Abstract
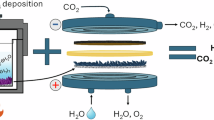
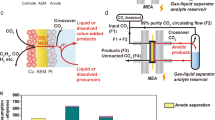
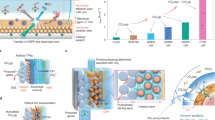
Electrochemical CO2 reduction can generate multi-carbon (C2+) products via a CO2-to-CO cascade followed by CO reduction (COR). However, COR energy efficiency remains below 40% due to sluggish ion transport within charge-selective membranes. Here we introduce an uncharged porous separator that enables facile transport of both ion types, reducing ohmic resistance and superconcentrating cations at the catalyst surface—lowering COR voltage by 150 mV at 200 mA cm−2. In previous electrolyser designs, porous separators were limited by cathode-to-anode H2 crossover; the low diffusivity of C2H4 and CO in water allows a separator three times thinner and 1.6 times more porous, markedly reducing overpotential. Operating at elevated temperatures with a nickel–iron-based anode further lowers voltage by 330 mV, leading to a full-cell voltage of 1.95 V at 200 mA cm−2 and an energy efficiency of 51% to C2+ products sustained over 250 h. The system also achieves a CO single-pass conversion of 97% and a C2H4 concentration of 87 wt% in the product gas stream.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and Supplementary Information files. Source data are provided with this paper.
References
O'Brien, C. P. et al. CO2 electrolyzers. Chem. Rev. 124, 3648–3693 (2024).
Chen, Y. et al. Catalyst design for electrochemical CO2 reduction to ethylene. Matter 7, 25–37 (2024).
Shin, H., Hansen, K. U. & Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 4, 911–919 (2021).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Chen, X. et al. Electrochemical CO2-to-ethylene conversion on polyamine-incorporated Cu electrodes. Nat. Catal. 4, 20–27 (2021).
García de Arquer, F. P. et al. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 367, 661–666 (2020).
Zhong, M. et al. Accelerated discovery of CO2 electrocatalysts using active machine learning. Nature 581, 178–183 (2020).
Dinh, C. T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
Ozden, A. et al. Carbon-efficient carbon dioxide electrolysers. Nat. Sustain. 5, 563–573 (2022).
Fan, M. et al. Cationic-group-functionalized electrocatalysts enable stable acidic CO2 electrolysis. Nat. Catal. 6, 763–772 (2023).
O’Brien, C. P. et al. Single pass CO2 conversion exceeding 85% in the electrosynthesis of multicarbon products via local CO2 regeneration. ACS Energy Lett. 6, 2952–2959 (2021).
Sisler, J. et al. Ethylene electrosynthesis: a comparative techno-economic analysis of alkaline vs membrane electrode assembly vs CO2-CO–C2H4 tandems. ACS Energy Lett. 6, 997–1002 (2021).
Ozden, A. et al. Cascade CO2 electroreduction enables efficient carbonate-free production of ethylene. Joule 5, 706–719 (2021).
Li, J. et al. Hydroxide is not a promoter of C2+ product formation in the electrochemical reduction of CO on copper. Angew. Chem. Int. Ed. 132, 4494–4499 (2020).
Jouny, M., Luc, W. & Jiao, F. High-rate electroreduction of carbon monoxide to multi-carbon products. Nat. Catal. 1, 745–755 (2018).
Ripatti, D. S., Veltman, T. R. & Kanan, M. W. Carbon monoxide gas diffusion electrolysis that produces concentrated C2 products with high single-pass conversion. Joule 3, 240–256 (2019).
Ozden, A. et al. Energy- and carbon-efficient CO2/CO electrolysis to multicarbon products via asymmetric ion migration–adsorption. Nat. Energy 8, 179–190 (2023).
Wang, X. et al. Efficient electrosynthesis of n-propanol from carbon monoxide using a Ag–Ru–Cu catalyst. Nat. Energy 7, 170–176 (2022).
Wu, H. et al. Selective and energy-efficient electrosynthesis of ethylene from CO2 by tuning the valence of Cu catalysts through aryl diazonium functionalization. Nat. Energy 9, 422–433 (2024).
Salvatore, D. A. et al. Designing anion exchange membranes for CO2 electrolysers. Nat. Energy 6, 339–348 (2021).
Hasa, B. et al. Benchmarking anion-exchange membranes for electrocatalytic carbon monoxide reduction. Chem. Catal. 3, 100450 (2023).
Du, N. et al. Anion-exchange membrane water electrolyzers. Chem. Rev. 122, 11830–11895 (2022).
Henkensmeier, D. et al. Overview: State-of-the-art commercial membranes for anion exchange membrane water electrolysis. J. Electrochem. Energy Convers. Storage 18, 024001 (2021).
Gilliam, R. J., Graydon, J. W., Kirk, D. W. & Thorpe, S. J. A review of specific conductivities of potassium hydroxide solutions for various concentrations and temperatures. Int. J. Hydrog. Energy 32, 359–364 (2007).
Monteiro, M. C. O. et al. Absence of CO2 electroreduction on copper, gold and silver electrodes without metal cations in solution. Nat. Catal. 4, 654–662 (2021).
Zi, X. et al. Breaking K+ concentration limit on Cu nanoneedles for acidic electrocatalytic CO2 reduction to multi-carbon products. Angew. Chem. Int. Ed. 62, e202309351 (2023).
Lee, H. I. et al. The synthesis of a Zirfon-type porous separator with reduced gas crossover for alkaline electrolyzer. Int. J. Energy Res. 44, 1875–1885 (2020).
Ha, T. H., Kim, J., Choi, H. & Oh, J. Selective zero-gap CO2 reduction in acid. ACS Energy Lett. 9, 4835–4842 (2024).
Perazio, A., Schreiber, M. W., Creissen, C. E. & Fontecave, M. Low-voltage acidic CO2 reduction enabled by a diaphragm-based electrolyzer. ChemElectroChem 11, e202400045 (2024).
Xu, Q. et al. Integrated reference electrodes in anion-exchange-membrane electrolyzers: impact of stainless-steel gas-diffusion layers and internal mechanical pressure. ACS Energy Lett. 6, 305–312 (2021).
Hu, C., Zhang, L. & Gong, J. Recent progress made in the mechanism comprehension and design of electrocatalysts for alkaline water splitting. Energy Environ. Sci. 12, 2620–2645 (2019).
Xie, X. et al. Oxygen evolution reaction in alkaline environment: material challenges and solutions. Adv. Funct. Mater. 32, 2110036 (2022).
Wang, N. et al. Boride-derived oxygen-evolution catalysts. Nat. Commun. 12, 6089 (2021).
Varcoe, J. R. & Slade, R. C. T. Prospects for alkaline anion-exchange membranes in low temperature fuel cells. Fuel Cells 5, 187–200 (2005).
Xue, J. et al. High-temperature anion-exchange membrane fuel cells with balanced water management and enhanced stability. Joule 8, 1457–1477 (2024).
Vos, R. E. & Koper, M. T. M. The effect of temperature on the cation-promoted electrochemical CO2 reduction on gold. ChemElectroChem 9, e202200239 (2022).
Vos, R. E. et al. How temperature affects the selectivity of the electrochemical CO2 reduction on copper. ACS Catal. 13, 8080–8091 (2023).
Li, J. et al. Constraining CO coverage on copper promotes high-efficiency ethylene electroproduction. Nat. Catal. 2, 1124–1131 (2019).
Tan, Y. C., Lee, K. B., Song, H. & Oh, J. Modulating local CO2 concentration as a general strategy for enhancing C–C coupling in CO2 electroreduction. Joule 4, 1104–1120 (2020).
da Cunha, S. C. & Resasco, J. Maximizing single-pass conversion does not result in practical readiness for CO2 reduction electrolyzers. Nat. Commun. 14, 5513 (2023).
Prajapati, A. et al. CO2-free high-purity ethylene from electroreduction of CO2 with 4% solar-to-ethylene and 10% solar-to-carbon efficiencies. Cell Rep. Phys. Sci. 3, 101053 (2022).
Gabardo, C. M. et al. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 3, 2777–2791 (2019).
Liu, W. et al. Electrochemical CO2 reduction to ethylene by ultrathin CuO nanoplate arrays. Nat. Commun. 13, 1877 (2022).
Acknowledgements
D.S. acknowledges support from the Canada Research Chairs Program. R.K.M. thanks the Natural Sciences and Engineering Research Council (NSERC), Hatch and the Government of Ontario for their support through graduate scholarships. This work is financially supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) and TOTAL SE.
Author information
Authors and Affiliations
Contributions
E.H.S. and D.S. supervised the work. R.K.M. conceived the idea. R.K.M. designed and carried out all the experiments with help from M.F., N.W. and Y.Z. R.K.M. prepared the manuscript. N.W. synthesized the NiFe–B anode material. F.L. performed the one-dimensional simulation. F.A. designed the electrolyser incorporating a reference electrode. C.P.O., P.P. and A.S.Z. prepared the hydrophobic carbon substrate. Y.L., W.N., K.X., Y.C., P.S., J.E.H. and J.W. assisted with experiments. M.L., J.K., Y.C.X., Y.X. and C.-T.D. contributed to data analysis and discussions. All authors discussed the results and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
There is a US provisional patent application (US 63/656,427) titled “Interposed electrode assembly for electroreduction of carbon oxides to C2+ products”, filed by R.K.M., D.S. and E.H.S. and their institutions. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Joel Ager III, Charles Creissen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–3, Figs. 1–31 and Tables 1–5.
Supplementary Data 1 (download XLSX )
Source data for Supplementary Figures.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miao, R.K., Fan, M., Wang, N. et al. CO electrolysers with 51% energy efficiency towards C2+ using porous separators. Nat Energy 10, 1197–1204 (2025). https://doi.org/10.1038/s41560-025-01846-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01846-1
This article is cited by
-
Scaling electrocatalysts for reduction of CO2 or CO to multicarbon products
Nature Reviews Materials (2026)
-
Swapping membranes for separators
Nature Energy (2025)