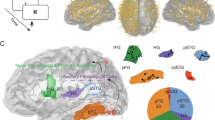
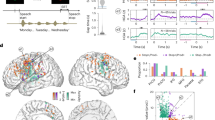
Abstract
Fluent speech production is mediated by serially ordering and preparing motor plans corresponding to target speech sounds, a process known as speech-motor sequencing. Here we used high-density direct cortical recordings while 14 participants spoke utterances with varying phonemic and syllabic sequence complexity after reading a target sequence and a delay period. Phasic activations corresponding to speech production and auditory feedback were observed, but also sustained neural activity that persisted throughout all task phases including the target presentation, the delay period and production of the sequence. Furthermore, sustained activity in a specific area, the middle precentral gyrus (mPrCG), was both modulated by sequence complexity and predicted reaction time, suggesting a role in speech-motor sequencing. Electrocortical stimulation of the mPrCG caused speech disfluencies resembling those seen in apraxia of speech. These results suggest that speech-motor sequencing is mediated by a distributed cortical network in which the mPrCG plays a central role.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data that support the main conclusions of the paper are available via Zenodo at https://doi.org/10.5281/zenodo.15346719 (ref. 78). Source data are provided with this paper. Specific statistical data is also provided in the Supplementary Data.
Code availability
Code for relevant analyses and figure creation is available via GitHub at https://github.com/ChangLabUcsf/Liu2025_Sequencing.
References
Levelt, W. J. M. Speaking: From Intention to Articulation (MIT, 1993).
Sternberg, S., Monsell, S., Knoll, R. L. & Wright, C. E. in Information Processing in Motor Control and Learning (ed. Stelmach, G. E.) 117–152 (Academic, 1978).
Lashley, K. S. in Cerebral Mechanisms in Behavior; The Hixon Symposium (ed. Jeffress, L. A.) 112–146 (Wiley, 1951).
Guenther, F. H. Neural Control of Speech (MIT, 2016).
Hickok, G. Computational neuroanatomy of speech production. Nat. Rev. Neurosci. 13, 135–145 (2012).
Van Der Merwe, A. New perspectives on speech motor planning and programming in the context of the four-level model and its implications for understanding the pathophysiology underlying apraxia of speech and other motor speech disorders. Aphasiology 35, 397–423 (2021).
Ogar, J., Slama, H., Dronkers, N., Amici, S. & Luisa Gorno-Tempini, M. Apraxia of speech: an overview. Neurocase 11, 427–432 (2005).
Strand, E. A., Duffy, J. R., Clark, H. M. & Josephs, K. The apraxia of speech rating scale: a tool for diagnosis and description of apraxia of speech. J. Commun. Disord. 51, 43–50 (2014).
Wertz, R. T., LaPointe, L. L. & Rosenbek, J. C. Apraxia of Speech in Adults: The Disorder and Its Management (Grune & Stratton, 1984).
Castellucci, G. A., Kovach, C. K., Howard, M. A., Greenlee, J. D. W. & Long, M. A. A speech planning network for interactive language use. Nature 602, 117–122 (2022).
Sahin, N. T., Pinker, S., Cash, S. S., Schomer, D. & Halgren, E. Sequential processing of lexical, grammatical, and phonological information within Broca’s area. Science 326, 445–449 (2009).
Chartier, J., Anumanchipalli, G. K., Johnson, K. & Chang, E. F. Encoding of articulatory kinematic trajectories in human speech sensorimotor cortex. Neuron 98, 1042–1054.e4 (2018).
Bouchard, K. E., Mesgarani, N., Johnson, K. & Chang, E. F. Functional organization of human sensorimotor cortex for speech articulation. Nature 495, 327–332 (2013).
Mugler, E. M. et al. Differential representation of articulatory gestures and phonemes in precentral and inferior frontal gyri. J. Neurosci. 38, 9803–9813 (2018).
Bohland, J. W. & Guenther, F. H. An fMRI investigation of syllable sequence production. NeuroImage 32, 821–841 (2006).
Svoboda, K. & Li, N. Neural mechanisms of movement planning: motor cortex and beyond. Curr. Opin. Neurobiol. 49, 33–41 (2018).
Hickok, G., Venezia, J. & Teghipco, A. Beyond Broca: neural architecture and evolution of a dual motor speech coordination system. Brain 146, 1775–1790 (2023).
Levelt, W. J. M., Roelofs, A. & Meyer, A. S. A theory of lexical access in speech production. Behav. Brain Sci. 22, 1–38 (1999).
Bohland, J. W., Bullock, D. & Guenther, F. H. Neural representations and mechanisms for the performance of simple speech sequences. J. Cogn. Neurosci. 22, 1504–1529 (2010).
Broca, P. P. Remarques Sur Le Siège de La Faculté Du Langage Articulé, Suivies d’une Observation d’aphémie (Perte de La Parole). Bull. Mém. Société Anat. Paris 6, 330–357 (1861).
Hillis, A. E. et al. Re-examining the brain regions crucial for orchestrating speech articulation. Brain 127, 1479–1487 (2004).
Castellucci, G. A. et al. Stimulation of caudal inferior and middle frontal gyri disrupts planning during spoken interaction. Curr. Biol. 34, 2719–2727 (2024).
Fedorenko, E. & Blank, I. A. Broca’s area is not a natural kind. Trends Cogn. Sci. 24, 270–284 (2020).
Mohr, J. P. et al. Broca aphasia: pathologic and clinical. Neurology 28, 311–311 (1978).
Andrews, J. P. Dissociation of Broca’s area from Broca’s aphasia in patients undergoing neurosurgical resections. J. Neurosurg. 138, 847–857 (2022).
Wilson, S. M. et al. Recovery from aphasia in the first year after stroke. Brain 146, 1021–1039 (2023).
Dronkers, N. F. A new brain region for coordinating speech articulation. Nature 384, 159–161 (1996).
Basilakos, A., Rorden, C., Bonilha, L., Moser, D. & Fridriksson, J. Patterns of poststroke brain damage that predict speech production errors in apraxia of speech and aphasia dissociate. Stroke 46, 1561–1566 (2015).
Graff-Radford, J. et al. The neuroanatomy of pure apraxia of speech in stroke. Brain Lang. 129, 43–46 (2014).
Levy, D. F. et al. Apraxia of speech with phonological alexia and agraphia following resection of the left middle precentral gyrus: illustrative case. J. Neurosurg. Case Lessons 5, CASE22504 (2023).
Chang, E. F. et al. Pure apraxia of speech after resection based in the posterior middle frontal gyrus. Neurosurgery 87, E383–E389 (2020).
Itabashi, R. et al. Damage to the left precentral gyrus is associated with apraxia of speech in acute stroke. Stroke 47, 31–36 (2016).
Josephs, K. A. et al. Characterizing a neurodegenerative syndrome: primary progressive apraxia of speech. Brain 135, 1522–1536 (2012).
Silva, A. B. et al. A neurosurgical functional dissection of the middle precentral gyrus during speech production. J. Neurosci. 42, 8416–8426 (2022).
Peeva, M. G. et al. Distinct representations of phonemes, syllables, and supra-syllabic sequences in the speech production network. NeuroImage 50, 626–638 (2010).
Ray, S. & Maunsell, J. H. R. Differences in gamma frequencies across visual cortex restrict their possible use in computation. Neuron 67, 885–896 (2010).
Steinschneider, M., Fishman, Y. I. & Arezzo, J. C. Spectrotemporal analysis of evoked and induced electroencephalographic responses in primary auditory cortex (A1) of the awake monkey. Cereb. Cortex 18, 610–625 (2008).
Leonard, M. K. et al. Large-scale single-neuron speech sound encoding across the depth of human cortex. Nature 626, 593–602 (2024).
Cheung, C., Hamilton, L. S., Johnson, K. & Chang, E. F. The auditory representation of speech sounds in human motor cortex. eLife 5, e12577 (2016).
Wilson, S. M., Saygin, A. P., Sereno, M. I. & Iacoboni, M. Listening to speech activates motor areas involved in speech production. Nat. Neurosci. 7, 701–702 (2004).
Dichter, B. K., Breshears, J. D., Leonard, M. K. & Chang, E. F. The control of vocal pitch in human laryngeal motor cortex. Cell 174, 21–31.e9 (2018).
Eichert, N., Papp, D., Mars, R. B. & Watkins, K. E. Mapping human laryngeal motor cortex during vocalization. Cereb. Cortex 30, 6254–6269 (2020).
Penfield, W. & Boldrey, E. Somatic motor and sensory representation in the cerebral cortex of man as studied by electrical stimulation. Brain 60, 389–443 (1937).
Leonard, M. K., Cai, R., Babiak, M. C., Ren, A. & Chang, E. F. The peri-Sylvian cortical network underlying single word repetition revealed by electrocortical stimulation and direct neural recordings. Brain Lang. 193, 58–72 (2019).
Lu, J. et al. Functional maps of direct electrical stimulation-induced speech arrest and anomia: a multicentre retrospective study. Brain 144, 2541–2553 (2021).
Rong, F., Isenberg, A. L., Sun, E. & Hickok, G. The neuroanatomy of speech sequencing at the syllable level. PLoS ONE 13, e0196381 (2018).
Churchland, M. M. & Shenoy, K. V. Delay of movement caused by disruption of cortical preparatory activity. J. Neurophysiol. 97, 348–359 (2007).
Duffy, J. R. Motor Speech Disorders: Substrates, Differential Diagnosis, and Management. (Elsevier Mosby, 2005).
Mailend, M.-L. & Maas, E. To lump or to split? Possible subtypes of apraxia of speech. Aphasiology 35, 592–613 (2021).
MacNeilage, P. F. The frame/content theory of evolution of speech production. Behav. Brain Sci. 21, 499–511 (1998).
Shattuck-Hufnagel, S. in The Production of Speech (ed. MacNeilage, P. F.) 109–136 (Springer, 1983).
Gordon, E. M. et al. A somato-cognitive action network alternates with effector regions in motor cortex. Nature 617, 351–359 (2023).
Long, M. A. et al. Functional segregation of cortical regions underlying speech timing and articulation. Neuron 89, 1187–1193 (2016).
Quigg, M., Geldmacher, D. S. & Elias, W. J. Conduction aphasia as a function of the dominant posterior perisylvian cortex: report of two cases. J. Neurosurg. 104, 845–848 (2006).
Cogan, G. B. et al. Manipulating stored phonological input during verbal working memory. Nat. Neurosci. 20, 279–286 (2017).
Miller, H. E. & Guenther, F. H. Modelling speech motor programming and apraxia of speech in the DIVA/GODIVA neurocomputational framework. Aphasiology 35, 424–441 (2021).
Binder, J. R. The Wernicke area: modern evidence and a reinterpretation. Neurology 85, 2170–2175 (2015).
Glasser, M. F. et al. A multi-modal parcellation of human cerebral cortex. Nature 536, 171–178 (2016).
Venezia, J. H., Richards, V. M. & Hickok, G. Speech-driven spectrotemporal receptive fields beyond the auditory cortex. Hear. Res. 408, 108307 (2021).
Kaestner, E. et al. The precentral gyrus contributions to the early time-course of grapheme-to-phoneme conversion. Neurobiol. Lang. 3, 18–45 (2022).
Dehaene, S. et al. Cerebral mechanisms of word masking and unconscious repetition priming. Nat. Neurosci. 4, 752–758 (2001).
Rapcsak, S. Z., Arthur, S. A. & Rubens, A. B. Lexical agraphia from focal lesion of the left precentral gyrus. Neurology 38, 1119–1119 (1988).
Segawa, J. A., Tourville, J. A., Beal, D. S. & Guenther, F. H. The neural correlates of speech motor sequence learning. J. Cogn. Neurosci. 27, 819–831 (2015).
Ozker, M., Doyle, W., Devinsky, O. & Flinker, A. A cortical network processes auditory error signals during human speech production to maintain fluency. PLoS Biol. 20, e3001493 (2022).
Chang, E. F., Kurteff, G. & Wilson, S. M. Selective interference with syntactic encoding during sentence production by direct electrocortical stimulation of the inferior frontal gyrus. J. Cogn. Neurosci. 30, 411–420 (2018).
Robinson, G. Dynamic aphasia: an inability to select between competing verbal responses? Brain 121, 77–89 (1998).
Parrell, B., Agnew, Z., Nagarajan, S., Houde, J. & Ivry, R. B. Impaired feedforward control and enhanced feedback control of speech in patients with cerebellar degeneration. J. Neurosci. 37, 9249–9258 (2017).
Lipski, W. J. et al. Subthalamic nucleus neurons encode syllable sequence and phonetic characteristics during speech. J. Neurophysiol. 132, 1382–1394 (2024).
Jordan, M. I. in Advances in Psychology Vol. 121 (ed. Donohoe, J. W.) 471–495 (Elsevier, 1997).
Averbeck, B. B., Chafee, M. V., Crowe, D. A. & Georgopoulos, A. P. Parallel processing of serial movements in prefrontal cortex. Proc. Natl Acad. Sci. USA 99, 13172–13177 (2002).
Kornysheva, K. et al. Neural competitive queuing of ordinal structure underlies skilled sequential action. Neuron 101, 1166–1180.e3 (2019).
Ariani, G., Kordjazi, N., Pruszynski, J. A. & Diedrichsen, J. The planning horizon for movement sequences. eNeuro 8, ENEURO.0085–21.2021 (2021).
Sanes, J. N. & Donoghue, J. P. Oscillations in local field potentials of the primate motor cortex during voluntary movement. Proc. Natl Acad. Sci. USA 90, 4470–4474 (1993).
Proix, T. et al. Imagined speech can be decoded from low- and cross-frequency intracranial EEG features. Nat. Commun. 13, 48 (2022).
Metzger, S. L. et al. Generalizable spelling using a speech neuroprosthesis in an individual with severe limb and vocal paralysis. Nat. Commun. 13, 6510 (2022).
Hamilton, L. S., Chang, D. L., Lee, M. B. & Chang, E. F. Semi-automated anatomical labeling and inter-subject warping of high-density intracranial recording electrodes in electrocorticography. Front. Neuroinform. 11, 62 (2017).
Hamilton, L. S., Edwards, E. & Chang, E. F. A spatial map of onset and sustained responses to speech in the human superior temporal gyrus. Curr. Biol. 28, 1860–1871.e4 (2018).
Liu, J. R., Zhao, L., Hullett, P. W. & Chang, E. F. Speech sequencing in the human precentral gyrus. Zenodo https://doi.org/10.5281/zenodo.15346719 (2025).
Acknowledgements
We thank all the participants who generously donated their time and effort to the collection of these data. We also thank the Chang Lab for helpful feedback and clinical support, in particular M. K. Leonard, D. F. Levy, A. B. Silva, Q. Greicius, C. Wang and M. P. Seaton, as well as R. C. Knowlton and J. K. Kleen. J.R.L. was supported by the National Institutes of Health grant no. U01 DC018671-01A1. L.Z. was supported by the National Institute on Deafness and Other Communication Disorders grant no. K99 DC020235. P.W.H. was supported by the National Center for Advancing Translational Sciences grant no. 5TL1TR001871-05.
Author information
Authors and Affiliations
Contributions
J.R.L. and E.F.C. conceptualized the research and designed the experiments. J.R.L., L.Z. and others collected the ECoG data. P.W.H. and J.R.L. collected the stimulation data. J.R.L. and L.Z. analysed the data. J.R.L., L.Z. and E.F.C. wrote the paper with input from all authors. E.F.C. supervised the project.
Corresponding author
Ethics declarations
Competing interests
J.R.L. and E.F.C. are inventors of pending provisional University of California, San Francisco patent applications relevant to neural decoding approaches. EFC is co-founder of Echo Neurotechnologies, LLC.
Peer review
Peer review information
Nature Human Behaviour thanks Anaïs Llorens, Nick Ramsey and Stephanie Ries for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–23 and Tables 1–8.
Supplementary Data (download XLSX )
Statistical source data for Supplementary Figs. 1 and 2, and summary electrode ROI counts.
Source data
Source Data Figs. 2, 3 and 5 (download XLSX )
Statistical source data for Figs. 2c, 3b, 3c, 5d and 5e.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J.R., Zhao, L., Hullett, P.W. et al. Speech sequencing in the human precentral gyrus. Nat Hum Behav 9, 2327–2344 (2025). https://doi.org/10.1038/s41562-025-02250-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41562-025-02250-1