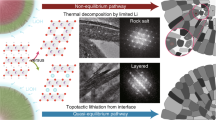
Abstract
Ni-rich layered oxide cathodes for lithium-ion batteries exhibit chemomechanical failures, with the consensus attributing this to high-voltage phase transitions. Existing mitigation strategies rely on compositional modifications (for example, doping), nanostructuring (for example, coatings and primary-particle engineering) and microstructure modifications, but these approaches increase synthesis complexity. Here, we demonstrate a simple synthesis strategy that enables exceptionally stable Ni-rich cathodes without doping, coating or concentration gradients. We show that chemomechanical failure is closely linked to microstructural non-uniformity (specifically, nanoscale pores), stemming from limited contact between solid-state reactants during calcination. By increasing the LiOH melting rate, we enhance liquid–solid interfacial contact between precursors, resulting in uniformly evolved microstructures. This uniformity leads to excellent cycle life by dissipating strain energy and mitigating chemomechanical failure even in the presence of high-voltage phase transition. Our findings challenge the prevailing belief that suppressing this phase transition and hierarchal material design are necessary for stable Ni-rich cathodes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information. Additional data in this paper are available via Dryad at https://doi.org/10.5061/dryad.mpg4f4rf3 (ref. 47).
References
Li, W., Erickson, E. M. & Manthiram, A. High-nickel layered oxide cathodes for lithium-based automotive batteries. Nat. Energy 5, 26–34 (2020).
Yang, J., Liang, X., Ryu, H. H., Yoon, C. S. & Sun, Y. K. Ni-rich layered cathodes for lithium-ion batteries: from challenges to the future. Energy Storage Mater. 63, 102969 (2023).
Manthiram, A., Knight, J. C., Myung, S. T., Oh, S. M. & Sun, Y. K. Nickel-rich and lithium-rich layered oxide cathodes: progress and perspectives. Adv. Energy Mater. 6, 1501010 (2016).
Bianchini, M. et al. There and back again—the journey of LiNiO2 as a cathode active material. Angew. Chem. Int. Ed. 58, 10434–10458 (2019).
Noh, H. J., Youn, S., Yoon, C. S. & Sun, Y. K. Comparison of the structural and electrochemical properties of layered Li[NixCoyMnz]O2 (x= 1/3, 0.5, 0.6, 0.7, 0.8 and 0.85) cathode material for lithium-ion batteries. J. Power Sources 233, 121–130 (2013).
Li, W., Asl, H. Y., Xie, Q. & Manthiram, A. Collapse of LiNi1–x–yCoxMnyO2 lattice at deep charge irrespective of nickel content in lithium-ion batteries. J. Am. Chem. Soc. 141, 5097–5101 (2019).
Ryu, H. H., Park, K. J., Yoon, C. S. & Sun, Y. K. Capacity fading of Ni-rich Li[NixCoyMn1–x–y]O2 (0.6≤ x≤ 0.95) cathodes for high-energy-density lithium-ion batteries: bulk or surface degradation?. Chem. Mater. 30, 1155–1163 (2018).
Lee, S., Su, L., Mesnier, A., Cui, Z. & Manthiram, A. Cracking vs. surface reactivity in high-nickel cathodes for lithium-ion batteries. Joule 7, 2430–2444 (2023).
Su, J., Raabe, D. & Li, Z. Hierarchical microstructure design to tune the mechanical behavior of an interstitial TRIP-TWIP high-entropy alloy. Acta Mater. 163, 40–54 (2019).
Tsang, M. Y. et al. Multilength scale hierarchy in metal-organic frameworks: synthesis, characterization and the impact on applications. Adv. Funct. Mater. 34, 2308376 (2024).
Sun, Y. et al. A breathable inorganic–organic interface for fabricating a crack-free nickel-rich cathode with long-term stability. Energy Environ. Sci. 17, 5124–5136 (2024).
Wang, L., Liu, T., Wu, T. & Lu, J. Strain-retardant coherent perovskite phase stabilized Ni-rich cathode. Nature 611, 61–67 (2022).
Zhao, C. et al. Suppressing strain propagation in ultrahigh-Ni cathodes during fast charging via epitaxial entropy-assisted coating. Nat. Energy 9, 345–356 (2024).
Kim, U.-H. et al. Heuristic solution for achieving long-term cycle stability for Ni-rich layered cathodes at full depth of discharge. Nat. Energy 5, 860–869 (2020).
Park, G.-T. et al. Introducing high-valence elements into cobalt-free layered cathodes for practical lithium-ion batteries. Nat. Energy 7, 946–954 (2022).
Kim, D.-H. et al. Stepwise dopant selection process for high-nickel layered oxide cathodes. Adv. Energy Mater. 12, 2200136 (2022).
Zhang, R. et al. Compositionally complex doping for zero-strain zero-cobalt layered cathodes. Nature 610, 67–73 (2022).
Sun, Y. K. et al. Nanostructured high-energy cathode materials for advanced lithium batteries. Nat. Mater. 11, 942–947 (2012).
Liu, T. et al. Rational design of mechanically robust Ni-rich cathode materials via concentration gradient strategy. Nat. Commun. 12, 6024 (2021).
Liang, L. et al. High-entropy doping promising ultrahigh-Ni Co-free single-crystalline cathode toward commercializable high-energy lithium-ion batteries. Sci. Adv. 10, eado4472 (2024).
Langdon, J. & Manthiram, A. A perspective on single-crystal layered oxide cathodes for lithium-ion batteries. Energy Storage Mater. 37, 143–160 (2021).
Yoon, M. et al. Eutectic salt-assisted planetary centrifugal deagglomeration for single-crystalline cathode synthesis. Nat. Energy 8, 482–491 (2023).
Busse, G. M. et al. Calcination heterogeneity in Li-rich layered oxides: a systematic study of Li2CO3 particle size. Chem. Mater. 35, 10658–10671 (2023).
Jo, S. et al. Solid-state reaction heterogeneity during calcination of lithium-ion battery cathode. Adv. Mater. 35, 2370067 (2023).
Bianchini, M., Fauth, F., Hartmann, P., Brezesinski, T. & Janek, J. An in situ structural study on the synthesis and decomposition of LiNiO2. J. Mater. Chem. A 8, 1808–1820 (2020).
Zuo, W. et al. Microstrain screening towards defect-less layered transition metal oxide cathodes. Nat. Nanotechnol. 19, 1644–1653 (2024).
Jun, Y. S. et al. Classical and nonclassical nucleation and growth mechanisms for nanoparticle formation. Annu. Rev. Phys. Chem. 73, 453–477 (2022).
Li, Z. et al. Gradient-porous-structured Ni-rich layered oxide cathodes with high specific energy and cycle stability for lithium-ion batteries. Nat. Commun. 15, 10216 (2024).
Park, H. et al. In situ multiscale probing of the synthesis of a Ni-rich layered oxide cathode reveals reaction heterogeneity driven by competing kinetic pathways. Nat. Chem. 14, 614–622 (2022).
Song, S. H. et al. Toward a nanoscale-defect-free Ni-rich layered oxide cathode through regulated pore evolution for long-lifespan Li rechargeable batteries. Adv. Funct. Mater. 34, 2306654 (2024).
Bianchini, M. et al. The interplay between thermodynamics and kinetics in the solid-state synthesis of layered oxides. Nat. Mater. 19, 1088–1095 (2020).
Kim, Y. & Lim, J. Exploring spectroscopic X-ray nano-imaging with Zernike phase contrast enhancement. Sci. Rep. 12, 2894 (2022).
Liu, Z. et al. Approaching ultimate synthesis reaction rate of Ni-rich layered cathodes for lithium-ion batteries. Nanomicro Lett. 16, 1–11 (2024).
Zhu, W. et al. Ultrafast non-equilibrium synthesis of cathode materials for Li-ion batteries. Adv. Mater. 35, 2208974 (2023).
Xu, Z. et al. Charge distribution guided by grain crystallographic orientations in polycrystalline battery materials. Nat. Commun. 11, 83 (2020).
Hou, D. et al. Effect of the grain arrangements on the thermal stability of polycrystalline nickel-rich lithium-based battery cathodes. Nat. Commun. 13, 3437 (2022).
Park, G.-T. et al. Ultrafine-grained Ni-rich layered cathode for advanced Li-ion batteries. Energy Environ. Sci. 14, 6616–6626 (2021).
Shen, F., Dixit, M. B., Xiao, X. & Hatzell, K. B. Effect of pore connectivity on Li dendrite propagation within LLZO electrolytes observed with synchrotron X-ray tomography. ACS Energy Lett. 3, 1056–1061 (2018).
Diallo, M. S. et al. Effect of solid-electrolyte pellet density on failure of solid-state batteries. Nat. Commun. 15, 858 (2024).
Kalnaus, S., Dudney, N. J., Westover, A. S., Herbert, E. & Hackney, S. Solid-state batteries: the critical role of mechanics. Science 381, eabg5998 (2023).
Wang, L. et al. Grain morphology and microstructure control in high-stable Ni-rich layered oxide cathodes. Adv. Funct. Mater. 33, 2212849 (2023).
Jung, C.-H. et al. New Insight into microstructure engineering of Ni-rich layered oxide cathode for high performance lithium ion batteries. Adv. Funct. Mater. 31, 2010095 (2021).
Jung, C.-H., Shim, H., Eum, D. & Hong, S. H. Challenges and recent progress in LiNixCoyMn1−x−yO2 (NCM) cathodes for lithium ion batteries. J. Korean Ceram. Soc. 58, 1–27 (2021).
Jo, S. et al. Nanoscale projection hard X-ray microscope for operando statistical analysis of chemical heterogeneity in lithium-ion battery cathodes. Small Methods 9, 2401087 (2025).
Jo, S., Kim, S. & Lim, J. TXM-Pal: a companion software for advanced data processing in spectroscopic X-ray microscopy. J. Synchrotron Radiat. 32, 815–822 (2025).
McCloskey, B. D., Bethune, D. S., Shelby, R. M., Girishkumar, G. & Luntz, A. C. Solvents’ critical role in nonaqueous lithium–oxygen battery electrochemistry. J. Phys. Chem. Lett. 2, 1161–1166 (2011).
Eum, D. Data from: Uniform pore structure enables negligible degradation in undoped and uncoated Ni-rich cathodes. Dryad https://doi.org/10.5061/dryad.mpg4f4rf3 (2026).
Acknowledgements
The battery aspect of this work was supported by the Assistant Secretary for Critical Minerals & Energy Innovation, Transportation Technologies Office, Battery Materials Research Program, US Department of Energy (DOE). The synthesis aspect of this work was supported by BASF Corporation through the California Research Alliance. X-ray imaging work was supported by the US Air Force Office Multidisciplinary University Research Initiative (MURI) programme under grant no. FA9550-23-1-0281. TGA-MS work was supported by the Regional Innovation System & Education (RISE) programme funded by the Ministry of Education (MOE) and the Jeollanamdo, Republic of Korea (grant no. 2025-RISE-14-003).
This research used resources of the National Synchrotron Light Source II, which is a DOE Office of Science User Facility under contract no. DE-SC0012704. Use of the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, was supported by the US Department of Energy (DOE), Office of Science, Office of Basic Energy Sciences under contract no. DE-AC02-76SF00515. This research also used resources of the Pohang Light Source II at the Pohang Accelerator Laboratory, Republic of Korea. Part of this work was performed at the Stanford Nano Shared Facilities (SNSF), supported by the National Science Foundation under grant no. ECCS-2026822. We thank P. Wallace for assistance with the Helios Hydra PFIB-SEM and S. Yoo for assistance with TXM/XNI image processing.
Author information
Authors and Affiliations
Contributions
D.E. and H.R. conceived the original idea, and D.E., H.R., T.S., Y.L. and W.C.C. designed the research. D.E. performed electrochemical tests and characterizations, such as OM, TXM, XNI and XRD. H.R. synthesized the materials and collected SEM, XANES and XRD data. T.S. analysed and processed the datasets of OM, TXM and XNI. S.S.L. conducted PFIM-SEM and interpreted the mechanical properties of the materials. Z.J. and Y.-K.T. contributed to XRD and OM experiments, respectively. H.P. provided constructive advice for experimental design. J.L. and B.D.M. conducted DEMS experiments. K.J. and S.J. carried out TGA-MS analysis and PXM measurements, respectively. E.P.K.L.C performed EIS measurements and fitting. N.B.L. and J.-H.L. offered valuable feedback throughout the project. D.E., H.R., T.S., Y.L. and W.C.C. wrote the manuscript, and D.E., Y.L. and W.C.C. supervised all aspects of the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Reviewer Recognition
Nature Energy thanks Lianzhou Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Reaction heterogeneity across multiple particles at 440 °.
a,b, PXM images showing spatial distribution of the Ni white-line energy across multiple particles quenched at 440 °C under heating rates of (a) 2 °C/min and (b) 10 °C/min.
Extended Data Fig. 3 Primary-particle morphology and size comparison in the pristine state.
a,b, Cross-sectional SEM images of the (a) 2 and (b) 10 °C/min electrodes in the pristine state, each shown as a pair of original and watershed-segmented images. c,d, Histograms of equivalent primary-particle sizes for the (c) 2 and (d) 10 °C/min samples, with mean particle sizes indicated.
Extended Data Fig. 4 Microstructural degradation across multiple particles after 100 cycles.
a,b, Cross-sectional SEM images of the (a) 2 and (b) 10 °C/min electrodes after 100 cycles. Scale bar, 10 µm.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–5, Figs. 1–28, Tables 1 and 2 and References.
Supplementary Video 1 (download MP4 )
2 °C per min.
Supplementary Video 2 (download MP4 )
10 °C per min.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Eum, D., Ramachandran, H., Sun, T. et al. Uniform pore structure enables negligible degradation in undoped and uncoated Ni-rich cathodes. Nat Energy (2026). https://doi.org/10.1038/s41560-026-01988-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41560-026-01988-w