. R. et al. The velocity of climate change. Nature 462, 1052–1055 (2009).
Burrows, M. T. et al. The pace of shifting climate in marine and terrestrial ecosystems. Science 334, 652–655 (2011).
Burrows, M. T. et al. Geographical limits to species-range shifts are suggested by climate velocity. Nature 507, 492–495 (2014).
Ackerly, D. D. et al. The geography of climate change: Implications for conservation biogeography. Divers. Distrib. 16, 476–487 (2010).
Feeley, K. J. et al. Upslope migration of Andean trees. J. Biogeogr. 38, 783–791 (2011).
Lenoir, J. et al. Species better track climate warming in the oceans than on land. Nat. Ecol. Evol. 4, 1044–1059 (2020).
Rubenstein, M. A. et al. Climate change and the global redistribution of biodiversity: substantial variation in empirical support for expected range shifts. Environ. Evid. 12, 7 (2023).
Dobrowski, S. Z. et al. The climate velocity of the contiguous United States during the 20th century. Glob. Change Biol. 19, 241–251 (2013).
Haesen, S. et al. Microclimate reveals the true thermal niche of forest plant species. Ecol. Lett. 26, 2043–2055 (2023).
Lembrechts, J. J., Nijs, I. & Lenoir, J. Incorporating microclimate into species distribution models. Ecography 42, 1267–1279 (2019).
Lenoir, J., Hattab, T. & Pierre, G. Climatic microrefugia under anthropogenic climate change: implications for species redistribution. Ecography 40, 253–266 (2017).
Maclean, I. M. D. & Early, R. Macroclimate data overestimate range shifts of plants in response to climate change. Nat. Clim. Change 13, 484–490 (2023).
Lenoir, J. & Svenning, J.-C. Climate-related range shifts—a global multidimensional synthesis and new research directions. Ecography 38, 15–28 (2015).
Brito-Morales, I. et al. Climate velocity can inform conservation in a warming world. Trends Ecol. Evol. 33, 441–457 (2018).
Vinod, N. et al. Thermal sensitivity across forest vertical profiles: Patterns, mechanisms, and ecological implications. New Phytol. 237, 22–47 (2023).
De Frenne, P. et al. Forest microclimates and climate change: Importance, drivers and future research agenda. Glob. Change Biol. 27, 2279–2297 (2021).
Hardwick, S. R. et al. The relationship between leaf area index and microclimate in tropical forest and oil palm plantation: forest disturbance drives changes in microclimate. Agric. For. Meteorol. 201, 187–195 (2015).
Jucker, T. et al. Canopy structure and topography jointly constrain the microclimate of human-modified tropical landscapes. Glob. Change Biol. 24, 5243–5258 (2018).
Fredston, A. L. et al. Reimagining species on the move across space and time. Trends Ecol. Evol. 40, 629–638 (2025).
Pinsky, M. L., Comte, L. & Sax, D. F. Unifying climate change biology across realms and taxa. Trends Ecol. Evol. 37, 672–682 (2022).
Heikkinen, R. K. et al. Fine-grained climate velocities reveal vulnerability of protected areas to climate change. Sci. Rep. 10, 1678 (2020).
Pincebourde, S. & Woods, H. A. There is plenty of room at the bottom: microclimates drive insect vulnerability to climate change. Curr. Opin. Insect Sci. 41, 63–70 (2020).
Potter, K. A., Woods, A. H. & Pincebourde, S. Microclimatic challenges in global change biology. Glob. Change Biol. 19, 2932–2939 (2013).
Trew, B. T. et al. Novel temperatures are already widespread beneath the world’s tropical forest canopies. Nat. Clim. Change 14, 753–759 (2024).
Fadrique, B. et al. Widespread but heterogeneous responses of Andean forests to climate change. Nature 564, 207–212 (2018).
Chan, W.-P. et al. Climate velocities and species tracking in global mountain regions. Nature 629, 114–120 (2024).
Ismaeel, A. et al. Patterns of tropical forest understory temperatures. Nat. Commun. 15, 549 (2024).
Menge, J. H., Magdon, P., Wöllauer, S. & Ehbrecht, M. Impacts of forest management on stand and landscape-level microclimate heterogeneity of European beech forests. Landsc. Ecol. 38, 903–917 (2023).
Wiens, J. A. Spatial scaling in ecology. Funct. Ecol. 3, 385–397 (1989).
Pottier, J. et al. The accuracy of plant assemblage prediction from species distribution models varies along environmental gradients. Glob. Ecol. Biogeogr. 22, 52–63 (2013).
Scheffers, B. R., Edwards, D. P., Diesmos, A., Williams, S. E. & Evans, T. A. Microhabitats reduce animal’s exposure to climate extremes. Glob. Change Biol. 20, 495–503 (2014).
Sanczuk, P. et al. Unexpected westward range shifts in European forest plants link to nitrogen deposition. Science 386, 193–198 (2024).
Ordonez, A. & Williams, J. W. Projected climate reshuffling based on multivariate climate-availability, climate-analog, and climate-velocity analyses: implications for community disaggregation. Clim. Change 119, 659–675 (2013).
Jammalamadaka, S. & Sarma, Y. A correlation coefficient for angular variables. Stat. Theory Data Anal. 2, 349–364 (1988).
Tsagris, M. et al. Directional: a collection of functions for directional data analysis. R package version 7.0 (2024).
Colwell, R. K. & Feeley, K. J. Still little evidence of poleward range shifts in the tropics, but lowland biotic attrition may be underway. Biotropica 57, e13358 (2025).
Blonder, B. et al. Extreme and highly heterogeneous microclimates in selectively logged topical forests. Front. For. Glob. Change 1, 5 (2018).
Tewksbury, J. J., Huey, R. B. & Deutsch, C. A. Putting the heat on tropical animals. Science 320, 1296–1297 (2008).
Sunday, J. M. et al. Thermal-safety margins and the necessity of thermoregulatory behavior across latitude and elevation. Proc. Natl Acad. Sci. USA 111, 5610–5615 (2014).
De Frenne, P. et al. Global buffering of temperatures under forest canopies. Nat. Ecol. Evol. 3, 744–749 (2019).
De Lombaerde, E. et al. Maintaining forest cover to enhance temperature buffering under future climate change. Sci. Total Environ. 810, 151338 (2022).
Normand, S. et al. Importance of abiotic stress as a range-limit determinant for European plants: insights from species responses to climatic gradients. Glob. Ecol. Biogeogr. 18, 437–449 (2009).
Scheffers, B. R. et al. Increasing arboreality with altitude: a novel biogeographic dimension. Proc. R. Soc. B. 280, 20131581 (2013).
Basham, E. W. & Scheffers, B. R. Vertical stratification collapses under seasonal shifts in climate. J. Biogeogr. 47, 1888–1898 (2020).
Xing, S. et al. Ecological patterns and processes in the vertical dimension of terrestrial ecosystems. J. Anim. Ecol. 92, 538–551 (2023).
Jorda, G. et al. Ocean warming compresses the three-dimensional habitat of marine life. Nat. Ecol. Evol. 4, 109–114 (2020).
What is GBIF? (GBIF, 2025); https://www.gbif.org/what-is-gbif
Curtis, P. G., Slay, C. M., Harris, N. L., Tyukavina, A. & Hansen, M. C. Classifying drivers of global forest loss. Science 361, 1108–1111 (2018).
Hartmann, H. et al. Climate change risks to global forest health: emergence of unexpected events of elevated tree mortality worldwide. Annu. Rev. Plant Biol. 73, 673–702 (2022).
Zellweger, F. et al. Forest microclimate dynamics drive plant responses to warming. Science 368, 772–775 (2020).
De Frenne, P. et al. Microclimate moderates plant responses to macroclimate warming. Proc. Natl Acad. Sci. USA 110, 18561–18565 (2013).
Inouye, D. W. The ecological and evolutionary significance of frost in the context of climate change. Ecol. Lett. 3, 457–463 (2000).
Murali, G., Iwamura, T., Meiri, S. & Roll, U. Future temperature extremes threaten land vertebrates. Nature 615, 461–467 (2023).
Reyer, C. P. O. et al. A plant’s perspective of extremes: terrestrial plant responses to changing climatic variability. Glob. Change Biol. 19, 75–89 (2013).
Maclean, I. Microclimf: Fast above, below or within canopy gridded microclimate modelling with R. R package version 0.1.0 (2021).
MacLean, I. Microclimf: Fast modelling of microclimate across real landscapes in R. Preprint at EcoEvoRxiv https://doi.org/10.32942/X2BD17 (2025).
Lembrechts, J. J. et al. SoilTemp: a global database of near-surface temperature. Glob. Change Biol. 26, 6616–6629 (2020).
Maclean, I. M. D., Mosedale, J. R. & Bennie, J. J. Microclima: an r package for modelling meso- and microclimate. Methods Ecol. Evol. 10, 280–290 (2019).
Karger, D. N. et al. Climatologies at high resolution for the Earth’s land surface areas. Sci. Data 4, 170122 (2017).
Karger, D. N. et al. Climatologies at high resolution for the Earth’s land surface areas. EnviDat https://doi.org/10.16904/envidat.228.v2.1 (2021).
De Frenne, P. et al. Ten practical guidelines for microclimate research in terrestrial ecosystems. Methods Ecol. Evolution 16, 269–294 (2025).
Maclean, I. M. D., Suggitt, A. J., Wilson, R. J., Duffy, J. P. & Bennie, J. J. Fine-scale climate change: Modelling spatial variation in biologically meaningful rates of warming. Glob. Change Biol. 23, 256–268 (2017).
Klinges, D. H. et al. Proximal microclimate: Moving beyond spatiotemporal resolution improves ecological predictions. Glob. Ecol. Biogeogr. 33, e13884 (2024).
Copernicus Climate Change Service. ERA5 hourly data on single levels from 1940 to present. Climate Data Store (CDS); https://doi.org/10.24381/cds.adbb2d47 (2021).
Hersbach, H. et al. ERA5 hourly data on single levels from 1940 to present. Copernicus Climate Change Service (C3S) Climate Data Store (CDS), https://doi.org/10.24381/cds.adbb2d47 (2023).
Duffy, J. P. Mcera5: Tools to acquire and process ERA5 data for use in microclimate modelling. R package version 0.2.0 (2021).
Isenburg, M. LAStools: Effecient LiDAR processing software; version 210128 (2014).
Milodowski, D. T. et al. The impact of logging on vertical canopy structure across a gradient of tropical forest degradation intensity in Borneo. J. Appl. Ecol. 58, 1764–1775 (2021).
Moeys, J. Soiltexture: Functions for soil texture plot, classification and transformation. R package version 1.5.1 (2018).
Poggio, L. et al. SoilGrids 2.0: producing soil information for the globe with quantified spatial uncertainty. SOIL 7, 217–240 (2021).
Helmer, E. H. et al. Detailed maps of tropical forest types are within reach: forest tree communities for Trinidad and Tobago mapped with multiseason Landsat and multiseason fine-resolution imagery. For. Ecol. Manag. 279, 147–166 (2012).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2021).
García Molinos, J., Schoeman, D. S., Brown, C. J. & Burrows, M. T. VoCC: An R package for calculating the velocity of climate change and related climatic metrics. Methods Ecol. Evol. 10, 2195–2202 (2019).
Soifer, L. Code for microclimates slow and alter the direction of climate velocities in tropical forests. Zenodo https://doi.org/10.5281/zenodo.17068852 (2025).
Acknowledgements
L.G.S. was supported by the W. Thomas Smith Scholarship from Davidson College.
Author information
Authors and Affiliations
Contributions
L.G.S., D.C. and J.B. conceptualized the study and developed the methodology. L.G.S. performed the formal analysis. H.A. provided the raw LiDAR data and developed the elevation and CHMs. L.G.S. wrote the original draft. L.G.S., D.C., J.B. and I.M.D.M. revised and edited the paper. L.G.S. developed the visualizations. D.C. supervised the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Climate Change thanks David Ackerly, Stef Haesen and Sylvain Pincebourde for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Maximum temperature velocity in the northern mountain range of Trinidad.
Climate velocity for maximum temperature of the warmest month in the northern mountain range of Trinidad calculated from free-air, land surface, and within-canopy climates at 1 km, 100 m, and 20 m spatial resolutions. Free-air velocities account for effects of topography but not vegetation on climate variability and are derived from CHELSA climate data (1 km resolution) and mechanistic microclimate models (100 m resolution). Land surface and within-canopy velocities account for effects of topography and vegetation on climate variability and are derived from mechanistic microclimate models. Within canopy velocities represent the average of 3D velocities in the top half of the canopy. Basemaps adapted from the Database of Global Administrative Areas (GADM).
Extended Data Fig. 2 Minimum temperature velocity in the northern mountain range of Trinidad.
Climate velocity for minimum temperature of the coldest month in the northern mountain range of Trinidad calculated from free-air, land surface, and within-canopy climates at 1 km, 100 m, and 20 m spatial resolutions. Free-air velocities account for effects of topography but not vegetation on climate variability and are derived from CHELSA climate data (1 km resolution) and mechanistic microclimate models (100 m resolution). Land surface and within-canopy velocities account for effects of topography and vegetation on climate variability and are derived from mechanistic microclimate models. Within canopy velocities represent the average of 3D velocities in the top half of the canopy. Basemaps adapted from the Database of Global Administrative Areas (GADM).
Extended Data Fig. 3 Ratio of vertical to horizontal movement for 3D within-canopy climate velocity vectors.
The y-axis represents relative height in the canopy, and the x-axis represents the natural log of the vertical distance divided by the horizontal distance of movement for maximum temperature of the warmest month and minimum temperature of the coldest month at 1 km, 100 m, and 20 m spatial resolutions. vertical distance = |z|cos( | θ | |) and horizontal distance = |z|sin( | θ | ); where z = climate velocity and θ = the angle of 3D climate velocity from horizontal, such that 90 represents direct upward movement and −90 represents direct downward movement. Positive values indicate vertical distance greater than horizontal distance and negative values indicate horizontal distance greater than vertical distance. Sample size (grid cells): ntmax,1km = 1811, ntmax,100m = 184638, ntmax,20m = 4321416, ntmin,1km = 1808, ntmin, 100m = 184629, ntmin,20m = 4321075.
Extended Data Fig. 4 Plant area index for 3D velocities directed downward and upward.
Violin plots representing the probability density distribution of plant area index (PAI) for 3D within-canopy climate velocities directed upward toward the canopy and downward toward the understory at 1 km, 100 m, and 20 m spatial resolutions for maximum temperature of the warmest month and minimum temperature of the coldest month. Lines indicate mean PAI. PAI of downward directed maximum temperature velocities was consistently higher than upward directed velocities across spatial grains with statistical significance assessed using two-sided t-tests (1 km: t175.43 = −8.17, p = 6.03 × 10−14, 100 m: t22390 = −86.05, p < 2.2 × 10−16, 20 m: t363815 = −287.17, p < 2.2 × 10−16). PAI of downward directed minimum temperature velocities was lower than upward directed velocities across spatial grains (1 km: t1038.9 = 11.36, p < 2.2 × 10−16, 100 m: t147917 = 69.35, p < 2.2 × 10−16, 20 m: t4305019 = 274.61, p < 2.2 × 10−16).
Extended Data Fig. 5 Vertical microclimate temperature profiles for downward and upward directed climate velocities.
Vertical profiles of maximum temperature of the warmest month and minimum temperature of the coldest month in the northern mountain range of Trinidad in grid cells containing climate velocities directed downward (solid lines) or upward (dashed lines). Temperature profiles represent the mean ± standard deviation at relative height intervals of 0.5, where relative height represents the absolute height divided by the height of the canopy. Sample sizes (n grid cells) vary by spatial resolution, vector direction, and temperature variable. At a 1 km resolution: ntmax,down = 1638, ntmax,up = 173, ntmin,down = 1407, ntmin,up = 401. At a 100 m resolution: ntmax,down = 162810, ntmax,up = 21828, ntmin,down = 123425, ntmin,up = 61204. At a 20 m resolution: ntmax,down = 3979468, ntmax,up = 341948, ntmin,down = 2247257, ntmin,up = 2073818.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Supplementary Table 1 and Supplementary Figs. 1–8.
Source data
Source Data Extended Data Table 1 (download CSV )
Tabular data used to produce Extended Data Table 1.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Soifer, L.G., Ball, J., Asmath, H. et al. Microclimates slow and alter the direction of climate velocities in tropical forests. Nat. Clim. Chang. 16, 95–101 (2026). https://doi.org/10.1038/s41558-025-02496-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41558-025-02496-7