Abstract
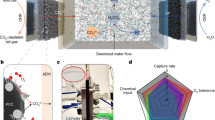
Electrified CO2 capture and release from air offers net-negative emissions, but today’s liquid-carbonate-based systems have a high energy cost (6–10 GJ per ton of CO2), and organic sorbents are oxygen sensitive. Here we report electrified CO2 surface mineralization/demineralization capture/release, wherein an inorganic capture sorbent, MnO2, is electrochemically reduced/activated to generate Mn(III), which mineralizes CO2 to form MnOOCO2H (operando Raman); the process is reversed under oxidative potential. This approach is built upon Mn redox reaction that resides within the water-stable bracket, offering tunable driving force (kinetics/productivity) with applied potential (energy). After optimizing the electrochemical protocol, we capture from air (0.04% CO2 and 21% O2) at 4.1 GJ per ton of CO2, with capacity and kinetics comparable to prior sorbents, low sensitivity to oxygen/humidity, 80% single-pass CO2 capture ratio and release under a pure CO2 carrier gas stream and pressure drop <150 Pa. The system operates >1,000 h with >90% capacity retention and scales to 20 cm2 without loss; remaining challenges include material utilization, electrolyte, gas flow/pressure drop and CO2-purity management.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that all data supporting the major findings are available in the main text or the Supplementary Information. Source data are provided with this paper.
References
Direct Air Capture 2022 (IEA, 2022).
Net Zero by 2050 (IEA, 2021).
Voskian, S. & Hatton, T. A. Faradaic electro-swing reactive adsorption for CO2 capture. Energy Environ. Sci. 12, 3530–3547 (2019).
Rahimi, M. et al. Carbon dioxide capture using an electrochemically driven proton concentration process. Cell Rep. Phys. Sci.1, 100033 (2020).
Sabatino, F., Gazzani, M., Gallucci, F. & van Sint Annaland, M. Modeling, optimization, and techno-economic analysis of bipolar membrane electrodialysis for direct air capture processes. Ind. Eng. Chem. Res. 61, 12668–12679 (2022).
Seo, H., Rahimi, M. & Hatton, T. A. Electrochemical carbon dioxide capture and release with a redox-active amine. J. Am. Chem. Soc. 144, 2164–2170 (2022).
Pang, S. et al. A phenazine-based high-capacity and high-stability electrochemical CO2 capture cell with coupled electricity storage. Nat. Energy 8, 1126–1136 (2023).
Jin, S., Wu, M., Gordon, R. G., Aziz, M. J. & Kwabi, D. G. pH swing cycle for CO2 capture electrochemically driven through proton-coupled electron transfer. Energy Environ. Sci. 13, 3706–3722 (2020).
Huebscher, R. G. & Babinsky, A. D. Electrochemical concentration and separation of carbon dioxide for advanced life support systems—carbonation cell system. SAE Trans. 78, 2164–2170 (1969).
Zhu, P. et al. Continuous carbon capture in an electrochemical solid-electrolyte reactor. Nature 618, 959–966 (2023).
Vermaas, D. A., Wiegman, S., Nagaki, T. & Smith, W. A. Ion transport mechanisms in bipolar membranes for (photo)electrochemical water splitting. Sustain. Energy Fuels 2, 2006–2015 (2018).
Liu, Y., Ye, H.-Z., Diederichsen, K. M., Van Voorhis, T. & Hatton, T. A. Electrochemically mediated carbon dioxide separation with quinone chemistry in salt-concentrated aqueous media. Nat. Commun. 11, 2278 (2020).
Jeziorek, D. et al. Theoretical and electrochemical study of the mechanism of anthraquinone-mediated one-electron reduction of oxygen: the involvement of adducts of dioxygen species to anthraquinones. J. Chem. Soc. Perkin Trans. 2, 229–236 (1997).
Lindskog, S. Structure and mechanism of carbonic anhydrase. Pharmacol. Ther. 74, 1–20 (1997).
Wilcox, J. Carbon Capture (Springer Science & Business Media, 2012).
Singh, A. K. et al. Electrochemical stability of metastable materials. Chem. Mater. 29, 10159–10167 (2017).
Persson, K. A., Waldwick, B., Lazic, P. & Ceder, G. Prediction of solid-aqueous equilibria: scheme to combine first-principles calculations of solids with experimental aqueous states. Phys. Rev. B 85, 235438 (2012).
Jain, A. et al. Commentary: the materials project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Patel, A. M., Nørskov, J. K., Persson, K. A. & Montoya, J. H. Efficient Pourbaix diagrams of many-element compounds. Phys. Chem. Chem. Phys. 21, 25323–25327 (2019).
Drage, T. C. et al. Materials challenges for the development of solid sorbents for post-combustion carbon capture. J. Mater. Chem. 22, 2815–2823 (2012).
Min, Y. J., Ganesan, A., Realff, M. J. & Jones, C. W. Direct air capture of CO2 using poly(ethyleneimine)-functionalized expanded poly(tetrafluoroethylene)/silica composite structured sorbents. ACS Appl. Mater. Interfaces 14, 40992–41002 (2022).
Li, H. et al. Capturing carbon dioxide from air with charged-sorbents. Nature 630, 654–659 (2024).
Seo, H. & Hatton, T. A. Electrochemical direct air capture of CO2 using neutral red as reversible redox-active material. Nat. Commun. 14, 313 (2023).
Zhou, Z. et al. Carbon dioxide capture from open air using covalent organic frameworks. Nature 635, 96–101 (2024).
Boualavong, J., Gorski, C. A. & Liu, Y. Translatable reporting of energy demand and rates in electrochemical carbon capture. iScience 28, 111781 (2025).
Simeon, F. et al. Electrochemical and molecular assessment of quinones as CO2-binding redox molecules for carbon capture. J. Phys. Chem. C 126, 1389–1399 (2022).
Wang, X. & Song, C. Carbon capture from flue gas and the atmosphere: a perspective. Front. Energy Res. https://doi.org/10.3389/fenrg.2020.560849 (2020).
Socolow, R. et al. Direct Air Capture of CO2 with Chemicals: A Technology Assessment for the APS Panel on Public Affairs (American Physical Society, 2011).
Barlow, J. M. & Yang, J. Y. Oxygen-stable electrochemical CO2 capture and concentration with quinones using alcohol additives. J. Am. Chem. Soc. 144, 14161–14169 (2022).
Wiegner, J. F., Grimm, A., Weimann, L. & Gazzani, M. Optimal design and operation of solid sorbent direct air capture processes at varying ambient conditions. Ind. Eng. Chem. Res. 61, 12649–12667 (2022).
Keith, D. W., Holmes, G., St. Angelo, D. & Heidel, K. A process for capturing CO2 from the atmosphere. Joule 2, 1573–1594 (2018).
Wurzbacher, J. A., Gebald, C., Brunner, S. & Steinfeld, A. Heat and mass transfer of temperature–vacuum swing desorption for CO2 capture from air. Chem. Eng. J. 283, 1329–1338 (2016).
Choi, S., Gray, M. L. & Jones, C. W. Amine-tethered solid adsorbents coupling high adsorption capacity and regenerability for CO2 capture from ambient air. ChemSusChem 4, 628–635 (2011).
Shi, X., Lin, Y. & Chen, X. Development of sorbent materials for direct air capture of CO2. MRS Bull. 47, 405–415 (2022).
Song, M. et al. Amine-functionalized MIL-101(Cr) fibers for direct air capture at cold temperatures using rapid temperature vacuum swing adsorption. Chem. Eng. J. 477, 147135 (2023).
Rahimi, M., Khurram, A., Hatton, T. A. & Gallant, B. Electrochemical carbon capture processes for mitigation of CO2 emissions. Chem. Soc. Rev. 51, 8676–8695 (2022).
Spiteri, A., Megerle, B., Calbry-Muzyka, A., Casas, N. & Wurzbacher, J. A. Method for capture of carbon dioxide from ambient air and corresponding adsorber structures with a plurality of parallel surfaces. US patent WO2021239747A1 (2021).
Ozkan, M. Atmospheric alchemy: the energy and cost dynamics of direct air carbon capture. MRS Energy Sustain. 12, 46–61 (2025).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Sundararaman, R. et al. JDFTx: software for joint density-functional theory. SoftwareX 6, 278–284 (2017).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Sundararaman, R. & Goddard, W. A. III. The charge-asymmetric nonlocally determined local-electric (CANDLE) solvation model. J. Chem. Phys. 142, 064107 (2015).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
This work made use of the Northwestern University Micro/Nano Fabrication Facility (NUFAB) facility of Northwestern University’s Atomic and Nanoscale Characterization Experimental Center (NUANCE), which has received support from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (National Science Foundation, NSF ECCS-2025633), the International Institute for Nanotechnology (IIN) and Northwestern’s Materials Research Science and Engineering Center (MRSEC) programme (NSF DMR-2308691). W.A.G. thanks Department of Energy (DOE) (SC-0022230) for support. DFT calculations were also supported through computational resources and staff contributions provided for the Quest high-performance computing facility at Northwestern University, which is jointly supported by the Office of the Provost, the Office of Research and Northwestern University Information Technology. Funding: this work was financially supported by TotalEnergies SE.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.X., E.H.S., Z.L. and B.P. Materials synthesis, device development, testing methodology development and experimental validation: Z.L. and B.P. Materials structural characterization: B.P. Raman experiments design, measurement and interpretation: H.Z. Simulation studies: C.B.M. Adsorption isotherm characterization: M.K.S. Supplementary three-electrode investigations: Z.L., H.S.J. and H.L. Supervision: W.A.G., K.O.K., O.K.F., K.X. and E.H.S. Funding acquisition: O.K.F. and E.H.S. Writing–original draft: Z.L., B.P. and H.Z. Writing–reviewing and editing: Z.L., B.P., M.K.S., H.L., K.O.K, O.K.F., K.X. and E.H.S.
Corresponding authors
Ethics declarations
Competing interests
Z.L., K.X. and E.H.S. are filling a patent based on this work. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1 and 2, Figs. 1–23 and Tables 1–3.
Supplementary Data (download XLSX )
Source data for Supplementary Figs. 11c, 19, 20d–e and 21c.
Source data
Source Data Fig. 3 (download XLSX )
Statistical source data for averaged results.
Source Data Fig. 4 (download XLSX )
Statistical source data for averaged results.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Z., Ze, H., Peng, B. et al. Electrified reversible surface mineralization of CO2 for direct air capture. Nat Energy (2026). https://doi.org/10.1038/s41560-026-01989-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41560-026-01989-9