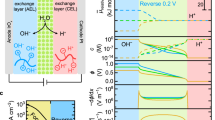
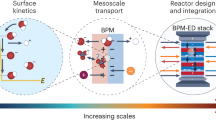
Abstract
The voltage penalty driving water dissociation (WD) at high current density is a major obstacle in the commercialization of bipolar membrane (BPM) technology for energy devices. Here we show that three materials descriptors, that is, electrical conductivity, microscopic surface area and (nominal) surface-hydroxyl coverage, effectively control the kinetics of WD in BPMs. Using these descriptors and optimizing mass loading, we design new earth-abundant WD catalysts based on nanoparticle SnO2 synthesized at low temperature with high conductivity and hydroxyl coverage. These catalysts exhibit exceptional performance in a BPM electrolyser with low WD overvoltage (ηwd) of 100 ± 20 mV at 1.0 A cm−2. The new catalyst works equivalently well with hydrocarbon proton-exchange layers as it does with fluorocarbon-based Nafion, thus providing pathways to commercializing advanced BPMs for a broad array of electrolysis, fuel-cell and electrodialysis applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data generated from this study and used to prepare the figures in the main manuscript are available with the digital identifier (https://doi.org/10.6084/m9.figshare.25769388). Further datasets generated during the study are available from the corresponding author on reasonable request. Source data are provided with this paper.
References
Frilette, V. J. Preparation and characterization of bipolar ion exchange membranes. J. Phys. Chem. 60, 435–439 (1956).
Pärnamäe, R. et al. Bipolar membranes: a review on principles, latest developments, and applications. J. Membr. Sci. 617, 118538 (2021).
Giesbrecht, P. K. & Freund, M. S. Recent advances in bipolar membrane design and applications. Chem. Mater. 32, 8060–8090 (2020).
Arana Juve, J.-M., Christensen, F. M. S., Wang, Y. & Wei, Z. Electrodialysis for metal removal and recovery: a review. Chem. Eng. J. 435, 134857 (2022).
Bazinet, L., Lamarche, F. & Ippersiel, D. Bipolar-membrane electrodialysis: applications of electrodialysis in the food industry. Trends Food Sci. Technol. 9, 107–113 (1998).
Chen, L., Xu, Q., Oener, S. Z., Fabrizio, K. & Boettcher, S. W. Design principles for water dissociation catalysts in high-performance bipolar membranes. Nat. Commun. 13, 3846–3846 (2022).
Oener, S. Z., Twight, L. P., Lindquist, G. A. & Boettcher, S. W. Thin cation-exchange layers enable high-current-density bipolar membrane electrolyzers via improved water transport. ACS Energy Lett. 6, 1–8 (2020).
Oener, S. Z., Foster, M. J. & Boettcher, S. W. Accelerating water dissociation in bipolar membranes and for electrocatalysis. Science 369, 1099–1103 (2020).
Ge, Z. et al. High-performance bipolar membrane for electrochemical water electrolysis. J. Membr. Sci. 656, 120660 (2022).
Marin, D. H. et al. Hydrogen production with seawater-resilient bipolar membrane electrolyzers. Joule 7, 765–781 (2023).
Xie, K. et al. Bipolar membrane electrolyzers enable high single-pass CO2 electroreduction to multicarbon products. Nat. Commun. 13, 3609–3609 (2022).
Li, Y. C. et al. Bipolar membranes inhibit product crossover in CO2 electrolysis cells. Adv. Sustain. Syst. 2, 1700187 (2018).
Salvatore, D. A. et al. Electrolysis of gaseous CO2 to CO in a flow cell with a bipolar membrane. ACS Energy Lett. 3, 149–154 (2017).
Disch, J., Ingenhoven, S. & Vierrath, S. Bipolar membrane with porous anion exchange layer for efficient and long-term stable electrochemical reduction of CO2 to CO. Adv. Energy Mater. 13, 2301614 (2023).
Li, Q. et al. Theoretical design strategies of bipolar membrane fuel cell with enhanced self-humidification behavior. J. Power Sources 307, 358–367 (2016).
Ünlü, M., Zhou, J. & Kohl, P. A. Hybrid anion and proton exchange membrane fuel cells. J. Phys. Chem. C 113, 11416–11423 (2009).
Bui, J. C. et al. Analysis of bipolar membranes for electrochemical CO2 capture from air and oceanwater. Energy Environ. Sci. 16, 5076–5095 (2023).
Bui, J. C. et al. Multi-scale physics of bipolar membranes in electrochemical processes. Nat. Chem. Eng. 1, 45–60 (2024).
Chen, L., Xu, Q. & Boettcher, S. W. Kinetics and mechanism of heterogeneous voltage-driven water-dissociation catalysis. Joule 7, 1867–1886 (2023).
Shehzad, M. A. et al. Shielded goethite catalyst that enables fast water dissociation in bipolar membranes. Nat. Commun. 12, 9 (2021).
Lucas, É. et al. Asymmetric bipolar membrane for high current density electrodialysis operation with exceptional stability. Preprint at https://chemrxiv.org/engage/chemrxiv/article-details/642df4da0784a63aee99154d (2023).
Powers, D. et al. Freestanding bipolar membranes with an electrospun junction for high current density water splitting. ACS Appl. Mater. Interfaces 14, 36092–36104 (2022).
Kole, S. et al. Bipolar membrane polarization behavior with systematically varied interfacial areas in the junction region. J. Mater. Chem. A 9, 2223–2238 (2021).
Bui, J. C., Corpus, K. R. M., Bell, A. T. & Weber, A. Z. On the nature of field-enhanced water dissociation in bipolar membranes. J. Phys. Chem. C 125, 24974–24987 (2021).
Cai, J. et al. Wien effect in interfacial water dissociation through proton-permeable graphene electrodes. Nat. Commun. 13, 5776–5776 (2022).
Onsager, L. Deviations from Ohm’s law in weak electrolytes. J. Chem. Phys. 2, 599–615 (1934).
Rodellar, C. G., Gisbert-Gonzalez, J. M., Sarabia, F., Roldan Cuenya, B. & Oener, S. Z. Ion solvation kinetics in bipolar membranes and at electrolyte–metal interfaces. Nat. Energy 10.1038/s41560-41024-01484-z (2024).
Henry, B. J. et al. A critical review of the application of polymer of low concern and regulatory criteria to fluoropolymers. Integr. Environ. Assess. Manag. 14, 316–334 (2018).
Lindquist, G. A. et al. Oxidative instability of ionomers in hydroxide-exchange-membrane water electrolyzers. Energy Environ. Sci. 16, 4373–4387 (2023).
Mineart, K. P., Lee, B. & Spontak, R. J. A solvent-vapor approach toward the control of block ionomer morphologies. Macromolecules 49, 3126–3137 (2016).
Tang, B. Y., Bisbey, R. P., Lodaya, K. M., Toh, W. L. & Surendranath, Y. Reaction environment impacts charge transfer but not chemical reaction steps in hydrogen evolution catalysis. Nat. Catal. 6, 339–350 (2023).
Dinh, H. Q., Toh, W. L., Chu, A. T. & Surendranath, Y. Neutralization short-circuiting with weak electrolytes erodes the efficiency of bipolar membranes. ACS Appl. Mater. Interfaces 15, 4001–4010 (2023).
Hohenadel, A., Gangrade, A. S. & Holdcroft, S. Spectroelectrochemical detection of water dissociation in bipolar membranes. ACS Appl. Mater. Interfaces 13, 46125–46133 (2021).
Reiter, R. S., White, W. & Ardo, S. Electrochemical characterization of commercial bipolar membranes under electrolyte conditions relevant to solar fuels technologies. J. Electrochem. Soc. 163, H3132 (2016).
Mitchell, J. B., Chen, L., Langworthy, K., Fabrizio, K. & Boettcher, S. W. Catalytic proton–hydroxide recombination for forward-bias bipolar membranes. ACS Energy Lett. 7, 3967–3973 (2022).
Xu, Q. et al. Integrated reference electrodes in anion-exchange-membrane electrolyzers: impact of stainless-steel gas-diffusion layers and internal mechanical pressure. ACS Energy Lett. 6, 305–312 (2021).
Boehm, H. P. Acidic and basic properties of hydroxylated metal oxide surfaces. Discuss. Faraday Soc. 52, 264–275 (1971).
Tamura, H., Mita, K., Tanaka, A. & Ito, M. Mechanism of hydroxylation of metal oxide surfaces. J. Colloid Interface Sci. 243, 202–207 (2001).
Mueller, R., Kammler, H. K., Wegner, K. & Pratsinis, S. E. OH surface density of SiO2 and TiO2 by thermogravimetric analysis. Langmuir 19, 160–165 (2003).
Chen, J. et al. Interactions of oxide surfaces with water revealed with solid-state NMR spectroscopy. J. Am. Chem. Soc. 142, 11173–11182 (2020).
Wang, G. et al. Hydrogen-treated TiO2 nanowire arrays for photoelectrochemical water splitting. Nano Lett. 11, 3026–3033 (2011).
Fujihara, S. et al. Hydrothermal routes to prepare nanocrystalline mesoporous SnO2 having high thermal stability. Langmuir 20, 6476–6481 (2004).
Sheng, W., Gasteiger, H. A. & Shao-Horn, Y. Hydrogen oxidation and evolution reaction kinetics on platinum: acid vs alkaline electrolytes. J. Electrochem. Soc. 157, B1529 (2010).
Schwämmlein, J. N. et al. Origin of superior HOR/HER activity of bimetallic Pt–Ru catalysts in alkaline media identified via Ru@Pt core–shell nanoparticles. J. Electrochem. Soc. 165, H229 (2018).
Tongwen, X. Electrodialysis processes with bipolar membranes (EDBM) in environmental protection—a review. Resour. Conserv. Recycl. 37, 1–22 (2002).
Bipolar Membrane Electrodialyzer [ACILYZER BPED products]. ASTOM http://www.astom-corp.jp/en/product/05.html (2023).
Culcasi, A., Gurreri, L., Cipollina, A., Tamburini, A. & Micale, G. A comprehensive multi-scale model for bipolar membrane electrodialysis (BMED). Chem. Eng. J. 437, 135317 (2022).
Mineart, K. P., Jiang, X., Jinnai, H., Takahara, A. & Spontak, R. J. Morphological investigation of midblock-sulfonated block ionomers prepared from solvents differing in polarity. Macromol. Rapid Commun. 36, 432–438 (2015).
Acknowledgements
The work investigating WD catalyst descriptors was supported by the US Office of Naval Research, grant N00014-20-1-2517 (S.W.B.). The design of the new high-performance catalyst was supported by US Advanced Research Projects Agency-Energy (ARPA-E), grant DE-AR0001502. The work applying the new catalyst to electrodialysis and with hydrocarbon cation-exchange membrane layers was supported by ARPA-E grant DE-AR0001540. P.V.S. acknowledges the Fulbright-Nehru postdoctoral fellowship supported by USIEF. K.M.W. and R.J.S. acknowledge support from the Kraton Corporation and the NC State College of Engineering. We acknowledge use of shared instrumentation in the Center for Advanced Materials Characterization in Oregon and the Phil and Penny Knight Campus. J. Razink collected electron-microscopy images. R. Wycisk and J. Doshi at eSpin Technologies are acknowledged for providing SPEEK membranes and for useful technical discussion.
Author information
Authors and Affiliations
Contributions
S.S. and S.W.B. conceived the experiments and led the project. S.S. performed most experiments. L.C. developed membrane-sensing experiments and data processing approaches. P.V.S. led the AEM and low-Pt BPM experiments. O.T.V. conceived the thickness measurements and assisted in the BPMED prototype experiment. C.R.S. performed the NMR experiments. K.M.W. and R.J.S. initiated discussions regarding and developed the TESET membrane. S.S. and S.W.B. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors have filed a patent application titled ‘Dissociation and recombination catalyst layers for reverse and forward-bias bipolar membranes’ (US20230264148A1), on materials reported in this manuscript and are working to commercialize advanced BPMs.
Peer review
Peer review information
Nature Materials thanks Christopher G. Arges, Severin Vierrath and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary figures and discussion.
Source data
Source Data Fig. 2 (download XLSX )
Source ascii data for figure.
Source Data Fig. 3 (download XLSX )
Source ascii data for figure.
Source Data Fig. 4 (download XLSX )
Source ascii data for figure.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sasmal, S., Chen, L., Sarma, P.V. et al. Materials descriptors for advanced water dissociation catalysts in bipolar membranes. Nat. Mater. 23, 1421–1427 (2024). https://doi.org/10.1038/s41563-024-01943-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41563-024-01943-8
This article is cited by
-
Stable acidic oxygen-evolving catalyst discovery through mixed accelerations
Nature Catalysis (2026)
-
Synthesis of deuterated acids and bases using bipolar membranes
Nature (2025)
-
Interconnected nanoconfining pore networks enhance catalyst CO2 interaction in electrified reactive capture
Nature Communications (2025)
-
Water dissociation efficiencies control the viability of reverse-bias bipolar membranes for CO2 electrolysis
Nature Chemical Engineering (2025)
-
Membrane electrode assembly for hydrogen peroxide electrosynthesis
Nature Reviews Clean Technology (2025)