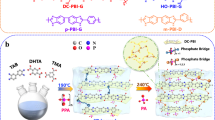
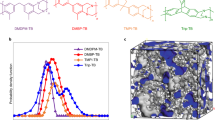
Abstract
High-temperature polymer electrolyte membrane fuel cells offer inherent impurity tolerance and simplified thermal/water management. However, conventional phosphoric acid-doped membranes must be relatively thick (typically >50 μm) to counteract H3PO4-induced mechanical degradation, leading to ohmic losses that limit fuel cell performance. Here we report a 20-μm-thin, mechanically robust phosphoric acid-doped membrane incorporating dynamic Cu-ion crosslinking for high-power-density high-temperature polymer electrolyte membrane fuel cells. The dynamic Cu-polymer coordination establishes dynamic crosslinking networks that provide exceptional toughness and extensibility, alongside spontaneous self-healing capability. Simultaneously, the Cu ions improve H3PO4 retention and proton dissociation through electrostatic interactions and polarization of O–H bonds in H3PO4 molecules. The resulting thin membranes exhibit minimal ohmic resistance (0.06 Ω cm2) while maintaining low H2 crossover current density (0.95 mA cm−2). Fuel cells incorporating this membrane achieved a peak power density of 3.08 W cm−2 at 200 °C (H2/O2), with negligible degradation over 503 h at 1.0 A cm−2 and 160 °C.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Source data are provided with this paper.
References
Yang, J. et al. Oxygen- and proton-transporting open framework ionomer for medium-temperature fuel cells. Science 385, 1115–1120 (2024).
Lim, K. H. et al. Protonated phosphonic acid electrodes for high power heavy-duty vehicle fuel cells. Nat. Energy 7, 248–259 (2022).
Cullen, D. A. et al. New roads and challenges for fuel cells in heavy-duty transportation. Nat. Energy 6, 462–474 (2021).
Jang, S. et al. Multiscale architectured membranes, electrodes, and transport layers for next-generation polymer electrolyte membrane fuel cells. Adv. Mater. 35, 2370311 (2023).
Liu, Z. et al. Pt catalyst protected by graphene nanopockets enables lifetimes of over 200,000 h for heavy-duty fuel cell applications. Nat. Nanotechnol. 20, 807–814 (2025).
Atanasov, V. et al. Synergistically integrated phosphonated poly(pentafluorostyrene) for fuel cells. Nat. Mater. 20, 370–377 (2020).
Li, W. et al. Dual-proton conductor for fuel cells with flexible operational temperature. Adv. Mater. 36, 2310584 (2024).
Zhang, Z. N. et al. Conquering heavy-loading decay of high-temperature polymer electrolyte membrane fuel cells with a conductive polymer-based phosphoric acid reservoir. ACS Energy Lett. 9, 2257–2264 (2024).
Liu, L. et al. Supramolecular complexation of metal oxide cluster and non-fluorinated polymer for large-scale fabrication of proton exchange membranes for high-power-density fuel cells. Angew. Chem. Int. Ed. 63, e202318355 (2024).
Zhang, L. et al. Double cross-linked 3D layered PBI proton exchange membranes for stable fuel cell performance above 200 °C. Nat. Commun. 15, 3409 (2024).
Cohen, L. A. et al. How low can you go? Nanoscale membranes for efficient water electrolysis. ACS Energy Lett. 9, 1624–1632 (2024).
Chen, Y. et al. Feasibility of using thin polybenzimidazole electrolytes in high-temperature proton exchange membrane fuel cells. Int. J. Hydrogen Energy 47, 28615–28625 (2022).
Zhang, J. et al. Proton conductor confinement strategy for polymer electrolyte membrane assists fuel cell operation in wide-range temperature. Adv. Funct. Mater. 33, 2214097 (2023).
Tang, H. et al. Fuel cells with an operational range of −20 °C to 200 °C enabled by phosphoric acid-doped intrinsically ultramicroporous membranes. Nat. Energy 7, 153–162 (2022).
Zong, X. et al. Quaternized-PAF architecture mediated proton channels to enhance ultra-robust operation for 200 °C proton exchange membrane fuel cells. Angew. Chem. Int. Ed. 64, e202509085 (2025).
Pang, X. et al. Confining phosphoric acid in quaternized COF channels for ultra-stable and fast anhydrous proton transport. Angew. Chem. Int. Ed. 64, e202423458 (2025).
Lee, S. et al. Self-assembled network polymer electrolyte membranes for application in fuel cells at 250 °C. Nat. Energy 9, 849–861 (2024).
Liu, X. et al. Ultrathin membranes prepared through interfacial polymer cross-linking for selective and fast ion transport. Nat. Chem. Eng. 2, 369–378 (2025).
Kissane, J., Pasaogullari, U. & Bliznakov, S. Novel strategy for engineering ultra-thin proton exchange membranes for advanced PEM fuel cells. ECS Meet. Abstr. MA2023-01, 2274 (2023).
Chen, Y. et al. Reinforced polybenzimidazole membranes by crosslinked fiber mats for high temperature polymer electrolyte membrane fuel cells. Electrochim. Acta 529, 146260 (2025).
Aili, D. et al. From polybenzimidazoles to polybenzimidazoliums and polybenzimidazolides. J. Mater. Chem. A 8, 12854–12886 (2020).
Tang, H. et al. Phosphoric-acid retention in high-temperature proton-exchange membranes. Chem-Eur J 28, 70 (2022).
Seselj, N. et al. Performance degradation and mitigation of high temperature polybenzimidazole-based polymer electrolyte membrane fuel cells. Chem. Soc. Rev. 52, 4046–4070 (2023).
Liu, G. et al. Soluble ultra-high molecular weight poly(4,4′-diphenylether-5,5′-bibenzimidazole) based membranes with remarkable mechanical strength and specific proton conductivity for high temperature proton exchange membrane fuel cells. J. Membr. Sci. 693, 122348 (2024).
Liu, T. et al. Super strong and tough polybenzimidazole/metal ions coordination networks: reinforcing mechanism, recyclability, and anti-counterfeiting applications. Macromol. Rapid Commun. 43, 2100643 (2021).
Wu, J. et al. Metal-coordinated polybenzimidazole membranes with preferential K+ transport. Nat. Commun. 14, 1149 (2023).
Yin, Q. et al. Construction of polyimide films with excellent dimensional stability and toughness via incorporating point-to-face multi-coordination structure. Composites, Part B 208, 108566 (2021).
Wu, J. et al. Dendrite-free zinc-based battery with high areal capacity via the region-induced deposition effect of Turing membrane. J. Am. Chem. Soc. 143, 13135–13144 (2021).
Sannigrahi, A., Arunbabu, D., Sankar, R. M. & Jana, T. Aggregation behavior of polybenzimidazole in aprotic polar solvent. Macromolecules 40, 2844–2851 (2007).
Gan, F. et al. Preparation of polyimide-metal complexes with tunable sol-gel transition by introduction of Zn(II)-coordination. Polym. Test. 111, 107615 (2022).
Hinge, V. K. et al. XAFS study of mixed ligand benzimidazole copper complexes having distorted coordination geometry. J. Mol. Struct. 1289, 135909 (2023).
Mukherjee, D. et al. Intermediate temperature superprotonic conductivity beyond 10−2 S cm−1 with low proton transfer energy barrier in a metal-organic framework. ACS Energy Lett. 10, 1216–1228 (2025).
Sahoo, R., Pal, S. C. & Das, M. C. Solid-state proton conduction driven by coordinated water molecules in metal-organic frameworks and coordination polymers. ACS Energy Lett. 7, 4490–4500 (2022).
Gao, W. et al. Ozonated graphene oxide film as a proton-exchange membrane. Angew. Chem. Int. Ed. 53, 3588–3593 (2014).
Li, J. et al. Tetramethyl poly(aryl ether ketone) modified by DABCO cationic polymer for high temperature proton exchange membrane fuel cells. J. Membr. Sci. 703, 122848 (2024).
Suarez, S. et al. Multinuclear NMR study of the effect of acid concentration on ion transport in phosphoric acid doped poly(benzimidazole) membranes. J. Phys. Chem. B 116, 12545–12551 (2012).
Murdock, L. A. & Benicewicz, B. C. Teaching an old dog new tricks: synthesis, processing, and application of polybenzimidazole (PBI) membranes. ACS Appl. Energy Mater. 7, 239–249 (2023).
Liu, B., Liu, Q., Pang, Y., Duan, Y. & Zhao, C. Antioxidant sulfide-linked polymer membrane with inherent microporosity enables fuel cells to achieve outstanding power density and durability over a wide temperature range. Adv. Funct. Mater. 35, 2408291 (2024).
Luo, J. et al. Boosting the power density of the H3PO4/polybenzimidazole high-temperature proton exchange membrane fuel cell to >1.2 W cm−2 via the deposition of acid-based polymer layers on the catalyst layers. J. Power Sources 589, 233756 (2024).
Chen, S. et al. Spacer-engineering construction of continuous proton transport networks for cardo poly(biphenyl indole) high-temperature proton exchange membranes. J. Mater. Chem. A 12, 17243–17251 (2024).
Mi, H. et al. Imidazole and triazine framed porous aromatic framework with rich proton transport sites for high performance high-temperature proton exchange membranes. J. Membr. Sci. 712, 123250 (2024).
Liu, B. et al. Poly(triphenylene-piperidine) membranes reinforced by carboxy intrinsic microporous polymers towards high output power at low phosphoric acid levels for HT-PEMFC. J. Membr. Sci. 692, 122273 (2024).
Ju, Q. et al. Effect of solvent-free membranes-forming processes on HT-PEM properties of highly soluble polybenzimidazole. J. Membr. Sci. 692, 122264 (2024).
Guan, X. et al. High hydrogen-bond density polymeric ionic liquid composited high temperature proton exchange membrane with exceptional long-term fuel cell performance. J. Membr. Sci. 717, 123523 (2025).
You, X. et al. High conductivity poly(meta-terphenyl alkylene)s proton exchange membranes for high temperature fuel cell. Chem. Eng. J. 487, 150535 (2024).
Chen, J., Wang, L. & Wang, L. Highly conductive polybenzimidazole membranes at low phosphoric acid uptake with excellent fuel cell performances by constructing long-range continuous proton transport channels using a metal-organic framework (UIO-66). ACS Appl. Mater. Interfaces 12, 41350–41358 (2020).
Sun, X. et al. The impact of imidazolium with steric hindrance on the dissociation of phosphoric acid and the performance of high-temperature proton exchange membranes. J. Mater. Chem. A 12, 24499–24507 (2024).
Ransil, B. J. Studies in molecular structure. IV. Potential curve for the interaction of two helium atoms in single-configuration LCAO MO SCF approximation. J. Chem. Phys. 34, 2109–2118 (1961).
Lu, T. & Chen, Q. Shermo: a general code for calculating molecular thermochemistry properties. Comput. Theor. Chem. 1200, 113249 (2021).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Schauperl, M. et al. Non-bonded force field model with advanced restrained electrostatic potential charges (RESP2). Commun. Chem. 3, 44 (2020).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Brehm, M. & Kirchner, B. TRAVIS - a free analyzer and visualizer for Monte Carlo and molecular dynamics trajectories. J. Chem. Inf. Model. 51, 2007–2023 (2011).
Brehm, M., Thomas, M., Gehrke, S. & Kirchner, B. TRAVIS—a free analyzer for trajectories from molecular simulation. J. Chem. Phys. 152, 164105 (2020).
Acknowledgements
We would like to thank the National of Key R&D Program of China (number 2024YFE0207700 (S.L.)), the National Natural Science Foundation of China (number 22178012 (S.L.), U22A20419 (Y.X.), 22409011 (W. Li), 22572005 (Jin Zhang)). The programme was also sponsored by Beijing Nova Program (20220484140, (Jin Zhang)). We thank J. Xu and X. Wang from National Center for Magnetic Resonance in Wuhan, State Key Laboratory of Magnetic Resonance Spectroscopy and Imaging, Wuhan Institute of Physics and Mathematics, Innovation Academy for Precision Measurement Science and Technology, Chinese Academy of Sciences for providing assistance in NMR test and analysis. We also thank P. An and the 1W1B-XAFS Beamline of Beijing Synchrotron Radiation Facility for providing technical support and assistance in XAFS data collection. We acknowledge the facilities and the scientific and technical assistance of the Analysis and Testing Center, Beihang University.
Author information
Authors and Affiliations
Contributions
Z.Z. conceived the project, analysed the data and wrote the manuscript. Q.Z., Jialin Zhang and W. Li helped with data collection and assisted fuel cell measurements. W. Liu performed the DFT and MD calculations. Jin Zhang guided the research and helped with data collection. H.W. and Y.X. supervised and guided this work. S.L. proposed the concept, supervised the research and made final revisions to the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Vladimir Atanasov, Cheng-Hui Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–8, Figs. 1–52 and Tables 1–10.
Supplementary Data (download XLSX )
Source data for supplementary figures.
Supplementary Video 1 (download MP4 )
Supplemental Video 1
Supplementary Video 2 (download MP4 )
Supplemental Video 2
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Fig. 7 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Z., Zhang, Q., Li, W. et al. Thin membranes with Cu-ion crosslinking for high temperature polymer electrolyte membrane fuel cells. Nat Energy (2026). https://doi.org/10.1038/s41560-026-02049-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41560-026-02049-y