Abstract
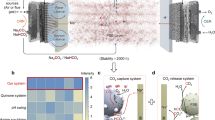
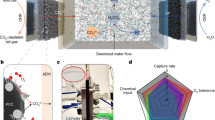
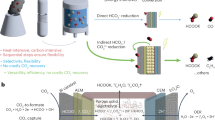
Carbon dioxide (CO2) and absorbent regeneration are the most energy-intensive processes in carbon capture loops. Conventional carbon capture technologies typically consume substantial amounts of heat and involve multiple steps for regeneration. Here we demonstrated one-step electrochemical regeneration of CO2 and alkaline absorbent from carbon-containing solutions in a modular porous solid electrolyte (PSE) reactor. By performing hydrogen evolution and oxidation redox reactions, our PSE reactor selectively split NaHCO3/Na2CO3 solutions, which typically come from air contactors after CO2 absorption, into NaOH absorbent in the catholyte and high-purity CO2 gas in the PSE layer. No chemicals were consumed and no by-products were generated. High Na+-ion transport number (~90%), high capture capacity retention (~90%), low energy consumptions (50 kJ molCO2−1 and 118 kJ molCO2−1 at 1 mA cm−2 and 100 mA cm−2 for bicarbonate, respectively) and long-term stability (>100 hours) were demonstrated. We achieved industrially relevant carbon regeneration rates of up to 1 A cm−2 (~18 mmol cm−2 h−1), highlighting the promising application potential.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data supporting the findings of this study are available in the main text, Supplementary Information and source data provided with this paper. Additional data related to this study may be requested from the corresponding author. Source data are provided with this paper.
References
Chu, S. Carbon capture and sequestration. Science 325, 1599 (2009).
Goeppert, A., Czaun, M., Prakash, G. S. & Olah, G. A. Air as the renewable carbon source of the future: an overview of CO2 capture from the atmosphere. Energy Environ. Sci. 5, 7833–7853 (2012).
Keith, D. W., Holmes, G., Angelo, D. S. & Heidel, K. A process for capturing CO2 from the atmosphere. Joule 2, 1573–1594 (2018).
Buckingham, J., Reina, T. R. & Duyar, M. S. Recent advances in carbon dioxide capture for process intensification. Carbon Capture Sci. Technol. 2, 100031 (2022).
Sanz-Pérez, E. S., Murdock, C. R., Didas, S. A. & Jones, C. W. Direct capture of CO2 from ambient air. Chem. Rev. 116, 11840–11876 (2016).
Rochelle, G. T. Amine scrubbing for CO2 capture. Science 325, 1652–1654 (2009).
Voskian, S. & Hatton, T. A. Faradaic electro-swing reactive adsorption for CO2 capture. Energy Environ. Sci. 12, 3530–3547 (2019).
Eisaman, M. D. et al. CO2 extraction from seawater using bipolar membrane electrodialysis. Energy Environ. Sci. 5, 7346–7352 (2012).
Zhu, P. et al. Continuous carbon capture in an electrochemical solid-electrolyte reactor. Nature 618, 959–966 (2023).
Shu, Q., Legrand, L., Kuntke, P., Tedesco, M. & Hamelers, H. V. Electrochemical regeneration of spent alkaline absorbent from direct air capture. Environ. Sci. Technol. 54, 8990–8998 (2020).
Liu, J. et al. Hydrogen-motivated electrolysis of sodium carbonate with extremely low cell voltage. Chem. Commun. 54, 3582–3585 (2018).
Muroyama, A. P. & Gubler, L. Carbonate regeneration using a membrane electrochemical cell for efficient CO2 capture. ACS Sustain. Chem. Eng. 10, 16113–16117 (2022).
Bougie, F. & Fan, X. Microwave regeneration of monoethanolamine aqueous solutions used for CO2 capture. Int. J. Greenh. Gas. Control 79, 165–172 (2018).
Bougie, F. & Iliuta, M. C. Analysis of regeneration of sterically hindered alkanolamines aqueous solutions with and without activator. Chem. Eng. Sci. 65, 4746–4750 (2010).
Yoo, M., Han, S.-J. & Wee, J.-H. Carbon dioxide capture capacity of sodium hydroxide aqueous solution. J. Environ. Manag. 114, 512–519 (2013).
Halliday, C. & Hatton, T. A. Sorbents for the capture of CO2 and other acid gases: a review. Ind. Eng. Chem. Res. 60, 9313–9346 (2021).
Stolaroff, J. K., Keith, D. W. & Lowry, G. V. Carbon dioxide capture from atmospheric air using sodium hydroxide spray. Environ. Sci. Technol. 42, 2728–2735 (2008).
Watkins, J. D. et al. Redox-mediated separation of carbon dioxide from flue gas. Energy Fuels 29, 7508–7515 (2015).
Renfrew, S. E., Starr, D. E. & Strasser, P. Electrochemical approaches toward CO2 capture and concentration. ACS Catal. 10, 13058–13074 (2020).
Rheinhardt, J. H., Singh, P., Tarakeshwar, P. & Buttry, D. A. Electrochemical capture and release of carbon dioxide. ACS Energy Lett. 2, 454–461 (2017).
Eisaman, M. D., Alvarado, L., Larner, D., Wang, P. & Littau, K. A. CO2 desorption using high-pressure bipolar membrane electrodialysis. Energy Environ. Sci. 4, 4031–4037 (2011).
Sharifian, R., Wagterveld, R., Digdaya, I., Xiang, C.-X. & Vermaas, D. Electrochemical carbon dioxide capture to close the carbon cycle. Energy Environ. Sci. 14, 781–814 (2021).
Willauer, H. D., DiMascio, F., Hardy, D. R. & Williams, F. W. Development of an electrolytic cation exchange module for the simultaneous extraction of carbon dioxide and hydrogen gas from natural seawater. Energy Fuels 31, 1723–1730 (2017).
Gurkan, B. et al. Perspective and challenges in electrochemical approaches for reactive CO2 separations. iScience 24, 103422 (2021).
Xia, R., Overa, S. & Jiao, F. Emerging electrochemical processes to decarbonize the chemical industry. JACS Au 2, 1054–1070 (2022).
Digdaya, I. A. et al. A direct coupled electrochemical system for capture and conversion of CO2 from oceanwater. Nat. Commun. 11, 4412 (2020).
Liu, Y., Ye, H.-Z., Diederichsen, K. M., Van Voorhis, T. & Hatton, T. A. Electrochemically mediated carbon dioxide separation with quinone chemistry in salt-concentrated aqueous media. Nat. Commun. 11, 2278 (2020).
Rau, G. H., Willauer, H. D. & Ren, Z. J. The global potential for converting renewable electricity to negative-CO2-emissions hydrogen. Nat. Clim. Change 8, 621–625 (2018).
Wang, M., Herzog, H. J. & Hatton, T. A. CO2 capture using electrochemically mediated amine regeneration. Ind. Eng. Chem. Res. 59, 7087–7096 (2020).
Welch, A. J., Dunn, E., DuChene, J. S. & Atwater, H. A. Bicarbonate or carbonate processes for coupling carbon dioxide capture and electrochemical conversion. ACS Energy Lett. 5, 940–945 (2020).
Gilliam, R. J. et al. Low voltage electrochemical process for direct carbon dioxide sequestration. J. Electrochem. Soc. 159, B627 (2012).
Park, H. S. et al. CO2 fixation by membrane separated NaCl electrolysis. Energies 8, 8704–8715 (2015).
Stucki, S., Schuler, A. & Constantinescu, M. Coupled CO2 recovery from the atmosphere and water electrolysis: feasibility of a new process for hydrogen storage. Int. J. Hydrogen Energy 20, 653–663 (1995).
Rahimi, M. et al. Carbon dioxide capture using an electrochemically driven proton concentration process. Cell Rep. Phys. Sci 1, 100033 (2020).
Prajapati, A. et al. Migration-assisted, moisture gradient process for ultrafast, continuous CO2 capture from dilute sources at ambient conditions. Energy Environ. Sci. 15, 680–692 (2022).
Legrand, L., Shu, Q., Tedesco, M., Dykstra, J. & Hamelers, H. Role of ion exchange membranes and capacitive electrodes in membrane capacitive deionization (MCDI) for CO2 capture. J. Colloid Interface Sci. 564, 478–490 (2020).
Willauer, H. D., DiMascio, F., Hardy, D. R. & Williams, F. W. Feasibility of CO2 extraction from seawater and simultaneous hydrogen gas generation using a novel and robust electrolytic cation exchange module based on continuous electrodeionization technology. Ind. Eng. Chem. Res. 53, 12192–12200 (2014).
de Lannoy, C.-F. et al. Indirect ocean capture of atmospheric CO2: part I. Prototype of a negative emissions technology. Int. J. Greenh. Gas. Control 70, 243–253 (2018).
Sharifian, R., Boer, L., Wagterveld, R. & Vermaas, D. Oceanic carbon capture through electrochemically induced in situ carbonate mineralization using bipolar membrane. Chem. Eng. J. 438, 135326 (2022).
Mehmood, A. et al. A novel high performance configuration of electrochemical cell to produce alkali for sequestration of carbon dioxide. Electrochim. Acta 219, 655–663 (2016).
Xia, C., Xia, Y., Zhu, P., Fan, L. & Wang, H. Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte. Science 366, 226–231 (2019).
Xia, C. et al. Continuous production of pure liquid fuel solutions via electrocatalytic CO2 reduction using solid-electrolyte devices. Nat. Energy 4, 776–785 (2019).
Kim, J. Y. T. et al. Recovering carbon losses in CO2 electrolysis using a solid electrolyte reactor. Nat. Catal. 5, 288–299 (2022).
Shin, H., Hansen, K. U. & Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 4, 911–919 (2021).
FY 2018 Progress Report for the DOE Hydrogen and Fuel Cells Program (US Department of Energy, 2019).
Acknowledgements
This work was supported by the Robert A. Welch Foundation (grant number C-2051-20230405) and the David and Lucile Packard Foundation (grant number 2020-71371).
Author information
Authors and Affiliations
Contributions
X.Z. and H.W. conceived the project and designed the experiments. X.Z. and Z.F. performed the experimental study. P.Z. performed the TEA study. Y.X. performed the energy consumption comparison of different methods. X.Z. and H.W. wrote the paper with support from all authors. H.W. supervised this project.
Corresponding authors
Ethics declarations
Competing interests
X.Z. and H.W. are listed as inventors on a patent application filed by the Rice University that pertains to this work. The other authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Ung Lee and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–7, Figs. 1–22, Tables 1–5 and Refs. 1–25.
Supplementary Data 1 (download XLSX )
Source data for supplementary figures.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data for Fig. 3.
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 4.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Fang, Z., Zhu, P. et al. Electrochemical regeneration of high-purity CO2 from (bi)carbonates in a porous solid electrolyte reactor for efficient carbon capture. Nat Energy 10, 55–65 (2025). https://doi.org/10.1038/s41560-024-01654-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-024-01654-z
This article is cited by
-
Energy-efficient indirect (bi)carbonate electroreduction in a porous solid electrolyte reactor
Nature Sustainability (2026)
-
Dual-confinement of reconstructed covalent organic framework for enhanced CO2 electrolysis in acid
Science China Materials (2026)
-
Electrochemical valorization of H2S in natural gas to sulfate under mild conditions
Nature Communications (2025)
-
Passive direct air capture via evaporative carbonate crystallization
Nature Chemical Engineering (2025)
-
Beyond molecular transformations in electrochemical porous solid electrolyte reactors
Nature Chemical Engineering (2025)