Abstract
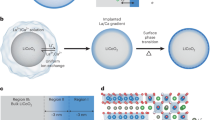
High-energy-density lithium-ion batteries for extreme conditions require cathodes that remain stable under harsh operation, including ultrahigh cutoff voltage and extreme temperatures. For Ni-rich layered cathodes, raising the charge voltage from 4.3 V to 4.8 V (versus Li+/Li) increases the energy density, yet this sacrifices cycling stability and remains challenging. Here we report a dopant-pairing method that achieves highly enriched Ti4+ (~9-nm surface layer) in LiNi0.8Co0.1Mn0.1O2 facilitated by Na+, enabling significantly enhanced high-voltage cyclability. Such high surface Ti4+ concentrations are unattainable without pairing Na+, representing a form of supersaturation within the layered cathode matrix. The enhanced stability is linked to improved structural integrity and reduced cathode–electrolyte side reactions (for example, O2 and CO2 evolution). In addition, ion transport is better preserved even after prolonged cycling at 4.8 V. This work highlights the power of supersaturated high-valence d0 cation Mz+ (z ≥ 4) in modifying the cathode–electrolyte interactions and degradation pathway.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data supporting the findings in the present work are included in the Article and its Supplementary Information. Source data are provided with this paper.
References
Li, M., Lu, J., Chen, Z. W. & Amine, K. 30 years of lithium-ion batteries. Adv. Mater. 30, 1800561 (2018).
Xu, G. L. et al. Challenges and strategies to advance high-energy nickel-rich layered lithium transition metal oxide cathodes for harsh operation. Adv. Funct. Mater. 30, 2004748 (2020).
Ahmed, S. et al. Cost of automotive lithium-ion batteries operating at high upper cutoff voltages. J. Power Sources 403, 56–65 (2018).
Xue, W. J. et al. Ultra-high-voltage Ni-rich layered cathodes in practical Li metal batteries enabled by a sulfonamide-based electrolyte. Nat. Energy 6, 495–505 (2021).
Jung, R., Metzger, M., Maglia, F., Stinner, C. & Gasteiger, H. A. Oxygen release and its effect on the cycling stability of LiNixMnyCozO2 (NMC) cathode materials for Li-ion batteries. J. Electrochem. Soc. 164, A1361 (2017).
Mukherjee, P. et al. Surface structural and chemical evolution of layered LiNi0.8Co0.15Al0.05O2 (NCA) under high voltage and elevated temperature conditions. Chem. Mater. 30, 8431–8445 (2018).
Yan, P. F. et al. Intragranular cracking as a critical barrier for high-voltage usage of layer-structured cathode for lithium-ion batteries. Nat. Commun. 8, 14101 (2017).
Gilbert, J. A., Shkrob, I. A. & Abraham, D. P. Transition metal dissolution, ion migration, electrocatalytic reduction and capacity loss in lithium-ion full cells. J. Electrochem. Soc. 164, A389 (2017).
Liu, X. et al. Probing the thermal-driven structural and chemical degradation of Ni-rich layered cathodes by Co/Mn exchange. J. Am. Chem. Soc. 142, 19745–19753 (2020).
Mu, L. Q. et al. Oxygen release induced chemomechanical breakdown of layered cathode materials. Nano Lett. 18, 3241–3249 (2018).
Namkoong, B. et al. High-energy Ni-rich cathode materials for long-range and long-Life electric vehicles. Adv. Energy Mater. 12, 2200615 (2022).
Cai, M. Z. et al. Stalling oxygen evolution in high-voltage cathodes by lanthurization. Nat. Energy 8, 159–168 (2023).
Csernica, P. M. et al. Persistent and partially mobile oxygen vacancies in Li-rich layered oxides. Nat. Energy 6, 642–652 (2021).
Xiao, P. H., Shi, T., Huang, W. X. & Ceder, G. Understanding surface densified phases in Ni-rich layered compounds. ACS Energy Lett. 4, 811–818 (2019).
Lin, F. et al. Surface reconstruction and chemical evolution of stoichiometric layered cathode materials for lithium-ion batteries. Nat. Commun. 5, 3529 (2014).
Xu, C. et al. Bulk fatigue induced by surface reconstruction in layered Ni-rich cathodes for Li-ion batteries. Nat. Mater. 20, 84–92 (2021).
Ahmed, S. et al. The Role of intragranular nanopores in capacity fade of nickel-rich layered Li(Ni1−x−yCoxMny)O2 cathode materials. ACS Nano 13, 10694–10704 (2019).
Ryu, H. H., Lim, H. W., Lee, S. G. & Sun, Y. K. Near-surface reconstruction in Ni-rich layered cathodes for high-performance lithium-ion batteries. Nat. Energy 9, 47–56 (2024).
Zhang, Y. R. et al. Revealing electrolyte oxidation via carbonate dehydrogenation on Ni-based oxides in Li-ion batteries by in situ Fourier transform infrared spectroscopy. Energy Environ. Sci. 13, 183–199 (2020).
Tu, S. B. et al. Fast-charging capability of graphite-based lithium-ion batteries enabled by Li3P-based crystalline solid-electrolyte interphase. Nat. Energy 8, 1365–1374 (2023).
Jagger, B. & Pasta, M. Solid electrolyte interphases in lithium metal batteries. Joule 7, 2228–2244 (2023).
Sun, Y. J. et al. Expandable Li percolation network: the effects of site distortion in cation-disordered rock-salt cathode material. J. Am. Chem. Soc. 145, 11717–11726 (2023).
Lee, J. et al. Determining the criticality of Li-excess for disordered-rocksalt Li-Ion battery cathodes. Adv. Energy Mater. 11, 2100204 (2021).
Zhang, J. N. et al. Trace doping of multiple elements enables stable battery cycling of LiCoO2 at 4.6V. Nat. Energy 4, 594–603 (2019).
Kang, K. et al. Synthesis and electrochemical properties of layered Li0.9Ni0.45Ti0.55O2. Chem. Mater. 15, 4503–4507 (2003).
Guo, S. H. et al. High-performance symmetric sodium-ion batteries using a new, bipolar O3-type material, Na0.8Ni0.4Ti0.6O2. Energy Environ. Sci. 8, 1237–1244 (2015).
Wang, P. F. et al. Na+/vacancy disordering promises high-rate Na-ion batteries. Sci. Adv. 4, eaar6018 (2018).
Yu, H. J., Guo, S. H., Zhu, Y. B., Ishida, M. & Zhou, H. S. Novel titanium-based O3-type NaTi0.5Ni0.5O2 as a cathode material for sodium ion batteries. Chem. Commun. 50, 457–459 (2014).
Ohzuku, T., Ueda, A. & Nagayama, M. Electrochemistry and structural chemistry of LiNiO2 (R3m) for 4 volt secondary lithium cells. J. Electrochem. Soc. 140, 1862 (1993).
Liu, T. C. et al. Ultrastable cathodes enabled by compositional and structural dual-gradient design. Nat. Energy 9, 1252–1263 (2024).
Lin, R. Q. et al. Hierarchical nickel valence gradient stabilizes high-nickel content layered cathode materials. Nat. Commun. 12, 2350 (2021).
Zhang, J. B. et al. Multifunctional solvent molecule design enables high-voltage Li-ion batteries. Nat. Commun. 14, 2211 (2023).
Wu, Z. J. et al. Cu–N synergism regulation to enhance anionic redox reversibility and activity of Li- and Mn-rich layered oxides cathode. Small 20, 2401645 (2024).
Cui, Z. H., Guo, Z. Z. & Manthiram, A. Irreparable interphase chemistry degradation induced by temperature pulse in lithium-ion batteries. Angew. Chem. Int. Ed. 135, e202313437 (2023).
Liu, W. et al. Inhibition of transition metals dissolution in cobalt-free cathode with ultrathin robust interphase in concentrated electrolyte. Nat. Commun. 11, 3629 (2020).
Zhang, Y. X. et al. Ultrahigh active material content and highly stable Ni-rich cathode leveraged by oxidative chemical vapor deposition. Energy Storage Mater. 48, 1–11 (2022).
Neudeck, S. et al. Molecular surface modification of NCM622 cathode material using organophosphates for improved Li-ion battery full-cells. ACS Appl. Mater. Interfaces 10, 20487–20498 (2018).
Yi, M. C., Dolocan, A. & Manthiram, A. Stabilizing the interphase in cobalt-free, ultrahigh-nickel cathodes for lithium-ion batteries. Adv. Funct. Mater. 33, 2213164 (2023).
Sun, H. H., Dolocan, A., Weeks, J. A., Heller, A. & Mullins, C. B. Stabilization of a highly Ni-rich layered oxide cathode through flower-petal grain arrays. ACS Nano 14, 17142–17150 (2020).
Zhang, J. X. et al. Interfacial design for a 4.6 V high-voltage single-crystalline LiCoO2 cathode. Adv. Mater. 34, 2108353 (2022).
Zhao, W. G. et al. High voltage operation of Ni-rich NMC cathodes enabled by stable electrode/electrolyte interphases. Adv. Energy Mater. 8, 1800297 (2018).
Acknowledgements
F.H. acknowledges the support by Science and Technology Commission of Shanghai Municipality (23DZ1200800).
Author information
Authors and Affiliations
Contributions
F.H. supervised the project. H.L. synthesized the materials and conducted the electrochemical testing and the characterizations. H.L., Y.D., Y.L. and F.H. analysed the data. H.L., Y.D. and F.H. wrote the paper. Y.T., Y.L. and W.M. contributed to pouch-type full cell assembly. All authors discussed and contributed to the writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Noriyoshi Matsumi and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–60, Notes 1 and 2 and Table 1.
Source data
Source Data Fig. 1 (download XLSX )
Source data for Fig. 1c,d,g.
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2a–i.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3d–h.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4b–g,l.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liao, H., Tang, Y., Ma, W. et al. Exceptional layered cathode stability at 4.8 V via supersaturated high-valence cation design. Nat Energy 10, 1107–1115 (2025). https://doi.org/10.1038/s41560-025-01831-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01831-8
This article is cited by
-
Stability through supersaturation
Nature Energy (2025)
-
Crystal Packing Factor-guided Design of Stress-resistant Alloying-type Anodes for Durable Sodium-ion Storage
Chemical Research in Chinese Universities (2025)