Abstract
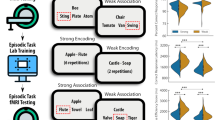
Although the distinction between episodic and semantic memory is supported by numerous neuropsychological studies, neuroimaging data have shown considerable overlap between regions that are activated during semantic and episodic remembering. This might indicate similar or shared mechanisms but might also result from inadequate task designs or poor functional magnetic resonance imaging signal coverage. Here we compared neural activations and representations associated with successful retrieval of episodic and semantic memories, using tasks that are more closely matched. A total of n = 40 participants recalled pairings between logos and brand names, where the pairings corresponded to real-world knowledge (semantic task) or were learned in an initial study phase (episodic task). Neither a priori-defined networks nor clusters generally activated by our task provided evidence for any difference between successful semantic and episodic retrieval, with the Bayes factor for the a priori networks supporting the null hypothesis of no difference. Protocol registration The Stage 1 protocol for this Registered Report was accepted in principle on 15 September 2021. The protocol, as accepted by the journal, can be found at https://osf.io/dm47y/.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Raw neuroimaging data, behavioural data and lab logs are available at https://openneuro.org/datasets/ds004495/. Processed data, pilot data and materials are available at https://osf.io/dm47y/.
Code availability
All code used for the study is available at https://osf.io/dm47y/.
References
Tulving, E. in Organization of Memory (eds Tulving, E. & Donaldson, W.) 381–403 (Academic Press, 1972).
Tulving, E. Memory and consciousness. Can. Psychol. Can. 26, 1–12 (1985).
Nebes, R. D., Martin, D. C. & Horn, L. C. Sparing of semantic memory in Alzheimer’s disease. J. Abnorm. Psychol. 93, 321–330 (1984).
Hodges, J. R. & Patterson, K. Is semantic memory consistently impaired early in the course of Alzheimer’s disease? Neuroanatomical and diagnostic implications. Neuropsychologia 33, 441–459 (1995).
Vargha-Khadem, F. et al. Differential effects of early hippocampal pathology on episodic and semantic memory. Science 277, 376–380 (1997).
Graham, K. S., Kropelnicki, A., Goldman, W. P. & Hodges, J. R. Two further investigations of autobiographical memory in semantic dementia. Cortex 39, 729–750 (2003).
McKinnon, M. C., Black, S. E., Miller, B., Moscovitch, M. & Levine, B. Autobiographical memory in semantic dementia: Implications for theories of limbic-neocortical interaction in remote memory. Neuropsychologia 44, 2421–2429 (2006).
Snowden, J., Griffiths, H. & Neary, D. Semantic dementia: autobiographical contribution to preservation of meaning. Cogn. Neuropsychol. 11, 265–288 (1994).
Westmacott, R. & Moscovitch, M. The contribution of autobiographical significance to semantic memory. Mem. Cognit. 31, 761–774 (2003).
Renoult, L., Irish, M., Moscovitch, M. & Rugg, M. D. From knowing to remembering: the semantic–episodic distinction. Trends Cogn. Sci. 23, 1041–1057 (2019).
King, D. R., Chastelaine, M. de, Elward, R. L., Wang, T. H. & Rugg, M. D. Recollection-related increases in functional connectivity predict individual differences in memory accuracy. J. Neurosci. 35, 1763–1772 (2015).
Thakral, P. P., Wang, T. H. & Rugg, M. D. Decoding the content of recollection within the core recollection network and beyond. Cortex 91, 101–113 (2017).
Binder, J. R. & Desai, R. H. The neurobiology of semantic memory. Trends Cogn. Sci. 15, 527–536 (2011).
Irish, M. & Vatansever, D. Rethinking the episodic–semantic distinction from a gradient perspective. Curr. Opin. Behav. Sci. 32, 43–49 (2020).
Irish, M. in The Cambridge Handbook of Imagination (ed. Abraham, A.) 447–465 (Cambridge Univ. Press, 2020).
Burianova, H. & Grady, C. L. Common and unique neural activations in autobiographical, episodic, and semantic retrieval. J. Cogn. Neurosci. 19, 1520–1534 (2007).
Burianova, H., McIntosh, A. R. & Grady, C. L. A common functional brain network for autobiographical, episodic, and semantic memory retrieval. NeuroImage 49, 865–874 (2010).
Rajah, M. N. & McIntosh, A. R. Overlap in the functional neural systems involved in semantic and episodic memory retrieval. J. Cogn. Neurosci. 17, 470–482 (2005).
Ryan, L., Cox, C., Hayes, S. M. & Nadel, L. Hippocampal activation during episodic and semantic memory retrieval: comparing category production and category cued recall. Neuropsychologia 46, 2109–2121 (2008).
Vatansever, D., Smallwood, J. & Jefferies, E. Varying demands for cognitive control reveals shared neural processes supporting semantic and episodic memory retrieval. Nat. Commun. 12, 2134 (2021).
Humphreys, G. F., Jung, J. & Lambon Ralph, M. A. The convergence and divergence of episodic and semantic functions across lateral parietal cortex. Cereb. Cortex 32, 5664–5681 (2022).
Nyberg, L. et al. Common prefrontal activations during working memory, episodic memory, and semantic memory. Neuropsychologia 41, 371–377 (2003).
St-Laurent, M., Abdi, H., Burianová, H. & Grady, C. L. Influence of aging on the neural correlates of autobiographical, episodic, and semantic memory retrieval. J. Cogn. Neurosci. 23, 4150–4163 (2011).
Nyberg, L., Lövdén, M., Riklund, K., Lindenberger, U. & Bäckman, L. Memory aging and brain maintenance. Trends Cogn. Sci. 16, 292–305 (2012).
Patterson, K. & Lambon Ralph, M. A. in Neurobiology of Language (eds. Hickok, G. & Small, S. L.) 765–775 (Academic Press, 2016); https://doi.org/10.1016/B978-0-12-407794-2.00061-4
Bonnici, H. M., Richter, F. R., Yazar, Y. & Simons, J. S. Multimodal feature integration in the angular gyrus during episodic and semantic retrieval. J. Neurosci. 36, 5462–5471 (2016).
Rugg, M. D. & Wilding, E. L. Retrieval processing and episodic memory. Trends Cogn. Sci. 4, 108–115 (2000).
Wetzels, R., Grasman, R. P. P. P. & Wagenmakers, E.-J. An encompassing prior generalization of the Savage–Dickey density ratio. Comput. Stat. Data Anal. 54, 2094–2102 (2010).
Frisby, S. L., Halai, A. D., Cox, C. R., Lambon Ralph, M. A. & Rogers, T. T. Decoding semantic representations in mind and brain. Trends Cogn. Sci. 27, 258–281 (2023).
Cabeza, R. et al. Attention-related activity during episodic memory retrieval: a cross-function fMRI study. Neuropsychologia 41, 390–399 (2003).
Shallice, T. et al. Brain regions associated with acquisition and retrieval of verbal episodic memory. Nature 368, 633–635 (1994).
Rugg, M. D., Fletcher, P. C., Frith, C. D., Frackowiak, R. S. J. & Dolan, R. J. Differential activation of the prefrontal cortex in successful and unsuccessful memory retrieval. Brain 119, 2073–2083 (1996).
Henson, R. N., Shallice, T. & Dolan, R. J. Right prefrontal cortex and episodic memory retrieval: a functional MRI test of the monitoring hypothesis. Brain J. Neurol. 122, 1367–1381 (1999).
Zahn, R. et al. Recovery of semantic word processing in global aphasia: a functional MRI study. Cogn. Brain Res. 18, 322–336 (2004).
Raposo, A., Moss, H. E., Stamatakis, E. A. & Tyler, L. K. Repetition suppression and semantic enhancement: An investigation of the neural correlates of priming. Neuropsychologia 44, 2284–2295 (2006).
Brambati, S. M., Benoit, S., Monetta, L., Belleville, S. & Joubert, S. The role of the left anterior temporal lobe in the semantic processing of famous faces. NeuroImage 53, 674–681 (2010).
Noonan, K. A., Jefferies, E., Visser, M. & Lambon Ralph, M. A. Going beyond inferior prefrontal involvement in semantic control: evidence for the additional contribution of dorsal angular gyrus and posterior middle temporal cortex. J. Cogn. Neurosci. 25, 1824–1850 (2013).
D’Mello, A. M., Centanni, T. M., Gabrieli, J. D. E. & Christodoulou, J. A. Cerebellar contributions to rapid semantic processing in reading. Brain Lang. 208, 104828 (2020).
D’Mello, A. M., Turkeltaub, P. E. & Stoodley, C. J. Cerebellar tDCS modulates neural circuits during semantic prediction: a combined tDCS-fMRI study. J. Neurosci. 37, 1604–1613 (2017).
Stoodley, C. J. The cerebellum and cognition: evidence from functional imaging studies. Cerebellum 11, 352–365 (2012).
Gatti, D., Vecchi, T. & Mazzoni, G. Cerebellum and semantic memory: a TMS study using the DRM paradigm. Cortex 135, 78–91 (2021).
Henson, R. Forward inference using functional neuroimaging: dissociations versus associations. Trends Cogn. Sci. 10, 64–69 (2006).
Gould, R. L. et al. Brain mechanisms of successful compensation during learning in Alzheimer disease. Neurology 67, 1011–1017 (2006).
Geerligs, L., Renken, R. J., Saliasi, E., Maurits, N. M. & Lorist, M. M. A brain-wide study of age-related changes in functional connectivity. Cereb. Cortex 25, 1987–1999 (2015).
Geerligs, L., Maurits, N. M., Renken, R. J. & Lorist, M. M. Reduced specificity of functional connectivity in the aging brain during task performance. Hum. Brain Mapp. 35, 319–330 (2014).
Rubin, D. C. A conceptual space for episodic and semantic memory. Mem. Cognit. 50, 464–477 (2022).
Addis, D. R. & Szpunar, K. K. Beyond the episodic–semantic continuum: the multidimensional model of mental representations. Philos. Trans. R. Soc. B 379, 20230408 (2024).
Tanguay, A. F. et al. The shared and unique neural correlates of personal semantic, general semantic, and episodic memory. eLife 12, e83645 (2023).
Curot, J. et al. Complex memories induced by intracranial electrical brain stimulation are related to complex networks. Cortex 183, 349–372 (2025).
Maguire, E. A., Kumaran, D., Hassabis, D. & Kopelman, M. D. Autobiographical memory in semantic dementia: A longitudinal fMRI study. Neuropsychologia 48, 123–136 (2010).
Hodges, J. R. & Patterson, K. Semantic dementia: a unique clinicopathological syndrome. Lancet Neurol. 6, 1004–1014 (2007).
Halai, A. D., Henson, R. N., Finoia, P. & Correia, M. M. Comparing the effect of multi-gradient echo and multi-band fMRI during a semantic task. Imaging Neurosci. 3, IMAG.a.1043 (2025).
Gilmore, A. W., Agron, A. M., González-Araya, E. I., Gotts, S. J. & Martin, A. A comparison of single- and multi-echo processing of functional MRI data during overt autobiographical recall. Front. Neurosci. 16, 854387 (2022).
Allen, T. A. & Fortin, N. J. The evolution of episodic memory. Proc. Natl Acad. Sci. USA 110, 10379–10386 (2013).
Tulving, E. & Markowitsch, H. J. Episodic and declarative memory: role of the hippocampus. Hippocampus 8, 198–204 (1998).
Eichenbaum, H. & Fortin, N. J. Bridging the gap between brain and behavior: cognitive and neural mechanisms of episodic memory. J. Exp. Anal. Behav. 84, 619–629 (2005).
Moscovitch, M., Cabeza, R., Winocur, G. & Nadel, L. Episodic memory and beyond: the hippocampus and neocortex in transformation. Annu. Rev. Psychol. 67, 105–134 (2016).
Hassabis, D., Kumaran, D. & Maguire, E. A. Using imagination to understand the neural basis of episodic memory. J. Neurosci. 27, 14365–14374 (2007).
Reas, E. T. & Brewer, J. B. Retrieval search and strength evoke dissociable brain activity during episodic memory recall. J. Cogn. Neurosci. 25, 219–233 (2013).
Kim, H., Daselaar, S. M. & Cabeza, R. Overlapping brain activity between episodic memory encoding and retrieval: roles of the task-positive and task-negative networks. NeuroImage 49, 1045–1054 (2010).
Tibon, R., Fuhrmann, D., Levy, D. A., Simons, J. S. & Henson, R. N. Multimodal integration and vividness in the angular gyrus during episodic encoding and retrieval. J. Neurosci. 39, 4365–4374 (2019).
Schönbrodt, F. D. & Wagenmakers, E.-J. Bayes factor design analysis: planning for compelling evidence. Psychon. Bull. Rev. 25, 128–142 (2018).
Keizer, K. Impulse noise addition. MATLAB https://uk.mathworks.com/matlabcentral/fileexchange/22141-impulse-noise-addition (2025).
Drucker, D. M., Kerr, W. T. & Aguirre, G. K. Distinguishing conjoint and independent neural tuning for stimulus features with fMRI adaptation. J. Neurophysiol. 101, 3310–3324 (2009).
Persichetti, A. S., Aguirre, G. K. & Thompson-Schill, S. L. Value is in the eye of the beholder: early visual cortex codes monetary value of objects during a diverted attention task. J. Cogn. Neurosci. 27, 893–901 (2014).
Halai, A. D., Welbourne, S. R., Embleton, K. & Parkes, L. M. A comparison of dual gradient-echo and spin-echo fMRI of the inferior temporal lobe. Hum. Brain Mapp. 35, 4118–4128 (2014).
Halai, A. D., Parkes, L. M. & Welbourne, S. R. Dual-echo fMRI can detect activations in inferior temporal lobe during intelligible speech comprehension. NeuroImage 122, 214–221 (2015).
Caballero-Gaudes, C. & Reynolds, R. C. Methods for cleaning the BOLD fMRI signal. NeuroImage 154, 128–149 (2017).
Jenkinson, M., Beckmann, C. F., Behrens, T. E. J., Woolrich, M. W. & Smith, S. M. FSL. NeuroImage 62, 782–790 (2012).
Smith, S. M. et al. Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage 23, S208–S219 (2004).
Woolrich, M. W. et al. Bayesian analysis of neuroimaging data in FSL. NeuroImage 45, S173–S186 (2009).
Cox, R. W. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 29, 162–173 (1996).
DuPre, E. et al. ME-ICA/Tedana: 0.0.9a. Zenodo https://doi.org/10.5281/zenodo.3786890 (2020).
Kundu, P., Inati, S. J., Evans, J. W., Luh, W.-M. & Bandettini, P. A. Differentiating BOLD and non-BOLD signals in fMRI time series using multi-echo EPI. NeuroImage 60, 1759–1770 (2012).
Kundu, P. et al. Integrated strategy for improving functional connectivity mapping using multiecho fMRI. Proc. Natl Acad. Sci. USA 110, 16187–16192 (2013).
Poser, B. A., Versluis, M. J., Hoogduin, J. M. & Norris, D. G. BOLD contrast sensitivity enhancement and artifact reduction with multiecho EPI: parallel-acquired inhomogeneity-desensitized fMRI. Magn. Reson. Med. 55, 1227–1235 (2006).
Tibon, R. & Levy, D. A. Striking a balance: analyzing unbalanced event-related potential data. Front. Psychol. 6, 555 (2015).
Bürkner, P.-C. brms: an R package for Bayesian multilevel models using Stan. J. Stat. Softw. 80, 1–28 (2017).
Bürkner, P.-C. Advanced Bayesian multilevel modeling with the R package brms. R J. 10, 395–411 (2018).
Binder, J. R., Desai, R. H., Graves, W. W. & Conant, L. L. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb. Cortex 19, 2767–2796 (2009).
Humphreys, G. F., Hoffman, P., Visser, M., Binney, R. J. & Ralph, M. A. L. Establishing task- and modality-dependent dissociations between the semantic and default mode networks. Proc. Natl Acad. Sci. USA 112, 7857–7862 (2015).
Mumford, J. A., Davis, T. & Poldrack, R. A. The impact of study design on pattern estimation for single-trial multivariate pattern analysis. NeuroImage 103, 130–138 (2014).
Abdulrahman, H. & Henson, R. N. Effect of trial-to-trial variability on optimal event-related fMRI design: implications for beta-series correlation and multi-voxel pattern analysis. NeuroImage 125, 756–766 (2016).
Barr, D. J., Levy, R., Scheepers, C. & Tily, H. J. Random effects structure for confirmatory hypothesis testing: Keep it maximal. J. Mem. Lang. 68, 255–278 (2013).
Bates, D., Kliegl, R., Vasishth, S. & Baayen, H. Parsimonious mixed models. Preprint at https://arxiv.org/abs/1506.04967 (2018).
Acknowledgements
R.T. was supported by a British Academy Postdoctoral Fellowship (grant no. SUAI/028 RG94188). G.H. was supported by an MRC Programme grant to Matt Lambon Ralph (grant no. MR/R023883/1) and MRC intramural funding (grant no. MC_UU_00005/18). J.A.Q. was supported by the Gates Cambridge Trust PhD studentship. R.H. and A.G. were supported by a UK Medical Research Council grant (grant no. SUAG/010 RG91365). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. We are grateful to A. Halai for advice on acquisition and preprocessing of multi-echo data, to L. Johnson for help with data collection and to D. Suchowski and I. Leitner for help with figure preparation.
Author information
Authors and Affiliations
Contributions
R.T. directed the project together with R.H., designed and planned the project, collected the data, carried out the analyses and wrote the paper with input from all authors. A.G. consulted on design and analyses, helped with data collection and provided input on the analyses and the paper. G.H. consulted on design and analyses and provided input on the analyses and the paper. J.A.Q. designed and executed sample size determination and provided input on the analyses and the paper. R.H. directed the project together with R.T., consulted on design and analyses and provided input on the analyses and the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Human Behaviour thanks Louis Renoult, Deniz Vatansever and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Supplementary information
Supplementary Information (download PDF )
Supplementary Analyses 1 and 2 and Fig. 1.
Supplementary Tables (download XLSX )
Supplementary Table 1 Exploratory data-defined ROIs. Supplementary Table 2 Univariate analyses results with exploratory data-defined ROIs. Supplementary Table 3 Similarity analyses results with exploratory data-defined ROIs.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tibon, R., Greve, A., Humphreys, G. et al. Neural activations and representations during episodic versus semantic memory retrieval. Nat Hum Behav (2026). https://doi.org/10.1038/s41562-025-02390-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41562-025-02390-4