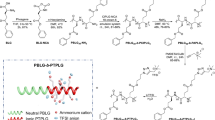
Abstract
Ion transport is essential to energy storage, cellular signalling and desalination. Polymers have been explored for decades as solid-state electrolytes by either adding salt to polar polymers or tethering ions to the backbone to create less flammable and more robust systems. New design paradigms are needed to advance the performance of solid polymer electrolytes beyond conventional systems. Here the role of a helical secondary structure is shown to greatly enhance the conductivity of solvent-free polymer electrolytes using cationic polypeptides with a mobile anion. Longer helices lead to higher conductivity, and random coil peptides show substantially lower conductivity. The macrodipole of the helix increases with peptide length, leading to larger dielectric constants. The hydrogen bonding of the helix also imparts thermal and electrochemical stability, while allowing for facile dissolution back to monomer in acid. Peptide polymer electrolytes present a promising platform for the design of next-generation ion-transporting materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and the Supplementary Information. The raw numbers for charts and graphs are available in the provided Source Data file whenever possible. Additional images are available from the corresponding author upon reasonable request. Source data are provided with this paper.
Change history
07 October 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41563-024-02040-6
References
Sun, H. T. et al. Hierarchical 3D electrodes for electrochemical energy storage. Nat. Rev. Mater. 4, 45–60 (2019).
Sun, Y. L., Liu, B., Liu, L. Y. & Yan, X. B. Ions transport in electrochemical energy storage devices at low temperatures. Adv. Funct. Mater. 32, 2109568 (2022).
Xiao, K., Jiang, L. & Antonietti, M. Ion transport in nanofluidic devices for energy harvesting. Joule 3, 2364–2380 (2019).
Chen, J. et al. Localized electrons enhanced ion transport for ultrafast electrochemical energy storage. Adv. Mater. 32, e1905578 (2020).
Yan, C. et al. Engineering 2D nanofluidic Li-ion transport channels for superior electrochemical energy storage. Adv. Mater. 29, 1703909 (2017).
Lin, P. & Yan, F. Organic thin-film transistors for chemical and biological sensing. Adv. Mater. 24, 34–51 (2012).
Jentsch, T. J. VRACs and other ion channels and transporters in the regulation of cell volume and beyond. Nat. Rev. Mol. Cell Biol. 17, 293–307 (2016).
Ratner, M. A. & Shriver, D. F. Ion-transport in solvent-free polymers. Chem. Rev. 88, 109–124 (1988).
Armand, M. & Tarascon, J. M. Building better batteries. Nature 451, 652–657 (2008).
Shen, C. T., Zhao, Q. J., Shan, N. S., Jing, B. B. & Evans, C. M. Conductivity–modulus–Tg relationships in solvent‐free, single lithium ion conducting network electrolytes. J. Polym. Sci. 58, 2376–2388 (2020).
Bouchet, R. et al. Single-ion BAB triblock copolymers as highly efficient electrolytes for lithium-metal batteries. Nat. Mater. 12, 452–457 (2013).
Sun, C. G. et al. Fast lithium ion transport in solid polymer electrolytes from polysulfide-bridged copolymers. Nano Energy 75, 104976 (2020).
Sharon, D. et al. Intrinsic ion transport properties of block copolymer electrolytes. ACS Nano 14, 8902–8914 (2020).
Liu, D. et al. Enhancing ionic conductivity in tablet–bottlebrush block copolymer electrolytes with well-aligned nanostructures via solvent vapor annealing. J. Mater. Chem. C. 10, 4247–4256 (2022).
Jia, D. et al. Multifunctional polymer bottlebrush-based gel polymer electrolytes for lithium metal batteries. Mater. Today Nano 15, 100128 (2021).
Deng, C. T. et al. Role of molecular architecture on ion transport in ethylene oxide-based polymer electrolytes. Macromolecules 54, 2266–2276 (2021).
Evans, C. M., Bridges, C. R., Sanoja, G. E., Bartels, J. & Segalman, R. A. Role of tethered ion placement on polymerized ionic liquid structure and conductivity: pendant versus backbone charge placement. ACS Macro Lett. 5, 925–930 (2016).
Sangoro, J. R. et al. Decoupling of ionic conductivity from structural dynamics in polymerized ionic liquids. Soft Matter 10, 3536–3540 (2014).
Jones, S. D. et al. Design of polymeric zwitterionic solid electrolytes with superionic lithium transport. ACS Cent. Sci. 8, 169–175 (2022).
Leigh, T. & Fernandez-Trillo, P. Helical polymers for biological and medical applications. Nat. Rev. Chem. 4, 291–310 (2020).
Ekladious, I., Colson, Y. L. & Grinstaff, M. W. Polymer–drug conjugate therapeutics: advances, insights and prospects. Nat. Rev. Drug Discov. 18, 273–294 (2019).
Gao, Y. et al. Winding-locked carbon nanotubes/polymer nanofibers helical yarn for ultrastretchable conductor and strain sensor. ACS Nano 14, 3442–3450 (2020).
Wang, M. X. et al. Conductance-stable and integrated helical fiber electrodes toward stretchy energy storage and self-powered sensing utilization. Chem. Eng. J. 457, 141164 (2023).
Liu, Y. S. et al. Controllable synthesis of Co@CoOx/helical nitrogen-doped carbon nanotubes toward oxygen reduction reaction as binder-free cathodes for Al–air batteries. ACS Appl. Mater. Interfaces 12, 16512–16520 (2020).
Zhao, M. Q. et al. Hierarchical vine-tree-like carbon nanotube architectures: in-situ CVD self-assembly and their use as robust scaffolds for lithium–sulfur batteries. Adv. Mater. 26, 7051–7058 (2014).
Jiang, Y. J. et al. ‘Metaphilic’ cell-penetrating polypeptide–vancomycin conjugate efficiently eradicates intracellular bacteria via a dual mechanism. ACS Cent. Sci. 6, 2267–2276 (2020).
Jiang, Y., Chen, Y., Song, Z., Tan, Z. & Cheng, J. Recent advances in design of antimicrobial peptides and polypeptides toward clinical translation. Adv. Drug. Deliv. Rev. 170, 261–280 (2021).
Nguyen, T. P. et al. Polypeptide organic radical batteries. Nature 593, 61–66 (2021).
Lightfoot, P., Mehta, M. A. & Bruce, P. G. Crystal structure of the polymer electrolyte poly(ethylene oxide)3:LiCF3SO3. Science 262, 883–885 (1993).
Ma, Y. A., Shen, Y. & Li, Z. B. Different cell behaviors induced by stereochemistry on polypeptide brush grafted surfaces. Mater. Chem. Front. 1, 846–851 (2017).
Papadopoulos, P., Floudas, G., Klok, H. A., Schnell, I. & Pakula, T. Self-assembly and dynamics of poly(γ-benzyl-l-glutamate) peptides. Biomacromolecules 5, 81–91 (2004).
Kricheldorf, H. R. & Mueller, D. Secondary structure of peptides. 3. Carbon-13 NMR cross polarization/magic angle spinning spectroscopic characterization of solid polypeptides. Macromolecules 16, 615–623 (1983).
Tsutsumi, A. et al. Relaxation phenomena of poly-γ-benzyl-l-glutamate, poly-γ-methyl-l-glutamate, and copoly(γ-methyl-l-glutamate, γ-benzyl-l-glutamate). J. Macromol. Sci., B 8, 413–430 (1973).
Evans, C. M., Sanoja, G. E., Popere, B. C. & Segalrnan, R. A. Anhydrous proton transport in polymerized ionic liquid block copolymers: roles of block length, ionic content, and confinement. Macromolecules 49, 395–404 (2016).
Drozd-Rzoska, A., Rzoska, S. J. & Starzonek, S. New paradigm for configurational entropy in glass-forming systems. Sci. Rep. 12, 3058 (2022).
Heres, M. et al. Ion transport in glassy polymerized ionic liquids: unraveling the impact of the molecular structure. Macromolecules 52, 88–95 (2019).
Cheng, S. J. et al. Ionic aggregation in random copolymers containing phosphonium ionic liquid monomers. J. Polym. Sci. A1 50, 166–173 (2012).
Hemp, S. T. et al. Comparing ammonium and phosphonium polymerized ionic liquids: thermal analysis, conductivity, and morphology. Macromol. Chem. Phys. 214, 2099–2107 (2013).
Xia, Y. C. et al. Accelerated polymerization of N-carboxyanhydrides catalyzed by crown ether. Nat. Commun. 12, 732 (2021).
Fan, F. et al. Effect of molecular weight on the ion transport mechanism in polymerized ionic liquids. Macromolecules 49, 4557–4570 (2016).
Zhao, Q. J. & Evans, C. M. Effect of molecular weight on viscosity scaling and ion transport in linear polymerized ionic liquids. Macromolecules 54, 3395–3404 (2021).
Keith, J. R., Mogurampelly, S., Aldukhi, F., Wheatle, B. K. & Ganesan, V. Influence of molecular weight on ion-transport properties of polymeric ionic liquids. Phys. Chem. Chem. Phys. 19, 29134–29145 (2017).
Timachova, K., Watanabe, H. & Balsara, N. P. Effect of molecular weight and salt concentration on ion transport and the transference number in polymer electrolytes. Macromolecules 48, 7882–7888 (2015).
Han, S. et al. Sequencing polymers to enable solid-state lithium batteries. Nat. Mater. 22, 1515–1522 (2023).
Wada, A. Dielectric properties of polypeptide solutions. II. Relation between the electric dipole moment and the molecular weight of α helix. J. Chem. Phys. 30, 328–329 (1959).
Choi, U. H. et al. Role of chain polarity on ion and polymer dynamics: molecular volume-based analysis of the dielectric constant for polymerized norbornene-based ionic liquids. Macromolecules 53, 10561–10573 (2020).
Wilcox, K. G., Dingle, M. E., Saha, A., Hore, M. J. A. & Morozova, S. Persistence length of α-helical poly-l-lysine. Soft Matter 18, 6550–6560 (2022).
Choe, S. & Sun, S. X. The elasticity of α-helices. J. Chem. Phys. 122, 244912 (2005).
Papadopoulos, P. et al. Thermodynamic confinement and α-helix persistence length in poly(γ-benzyl-l-glutamate)-b-poly(dimethyl siloxane)-b-poly(γ-benzyl-l-glutamate) triblock copolymers. Biomacromolecules 7, 618–626 (2006).
Zhao, Q. J., Bennington, P., Nealey, P. F., Patel, S. N. & Evans, C. M. Ion specific, thin film confinement effects on conductivity in polymerized ionic liquids. Macromolecules 54, 10520–10528 (2021).
Acknowledgements
This work is partially supported by the United States National Science Foundation (NSF CHE 17-09820 to J.C. and CHE 19-05097 to J.C. and P.V.B. for peptide synthesis, and DMR-1751291 to C.M.E. for polymerized ionic liquid physics). The work is also partially supported by the US Department of Energy, Office of Basic Energy Science, Division of Materials Sciences and Engineering under award #DE-SC0020858 (ionic conductivity and dielectric measurements). The authors acknowledge the facility and instrumental support from the Materials Research Laboratory, the SCS NMR Laboratory, Beckman Institute, University of Illinois Urbana-Champaign. Specifically, the Q-Tof Ultima mass spectrometer was purchased in part with a grant from the National Science Foundation, Division of Biological Infrastructure (DBI-0100085).
Author information
Authors and Affiliations
Contributions
C.M.E conceived of using the helix to enhance conductivity, and Y.C and T.X. conceived of the polymer electrolyte design and synthesis. Solid 13C NMR was conducted by S.J. Dielectric spectroscopy was conducted by C.C. Y.C. and C.M.E. wrote the manuscript with contributions and critical feedback from T.X., C.C., S.J., J.C. and P.V.B. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Darrin Pochan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Schemes 1–6, Figs. 1–51 and Tables 1 and 2.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Xue, T., Chen, C. et al. Helical peptide structure improves conductivity and stability of solid electrolytes. Nat. Mater. 23, 1539–1546 (2024). https://doi.org/10.1038/s41563-024-01966-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41563-024-01966-1