Abstract
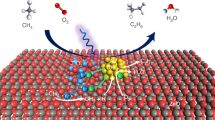
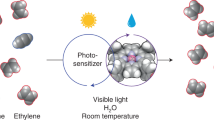
Photocatalytic oxidative coupling of methane (POCM) enables the production of value-added fuels and chemicals using renewable solar energy. Unfortunately, despite recent advances in the production of C2 chemicals (for example, ethane), POCM systems that selectively produce industrially useful and transportable C3+ hydrocarbons remain elusive. Here we report that Au-embedded porous TiO2, activated by steam during the POCM process, enables efficient and selective flow synthesis of propane with a productivity of 1.4 mmol h−1. At this productivity, we achieve a high propane selectivity of 91.3% and an apparent quantum efficiency of 39.7% at a wavelength of 365 nm. Mechanistic studies reveal that the tensile-strained Au and the nanopore-confined catalytic microenvironment jointly stabilize key ethane intermediates, boosting deeper C2–C1 coupling to form propane. Meanwhile, the steam-activated surface lattice oxygen on TiO2 accelerates hydrogen species transfer, thus enhancing POCM kinetics. This approach is economically feasible for practical application under concentrated solar light.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the Article and its Supplementary Information. Source data are provided with this paper.
References
BP Statistical Review of World Energy July (bp, 2023); http://www.bp.com/statisticalreview
Annual Energy Outlook 2019 (EIA, 2019); https://www.eia.gov/outlooks/archive/aeo19/
Luna, P. D. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Song, R. et al. Ethylene production via photocatalytic dehydrogenation of ethane using LaMn1−xCuxO3. Nat. Energy 9, 750–760 (2024).
Zhang, L. et al. Visible-light-driven non-oxidative dehydrogenation of alkanes at ambient conditions. Nat. Energy 7, 1042–1051 (2022).
Li, X., Wang, C. & Tang, J. Methane transformation by photocatalysis. Nat. Rev. Mater. 7, 617–632 (2022).
Wang, P., Shi, R., Zhao, J. & Zhang, T. Photodriven methane conversion on transition metal oxide catalyst: recent progress and prospects. Adv. Sci. 11, 2305471 (2023).
Sushkevich, V. L., Palagin, D., Ranocchiari, M. & Bokhoven, J. A. V. Selective anaerobic oxidation of methane enables direct synthesis of methanol. Science 356, 523–527 (2017).
Schwach, P., Pan, X. & Bao, X. Direct conversion of methane to value-added chemicals over heterogeneous catalysts: challenges and prospects. Chem. Rev. 117, 8497–8520 (2017).
Wang, P., Zhao, G., Wang, Y. & Lu, Y. MnTiO3-driven low-temperature oxidative coupling of methane over TiO2-doped Mn2O3-Na2WO4/SiO2 catalyst. Sci. Adv. 3, e1603180 (2017).
Wortman, J., Igenegbai, V. O., Almallahi, R., Motagamwala, A. H. & Linic, S. Optimizing hierarchical membrane/catalyst systems for oxidative coupling of methane using additive manufacturing. Nat. Mater. 22, 1523–1530 (2023).
Li, X. et al. Efficient hole abstraction for highly selective oxidative coupling of methane by Au-sputtered TiO2 photocatalysts. Nat. Energy 8, 1013–1022 (2023).
Song, S. et al. A selective Au-ZnO/TiO2 hybrid photocatalyst for oxidative coupling of methane to ethane with dioxygen. Nat. Catal. 4, 1032–1042 (2021).
Wang, P. et al. Selective photocatalytic oxidative coupling of methane via regulating methyl intermediates over metal/ZnO nanoparticles. Angew. Chem. Int. Ed. 62, e202304301 (2023).
Li, X. et al. PdCu nanoalloy decorated photocatalysts for efficient and selective oxidative coupling of methane in flow reactors. Nat. Commun. 14, 6343 (2023).
Zhai, G. et al. Highly efficient, selective, and stable photocatalytic methane coupling to ethane enabled by lattice oxygen looping. Sci. Adv. 10, eado4390 (2024).
Kallio, P., Pásztor, A., Akhtar, M. K. & Jones, P. R. An engineered pathway for the biosynthesis of renewable propane. Nat. Commun. 5, 4731 (2014).
Zeng, L. et al. Stable anchoring of single rhodium atoms by indium in zeolite alkane dehydrogenation catalysts. Science 383, 998–1004 (2019).
Burgess, D. R. Thermochemical data. in NIST Chemistry WebBook NIST Standard Reference Database Number 69 (eds Linstrom, P. J. & Mallard, W. G.) (National Institute of Standards and Technology, 2012); http://webbook.nist.gov
Yan, H. et al. Tandem In2O3-Pt/Al2O3 catalyst for coupling of propane dehydrogenation to selective H2 combustion. Science 371, 1257–1260 (2021).
Wang, W. et al. Tandem propane dehydrogenation and surface oxidation catalysts for selective propylene synthesis. Science 381, 886–890 (2023).
Waku, T., Yu, S. Y. & Iglesia, E. Staged O2 introduction and selective H2 combustion during catalytic reactions of alkanes on cation-exchanged H-ZSM5. Ind. Eng Chem. Res. 42, 3680–3689 (2003).
Zhuang, T.-T. et al. Copper nanocavities confine intermediates for efficient electrosynthesis of C3 alcohol fuels from carbon monoxide. Nat. Catal. 1, 946–951 (2018).
Zhang, Y. et al. Single-atom Cu anchored catalysts for photocatalytic renewable H2 production with a quantum efficiency of 56%. Nat. Commun. 13, 58 (2022).
Feng, X., Jiang, K., Fan, S. & Kanan, M. W. Grain-boundary-dependent CO2 electroreduction activity. J. Am. Chem. Soc. 137, 4606–4609 (2015).
Mariano, R. G. et al. Microstructural origin of locally enhanced CO2 electroreduction activity on gold. Nat. Mater. 20, 1000–1006 (2021).
Mariano, R. G., McKelvey, K., White, H. S. & Kanan, M. W. Selective increase in CO2 electroreduction activity at grain-boundary surface terminations. Science 358, 1187–1192 (2017).
Li, Z. et al. A silver catalyst activated by stacking faults for the hydrogen evolution reaction. Nat. Catal. 2, 1107–1114 (2019).
Chen, L.-W. et al. Multi-twinned gold nanoparticles with tensile surface steps for efficient electrocatalytic CO2 reduction. Sci. China Chem. 65, 2188–2196 (2022).
Jiang, Y. et al. Enabling specific photocatalytic methane oxidation by controlling free radical type. J. Am. Chem. Soc. 145, 2698–2707 (2023).
Li, M. et al. Ultrafine jagged platinum nanowires enable ultrahigh mass activity for the oxygen reduction reaction. Science 354, 1414–1418 (2016).
Wang, H. et al. Direct and continuous strain control of catalysts with tunable battery electrode materials. Science 354, 1031–1036 (2016).
Luo, M. et al. PdMo bimetallene for oxygen reduction catalysis. Nature 574, 81–85 (2019).
Xu, M. et al. Insights into interfacial synergistic catalysis over Ni@TiO2–x catalyst toward water–gas shift reaction. J. Am. Chem. Soc. 140, 11241–11251 (2018).
Gu, H. et al. Water enables lattice oxygen activation of transition metal oxides for volatile organic compound oxidation. ACS Catal. 12, 11272–11280 (2022).
Nie, L. et al. Activation of surface lattice oxygen in single-atom Pt/CeO2 for low-temperature CO oxidation. Science 358, 1419–1423 (2017).
Dinh, C.-T. et al. Multi-site electrocatalysts for hydrogen evolution in neutral media by destabilization of water molecules. Nat. Energy 4, 107–114 (2019).
Zhang, X. et al. A stable low-temperature H2-production catalyst by crowding Pt on α-MoC. Nature 589, 396–401 (2021).
Yuan, W. et al. Visualizing H2O molecules reacting at TiO2 active sites with transmission electron microscopy. Science 367, 428–430 (2020).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. J. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide: an LSDA+U study. Phys. Rev. B 57, 1505–1510 (1998).
Wang, V., Xu, N., Liu, J.-C., Tang, G. & Geng, W.-D. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput. Phys. Commun. 267, 108033 (2021).
Wang, Y. et al. Photocatalytic oxidative coupling of methane over Au1Ag single-atom alloy modified ZnO with oxygen and water vapor: synergy of gold and silver. Angew. Chem. Int. Ed. 62, e202310525 (2023).
Sun, X. et al. Directional activation of oxygen by the Au-loaded ZnAl-LDH with defect structure for highly efficient photocatalytic oxidative coupling of methane. Small 20, 2310857 (2024).
Chen, Z. et al. Non-oxidative coupling of methane: N-type doping of niobium single atoms in TiO2–SiO2 induces electron localization. Angew. Chem. Int. Ed. 60, 11901–11909 (2021).
Chen, Z. et al. High-density frustrated Lewis pairs based on lamellar Nb2O5 for photocatalytic non-oxidative methane coupling. Nat. Commun. 14, 2000 (2023).
Ma, J. et al. Design of frustrated Lewis pair in defective TiO2 for photocatalytic non-oxidative methane coupling. Chem. Catal. 2, 1775–1792 (2022).
Wu, S. et al. Ga-doped and Pt-loaded porous TiO2–SiO2 for photocatalytic nonoxidative coupling of methane. J. Am. Chem. Soc. 141, 6592–6600 (2019).
Yu, X. et al. Stoichiometric methane conversion to ethane using photochemical looping at ambient temperature. Nat. Energy 5, 511–519 (2020).
Amano, F. & Ishimaru, M. Hydroxyl radical formation on metal-loaded Ga2O3 photocatalysts for dehydrogenative coupling of methane to ethane with water. Energy Fuels 36, 5393–5402 (2022).
Zhang, W. et al. High-performance photocatalytic nonoxidative conversion of methane to ethane and hydrogen by heteroatoms-engineered TiO2. Nat. Commun. 13, 2806 (2022).
Chen, Y. et al. Continuous flow system for highly efficient and durable photocatalytic oxidative coupling of methane. J. Am. Chem. Soc. 146, 2465–2473 (2024).
Li, X. et al. Platinum-and CuOx-decorated TiO2 photocatalyst for oxidative coupling of methane to C2 hydrocarbons in a flow reactor. Angew. Chem. Int. Ed. 59, 19702–19707 (2020).
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (grant nos 22405069, 51972094, 52371220, 22322101 and 22375017), the National Key Research and Development Program of China (grant no. 2020YFB1506300), the Beijing Institute of Technology Research Fund Program, the Hebei Provincial Department of Science and Technology (grant no. 236Z4403G), the Science Research Project of Hebei Education Department (grant no. JZX2024008), the Hebei Province Optoelectronic Information Materials Laboratory Performance Subsidy Fund Project (grant no. 22567634H), Hebei Education Department (grant no. BJ2025011) and the Research Innovation Team Project of Hebei University (grant nos 150000321008 and IT2023A04).
Author information
Authors and Affiliations
Contributions
Y.H., B.W. and J.Y. designed the research. W.N., L.C. and Y.H. performed the catalyst preparation, characterization, photocatalytic tests and data analysis. H.L., X.G., Z.W., H.-Z.H., J.L., J.W., C.S., S.N., L.G., Y.L., C.L. and S.W. assisted with the material synthesis, characterizations and catalysis measurements. W.N., Y.H., L.C., B.W. and J.Y. wrote the paper. Y.H., S.W, B.W. and J.Y. supervised the research. All authors discussed the results and assisted during paper preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–78, Tables 1–7 and Note 1.
Source data
Source Data Fig. 1 (download XLSX )
Source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nie, W., Chen, L., Hao, Y. et al. Photocatalytic oxidative coupling of methane to C3+ hydrocarbons via nanopore-confined microenvironments. Nat Energy 10, 1095–1106 (2025). https://doi.org/10.1038/s41560-025-01834-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01834-5
This article is cited by
-
Orchestrated multi-physics field-engineering toward valorized C2+ chemicals from CO2/CH4
Science China Materials (2026)