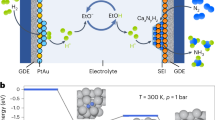
Abstract
Ammonia is a key component of fertilizers, a crucial industrial chemical and a carbon-free fuel. Electrosynthesis of ammonia from nitrogen under ambient conditions presents an attractive alternative to the centralized Haber–Bosch process. Although lithium- and calcium-mediated nitrogen reduction (Li-NRR and Ca-NRR) show promise, long-term continuous ammonia electrosynthesis at high rates will be needed for industrial application. In this Perspective we argue that for Li-NRR and Ca-NRR to operate sustainably, the use of continuous-flow reactors—in which NRR is coupled with the hydrogen oxidation reaction, avoiding non-sustainable proton sources and electrolyte oxidation—is essential. Providing the necessary protons via hydrogen oxidation is vital for the sustainable production of ammonia and long-term system stability. We propose strategies such as optimizing the solid–electrolyte interphase design, refining the electrode and reactor engineering to enhance the system stability and ammonia production rate. We also strongly advocate the exploration of electrocatalytic routes for surpassing the theoretical energy efficiency limit of Li/Ca-NRR.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Source data are provided with this paper.
References
Nielsen, A. (ed.) Ammonia: Catalysis and Manufacture (Springer, 1995)
Choudhary, T. V., Sivadinarayana, C. & Goodman, D. W. Catalytic ammonia decomposition: COx-free hydrogen production for fuel cell applications. Catal. Lett. 72, 197–201 (2001).
Schlögl, R. Catalytic synthesis of ammonia—a “never-ending story”? Angew. Chem. Int. Ed. 42, 2004–2008 (2003).
Haber, F. & Le Rossignol, R. Über die technische Darstellung von Ammoniak aus den Elementen. Z. Elektrochem. Angew. Phys. Chem. 19, 53–72 (1913).
MacFarlane, D. R. et al. A roadmap to the ammonia economy. Joule 4, 1186–1205 (2020).
Iriawan, H. et al. Methods for nitrogen activation by reduction and oxidation. Nat. Rev. Methods Primers 1, 56 (2021).
Choi, J. et al. Identification and elimination of false positives in electrochemical nitrogen reduction studies. Nat. Commun. 11, 5546 (2020).
Andersen, S. Z. et al. A rigorous electrochemical ammonia synthesis protocol with quantitative isotope measurements. Nature 570, 504–508 (2019).
Fu, X. et al. Calcium-mediated nitrogen reduction for electrochemical ammonia synthesis. Nat. Mater. 23, 101–107 (2023).
Fichter, F., Girard, P. & Erlenmeyer, H. Elektrolytische Bindung von komprimiertem Stickstoff bei gewöhnlicher Temperatur. Helv. Chim. Acta 13, 1228–1236 (1930).
Tsuneto, A., Kudo, A. & Sakata, T. Efficient electrochemical reduction of N2 to NH3 catalyzed by lithium. Chem. Lett. 22, 851–854 (1993).
Andersen, S. Z. et al. Increasing stability, efficiency, and fundamental understanding of lithium-mediated electrochemical nitrogen reduction. Energy Environ. Sci. 13, 4291–4300 (2020).
Lazouski, N., Schiffer, Z. J., Williams, K. & Manthiram, K. Understanding continuous lithium-mediated electrochemical nitrogen reduction. Joule 3, 1127–1139 (2019).
Westhead, O., Jervis, R. & Stephens, I. E. L. Is lithium the key for nitrogen electroreduction? Science 372, 1149–1150 (2021).
Krebsz, M. et al. Reduction of dinitrogen to ammonium through a magnesium-based electrochemical process at close-to-ambient temperature. Energy Environ. Sci. 17, 4481–4487 (2024).
Du, H.-L. et al. Electroreduction of nitrogen with almost 100% current-to-ammonia efficiency. Nature 609, 722–727 (2022).
Li, K. et al. Enhancement of lithium-mediated ammonia synthesis by addition of oxygen. Science 374, 1593–1597 (2021).
Suryanto, B. H. R. et al. Nitrogen reduction to ammonia at high efficiency and rates based on a phosphonium proton shuttle. Science 372, 1187–1191 (2021).
Li, K. et al. Increasing current density of Li-mediated ammonia synthesis with high surface area copper electrodes. ACS Energy Lett. 7, 36–41 (2022).
Li, S. et al. Electrosynthesis of ammonia with high selectivity and high rates via engineering of the solid–electrolyte interphase. Joule 6, 2083–2101 (2022).
Lazouski, N. et al. Non-aqueous gas diffusion electrodes for rapid ammonia synthesis from nitrogen and water-splitting-derived hydrogen. Nat. Catal. 3, 463–469 (2020).
Fu, X. et al. Continuous-flow electrosynthesis of ammonia by nitrogen reduction and hydrogen oxidation. Science 379, 707–712 (2023).
Hodgetts, R. Y. et al. Electrocatalytic oxidation of hydrogen as an anode reaction for the Li-mediated N2 reduction to ammonia. ACS Catal. 12, 5231–5246 (2022).
Du, H.-L. et al. The chemistry of proton carriers in high-performance lithium-mediated ammonia electrosynthesis. Energy Environ. Sci. 16, 1082–1090 (2023).
Sažinas, R. et al. Oxygen-enhanced chemical stability of lithium-mediated electrochemical ammonia synthesis. J. Phys. Chem. Lett. 13, 4605–4611 (2022).
Aouissi, A., Al-Deyab, S. S. & Al-Shahri, H. The cationic ring-opening polymerization of tetrahydrofuran with 12-tungstophosphoric acid. Molecules 15, 1398–1407 (2010).
Krishnamurthy, D. et al. Closed-loop electrolyte design for lithium-mediated ammonia synthesis. ACS Cent. Sci. 7, 2073–2082 (2021).
Lazouski, N. et al. Proton donors induce a differential transport effect for selectivity toward ammonia in lithium-mediated nitrogen reduction. ACS Catal. 12, 5197–5208 (2022).
Fu, X. et al. Phenol as proton shuttle and buffer for lithium-mediated ammonia electrosynthesis. Nat. Commun. 15, 2417 (2024).
Li, Y. et al. Extending ring–chain coupling empirical law to lithium-mediated electrochemical ammonia synthesis. Angew. Chem. Int. Ed. 63, e202311413 (2023).
Cai, X. et al. Lithium-mediated ammonia electrosynthesis with ether-based electrolytes. J. Am. Chem. Soc. 145, 25716–25725 (2023).
Li, S. et al. Long-term continuous ammonia electrosynthesis. Nature 629, 92–97 (2024).
Cai, X. et al. Lithium-mediated electrochemical nitrogen reduction: mechanistic insights to enhance performance. iScience 24, 103105 (2021).
Lazouski, N. et al. Cost and performance targets for fully electrochemical ammonia production under flexible operation. ACS Energy Lett. 7, 2627–2633 (2022).
Steinberg, K. et al. Imaging of nitrogen fixation at lithium solid electrolyte interphases via cryo-electron microscopy. Nat. Energy 8, 138–148 (2023).
Singh, A. R. et al. Electrochemical ammonia synthesis—the selectivity challenge. ACS Catal. 7, 706–709 (2017).
Smith, C., Hill, A. K. & Torrente-Murciano, L. Current and future role of Haber–Bosch ammonia in a carbon-free energy landscape. Energy Environ. Sci. 13, 331–344 (2020).
Innovation Outlook: Renewable Ammonia (International Renewable Energy Agency & Ammonia Energy Association, 2022)
Weng, G. et al. A high-efficiency electrochemical proton-conducting membrane reactor for ammonia production at intermediate temperatures. Joule 7, 1333–1346 (2023).
Acknowledgements
We gratefully acknowledge the funding by Villum Fonden, part of the Villum Center for the Science of Sustainable Fuels and Chemicals (V-SUSTAIN grant 9455), Innovationsfonden (E-ammonia grant 9067-00010B), the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement number 741860) and MSCA European Postdoctoral Fellowships (Electro-Ammonia Project 101059643).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Rebecca Hodgetts and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, S., Fu, X., Nørskov, J.K. et al. Towards sustainable metal-mediated ammonia electrosynthesis. Nat Energy 9, 1344–1349 (2024). https://doi.org/10.1038/s41560-024-01622-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-024-01622-7
This article is cited by
-
Modulated metal-support interactions for efficient nitrate electroreduction at positive potentials
Nature Communications (2026)
-
Revealing and modulating catalyst reconstruction for highly efficient electrosynthesis of ammonia
Nature Communications (2025)
-
Ammonia as a renewable energy carrier from synthesis to utilization
Nature Reviews Clean Technology (2025)
-
Metastable dual-defect states drive deep protonation for selective CO2 photomethanation
Nature Communications (2025)
-
Advancing lithium-mediated ammonia synthesis
Nature Reviews Chemistry (2025)