Abstract
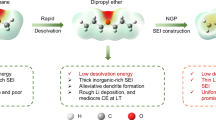
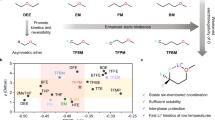
Short circuits in lithium metal batteries caused by separator failure at elevated temperatures present a critical thermal safety challenge. Smart, temperature-responsive materials offer a promising way to prevent short circuits, yet practical systems with sufficiently fast response times have not been realized. Here we propose a thermo-responsive electrolyte that undergoes a rapid liquid-to-solid phase transition upon heating, offering a highly effective strategy to enhance lithium metal battery safety. The electrolyte leverages LiPF6 to initiate cationic polymerization, enabling solidification within seconds at a temperature threshold near the separator’s melting point. This fast phase change forms an effective heat shield that prevents internal short circuits and thermal runaway. Demonstrated in LiFePO4||Li pouch cells, the electrolyte ensures stable operation up to 90 °C and completely suppresses thermal runaway. Notably, the transition temperature can be tuned between 100 °C and 150 °C, allowing compatibility with various commercial separators. This ultrafast thermo-responsive electrolyte offers a pathway towards the design of intrinsically safe lithium metal batteries.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All of the data are included within this article and its Supplementary Information. Source data are provided with this paper.
References
Wang, C.-Y. et al. Fast charging of energy-dense lithium-ion batteries. Nature 611, 485–490 (2022).
Kim, S. et al. Lithium-metal batteries: from fundamental research to industrialization. Adv. Mater. 35, 2206625 (2023).
He, M. et al. Industry needs for practical lithium-metal battery designs in electric vehicles. Nat. Energy 9, 1199–1205 (2024).
Fang, C. et al. Quantifying inactive lithium in lithium metal batteries. Nature 572, 511–515 (2019).
Sun, H. et al. High-safety and high-energy-density lithium metal batteries in a novel ionic-liquid electrolyte. Adv. Mater. 32, 2001741 (2020).
Zou, P. et al. Directing lateral growth of lithium dendrites in micro-compartmented anode arrays for safe lithium metal batteries. Nat. Commun. 9, 464 (2018).
Xu, J. et al. Electrolyte design for Li-ion batteries under extreme operating conditions. Nature 614, 694–700 (2023).
Meng, Y. et al. Designing phosphazene-derivative electrolyte matrices to enable high-voltage lithium metal batteries for extreme working conditions. Nat. Energy 8, 1023–1033 (2023).
Liu, K., Liu, Y., Lin, D., Pei, A. & Cui, Y. Materials for lithium-ion battery safety. Sci. Adv. 4, eaas9820 (2018).
Masalkovaitė, K., Gasper, P. & Finegan, D. P. Predicting the heat release variability of Li-ion cells under thermal runaway with few or no calorimetry data. Nat. Commun. 15, 8399 (2024).
Song, I. T. et al. Thermal runaway prevention through scalable fabrication of safety reinforced layer in practical Li-ion batteries. Nat. Commun. 15, 8294 (2024).
Wu, H., Zhuo, D., Kong, D. & Cui, Y. Improving battery safety by early detection of internal shorting with a bifunctional separator. Nat. Commun. 5, 5193 (2014).
Xu, J., Lan, C., Qiao, Y. & Ma, Y. Prevent thermal runaway of lithium-ion batteries with minichannel cooling. Appl. Therm. Eng 110, 883–890 (2017).
Liu, Y. et al. Anion-repulsive polyoxometalate@MOF-modified separators for dendrite-free and high-rate lithium batteries. Interdiscip. Mater. 4, 190–200 (2025).
Zhang, W. et al. Single-phase local-high-concentration solid polymer electrolytes for lithium-metal batteries. Nat. Energy 9, 386–400 (2024).
Zhang, J.-F. et al. ZIF-8-functionalized polymer electrolyte with enhanced performance for high-temperature solid-state lithium metal batteries. Rare Met. 43, 984–994 (2024).
Huang, Z. et al. A salt-philic, solvent-phobic interfacial coating design for lithium metal electrodes. Nat. Energy 8, 577–585 (2023).
Zhang, Z. et al. Rapid thermal shutdown of deep-eutectic-polymer electrolyte enabling overheating self-protection of lithium metal batteries. Adv. Sci. 11, 2409628 (2024).
Jiang, F.-N. et al. Thermoresponsive electrolytes for safe lithium-metal batteries. Adv. Mater. 35, 2209114 (2023).
Zhou, Q. et al. A temperature-responsive electrolyte endowing superior safety characteristic of lithium metal batteries. Adv. Energy Mater. 10, 1903441 (2020).
Zhang, X. et al. Deciphering the thermal failure mechanism of anode-free lithium metal pouch batteries. Adv. Energy Mater. 13, 2203648 (2023).
Dong, T. et al. A smart polymer electrolyte coordinates the trade-off between thermal safety and energy density of lithium batteries. Energy Storage Mater. 58, 123–131 (2023).
Lanceros-Méndez, S., Mano, J. F., Costa, A. M. & Schmidt, V. H. FTIR and DSC studies of mechanically deformed β-PVDF films. J. Macromol. Sci. Part B 40, 517–527 (2001).
Xu, Y. et al. Enhanced-quantum yield sulfur/nitrogen co-doped fluorescent carbon nanodots produced from biomass Enteromorpha prolifera: synthesis, posttreatment, applications and mechanism study. Sci. Rep. 7, 4499 (2017).
Hwang, S. S., Cho, C. G. & Kim, H. Room temperature cross-linkable gel polymer electrolytes for lithium ion batteries by in situ cationic polymerization of divinyl ether. Electrochem. Commun. 12, 916–919 (2010).
Dong, Y. & Edgar, K. J. Imparting functional variety to cellulose ethers via olefin cross-metathesis. Polym. Chem. 6, 3816–3827 (2015).
Liu, F.-Q. et al. Upgrading traditional liquid electrolyte via in situ gelation for future lithium metal batteries. Sci. Adv. 4, eaat5383 (2018).
Han, J.-G., Kim, K., Lee, Y. & Choi, N.-S. Scavenging materials to stabilize LiPF6-containing carbonate-based electrolytes for Li-ion batteries. Adv. Mater. 31, 1804822 (2019).
Jiang, H.-Z. et al. Electrophilically trapping water for preventing polymerization of cyclic ether towards low-temperature Li metal battery. Angew. Chem. Int. Ed. 62, e202300238 (2023).
Liu, X. et al. Why LiFePO4 is a safe battery electrode: Coulomb repulsion induced electron-state reshuffling upon lithiation. Phys. Chem. Chem. Phys. 17, 26369–26377 (2015).
Hu, J., Huang, W., Yang, L. & Pan, F. Structure and performance of the LiFePO4 cathode material: from the bulk to the surface. Nanoscale 12, 15036–15044 (2020).
Zhang, G. et al. A monofluoride ether-based electrolyte solution for fast-charging and low-temperature non-aqueous lithium metal batteries. Nat. Commun. 14, 1081 (2023).
Yang, A. et al. Benchmarking the safety performance of organic electrolytes for rechargeable lithium batteries: a thermochemical perspective. ACS Energy Lett. 8, 836–843 (2023).
Chen, S., Cook, W. D. & Chen, F. Thermal and photopolymerization of divinyl ethers using an iodonium initiator: the effect of temperature. Polym. Int. 56, 1423–1431 (2007).
Zhou, H. et al. In situ formed polymer gel electrolytes for lithium batteries with inherent thermal shutdown safety features. J. Mater. Chem. A 7, 16984–16991 (2019).
Xia, L., Wang, D., Yang, H., Cao, Y. & Ai, X. An electrolyte additive for thermal shutdown protection of Li-ion batteries. Electrochem. Commun. 25, 98–100 (2012).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Perdew, J. P. et al. Restoring the density-gradient expansion for exchange in solids and surfaces. Phys. Rev. Lett. 100, 136406 (2008).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Liu, Y. & Goddard, W. A. III. A universal damping function for empirical dispersion correction on density functional theory. Mater. Trans. 50, 1664–1670 (2009).
Moellmann, J. & Grimme, S. DFT-D3 study of some molecular crystals. J. Phys. Chem. C. 118, 7615–7621 (2014).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2023YFB2406100), the National Natural Science Foundation of China (grant numbers 92372207 and 92572106) and Zhejiang Province Natural Science Foundation (LQN25E020003).
Author information
Authors and Affiliations
Contributions
Y.Y. conceived the idea of the smart electrolyte. C.Y., W.H. and M.Z. processed and analysed the experimental data and prepared the figures. X.L. performed the calculation part of the work. X.Z. and D.X. conducted the assembly and testing of the pouch cells. X.Z., Y.Z. and M.W. investigated the mechanism of electrolyte polymerization. Y.Y. and J.L. supervised the research. Y.Y. and J.L. wrote the paper. M.Z., J.Y. and W.C. helped revise the paper. All authors discussed the results and commented on the paper writing. C.Y., W.H. and M.Z. contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Jie Sun and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–36, discussion and Note 1.
Source data
Source Data Fig. 2c,e (download XLSX )
Unprocessed data of NMR and the ternary phase diagram.
Source Data Fig. 4b,c (download XLSX )
Unprocessed data of temperature–time curves and dT/dt–T curves from ARC tests.
Source Data Fig. 5b (download XLSX )
Calculation data of the activation energy (∆E) of various polymerization types.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, C., Hu, W., Zheng, M. et al. Ultrafast thermo-responsive electrolyte for enhanced safety in lithium metal batteries. Nat Energy 10, 1493–1502 (2025). https://doi.org/10.1038/s41560-025-01905-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41560-025-01905-7
This article is cited by
-
Solidifying safety on cue
Nature Energy (2025)