Abstract
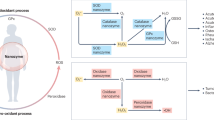
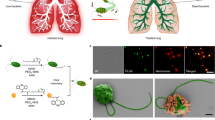
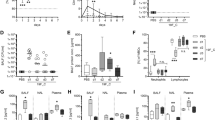
Pneumonia is a ubiquitous disease caused by viral and bacterial infections, characterized by high levels of reactive oxygen species in inflamed areas. Therapeutic strategies targeting reactive oxygen species levels in pneumonia have limited success due to the intricate nature of lung tissues and lung inflammatory responses. Here we describe an inhalable, non-invasive therapeutic platform composed of engineered cerium-based tannic acid nanozymes bound to a self-assembling peptide. In vitro and in vivo studies show that the nanozyme is internalized mostly by activated macrophages and epithelial cells in the inflamed sites. In the oxidative environments of a mouse model of viral pneumonia, nanozyme aggregates into catalytically active structures that reduce reactive oxygen species levels and inflammatory cytokine production and promote macrophage polarization to the prohealing (M2) phenotype. Moreover, the nanozyme attenuates bacterial inflammation and reduces tissue damage in a mouse viral pneumonia model with secondary bacterial infection. Overall, this nanozyme platform is a promising strategy for treating pneumonia and its associated conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information and are available from the corresponding authors upon request. Source data are provided with this paper.
References
Ruuskanen, O., Lahti, E., Jennings, L. C. & Murdoch, D. R. Viral pneumonia. Lancet 377, 1264–1275 (2011).
Wang, C. C. et al. Airborne transmission of respiratory viruses. Science 373, abd9149 (2021).
Zhou, P. et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579, 270–273 (2020).
Lessler, J. et al. Incubation periods of acute respiratory viral infections: a systematic review. Lancet Infect. Dis. 9, 291–300 (2009).
Imai, Y. et al. Identification of oxidative stress and toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell 133, 235–249 (2008).
Iwasaki, A. & Pillai, P. S. Innate immunity to influenza virus infection. Nat. Rev. Immunol. 14, 315–328 (2014).
To, E. E. et al. Endosomal NOX2 oxidase exacerbates virus pathogenicity and is a target for antiviral therapy. Nat. Commun. 8, 69 (2017).
Noreng, S. et al. Structure of the core human NADPH oxidase NOX2. Nat. Commun. 13, 6079 (2022).
Kim, S. Y. et al. Pro-inflammatory hepatic macrophages generate ROS through NADPH oxidase 2 via endocytosis of monomeric TLR4–MD2 complex. Nat. Commun. 8, 2247 (2017).
Wallace, D. C. Mitochondrial diseases in man and mouse. Science 283, 1482–1488 (1999).
Dickinson, B. C. & Chang, C. J. Chemistry and biology of reactive oxygen species in signaling or stress responses. Nat. Chem. Biol. 7, 504–511 (2011).
Wang, Y. et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet 395, 1569–1578 (2020).
Hung, I. F. et al. Triple combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet 395, 1695–1704 (2020).
Gubareva, L. V., Kaiser, L. & Hayden, F. G. Influenza virus neuraminidase inhibitors. Lancet 355, 827–835 (2000).
Choudhary, M. C. et al. Emergence of SARS-CoV-2 escape mutations during Bamlanivimab therapy in a phase II randomized clinical trial. Nat. Microbiol. 7, 1906–1917 (2022).
Gillin, J. C., Jacobs, L. S., Fram, D. H. & Snyder, F. Acute effect of a glucocorticoid on normal human sleep. Nature 237, 398–399 (1972).
Perretti, M. et al. Endogenous lipid- and peptide-derived anti-inflammatory pathways generated with glucocorticoid and aspirin treatment activate the lipoxin A4 receptor. Nat. Med. 8, 1296–1302 (2002).
Snelgrove, R. J. et al. A critical function for CD200 in lung immune homeostasis and the severity of influenza infection. Nat. Immunol. 9, 1074–1083 (2008).
Crotty, S. et al. The broad-spectrum antiviral ribonucleoside ribavirin is an RNA virus mutagen. Nat. Med. 6, 1375–1379 (2000).
Driouich, J.-S. et al. Favipiravir antiviral efficacy against SARS-CoV-2 in a hamster model. Nat. Commun. 12, 1735 (2021).
Shannon, A. et al. Rapid incorporation of favipiravir by the fast and permissive viral RNA polymerase complex results in SARS-CoV-2 lethal mutagenesis. Nat. Commun. 11, 4682 (2020).
Gao, L. et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2, 577–583 (2007).
Richter, D. Decomposition of hydrogen peroxide by catalase. Nature 129, 870 (1932).
Sies, H. Strategies of antioxidant defense. Eur. J. Biochem. 215, 213–219 (1993).
Liu, B., Sun, Z., Huang, P. J. & Liu, J. Hydrogen peroxide displacing DNA from nanoceria: mechanism and detection of glucose in serum. J. Am. Chem. Soc. 137, 1290–1295 (2015).
Allawadhi, P. et al. Nanoceria as a possible agent for the management of COVID-19. Nano Today 35, 100982 (2020).
Fu, X. et al. Small molecule-assisted assembly of multifunctional ceria nanozymes for synergistic treatment of atherosclerosis. Nat. Commun. 13, 6528 (2022).
Zhang, Y. et al. Nanozymes for nanohealthcare. Nat. Rev. Methods Prim. 4, 36 (2024).
Ma, X. et al. Prussian blue nanozyme as a pyroptosis inhibitor alleviates neurodegeneration. Adv. Mater. 34, 2106723 (2022).
Wang, Z. et al. A thrombin-activated peptide-templated nanozyme for remedying ischemic stroke via thrombolytic and neuroprotective actions. Adv. Mater. 36, 2210144 (2024).
Yu, P. et al. Mimicking antioxidases and hyaluronan synthase: a zwitterionic nanozyme for photothermal therapy of osteoarthritis. Adv. Mater. 35, 2303299 (2023).
Wang, H. et al. Genetically engineered and enucleated human mesenchymal stromal cells for the targeted delivery of therapeutics to diseased tissue. Nat. Biomed. Eng. 6, 882–897 (2022).
Deng, C. et al. Targeted apoptosis of macrophages and osteoclasts in arthritic joints is effective against advanced inflammatory arthritis. Nat. Commun. 12, 2174 (2021).
Huang, Y. et al. Nanotechnology’s frontier in combatting infectious and inflammatory diseases: prevention and treatment. Signal Transduct. Target. Ther. 9, 34 (2024).
Cui, L. et al. CeO2 nanoparticles induce pulmonary fibrosis via activating S1P pathway as revealed by metabolomics. Nano Today 45, 101559 (2022).
Pauluhn, J. Kinetic modeling of the retention and fate of inhaled cerium oxide nanoparticles in rats: the cumulative displacement volume of agglomerates determines the outcome. Regul. Toxicol. Pharm. 86, 319–331 (2017).
Gupta, M. K. et al. Cell protective, ABC triblock polymer-based thermoresponsive hydrogels with ROS-triggered degradation and drug release. J. Am. Chem. Soc. 136, 14896–14902 (2014).
Shim, M. S. & Xia, Y. A reactive oxygen species (ROS)-responsive polymer for safe, efficient, and targeted gene delivery in cancer cells. Angew. Chem. Int. Ed. 52, 6926–6929 (2013).
Kanekiyo, M. et al. Self-assembling influenza nanoparticle vaccines elicit broadly neutralizing H1N1 antibodies. Nature 499, 102–106 (2013).
Porotto, M., Greengard, O., Poltoratskaia, N., Horga, M. A. & Moscona, A. Human parainfluenza virus type 3 HN-receptor interaction: effect of 4-guanidino-Neu5Ac2en on a neuraminidase-deficient variant. J. Virol. 75, 7481–7488 (2001).
Zuo, Y. et al. Neutrophil extracelluar traps in COVID-19. JCI Insight 5, e138999 (2020).
Chen, H. Y. et al. Clinical and epidemiological characteristics of a fatal case of avian influenza A H10N8 virus infection: a descriptive study. Lancet 383, 714–721 (2014).
Myerson, J. W. et al. Supramolecular arrangement of protein in nanoparticle structures predicts nanoparticle tropism for neutrophils in acute lung inflammation. Nat. Nanotechnol. 17, 86–97 (2022).
Zhang, Q. et al. Neutrophil membrane-coated nanoparticles inhibit synovial inflammation and alleviate joint damage in inflammatory arthritis. Nat. Nanotechnol. 13, 1182–1190 (2018).
Guo, J. et al. Engineering multifunctional capsules through the assembly of metal–phenolic networks. Angew. Chem. Int. Ed. 53, 5546–5551 (2014).
He, Q. et al. Safeguarding osteointegration in diabetic patients: a potent ‘chain armor’ coating for scavenging ROS and macrophage reprogramming in a microenvironment-responsive manner. Adv. Funct. Mater. 31, 2101611 (2021).
Cheng, D. B. et al. Endogenous reactive oxygen species-triggered morphology transformation for enhanced cooperative interaction with mitochondria. J. Am. Chem. Soc. 141, 7235–7239 (2019).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (32271400 to B.Z., 82271872 to W.T., 32100755 to W.T., 82341046 to W.T. and 32188101 to G.C.), National Key Research and Development Program of China (2021YFC2600503 to B.Z., 2021YFC2300200 to G.C., 2021YFC2302405 to G.C., 2022YFC2303200 to G.C. and 2022YFC2303400 to G.C.), Key Project of Tianjin Natural Science Foundation (21JCQNJC00690 to B.Z.), Elderly Health & Happiness Major Program of China Ageing Development Foundation (EHH20211002 and EHH20211001 to B.Z.), Shenzhen Bay Laboratory Startup Fund (no. 21330111 to W.T.), Shenzhen San-Ming Project for Prevention and Research on Vector-borne Diseases (SZSM202211023 to G.C.), Yunnan Provincial Science and Technology Project at Southwest United Graduate School (202302AO370010 to G.C.), New Cornerstone Science Foundation through the New Cornerstone Investigator Program (to G.C.) and Xplorer Prize from the Tencent Foundation (to G.C.).
Author information
Authors and Affiliations
Contributions
B.Z., G.C. and L.Y. designed and supervised the study, and wrote and revised the paper. W.P., W.T., B.L. and C.T. performed the experiments and analysed the data. H.W., S.G., X.Z. and S.F. assisted with the animal experiments. W.P., W.T., B.L., P.D. and T.W. contributed to the experimental design and the writing of the paper. All authors reviewed, critiqued and provided comments on the text of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Ke Cheng, Gregg Duncan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–7, Table 1, Discussion and Methods.
Supplementary Data 1 (download XLSX )
Source data for Supplementary Figs. 1–5 and 7.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peng, W., Tai, W., Li, B. et al. Inhalable nanocatalytic therapeutics for viral pneumonia. Nat. Mater. 24, 637–648 (2025). https://doi.org/10.1038/s41563-024-02041-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41563-024-02041-5
This article is cited by
-
Microrobots for pulmonary drug delivery
Nature Reviews Bioengineering (2026)
-
Dual-function TA@CaCO3 nanomedicine targeting M2 macrophage lipid peroxidation sensitivity in rheumatoid arthritis therapy
Journal of Nanobiotechnology (2025)
-
Targeting alveolar macrophages: a promising intervention for pulmonary infection and acute lung injury
Cellular & Molecular Biology Letters (2025)
-
Designing supramolecular catalytic systems for mammalian synthetic metabolism
Nature Reviews Materials (2025)
-
Inhalable nanozymes against pneumonia
Nature Materials (2025)