Abstract
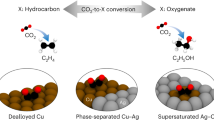
CO electroreduction has recently been explored as an alternative to CO2 electroreduction for multicarbon product formation, because it bypasses the large carbon loss associated with CO2 electroreduction. Although ethylene is generally obtained as the major product, shifting electrolysis towards the production of alcohols is an industrially promising path forward. Here we report a trimetallic-copper-based catalyst, consisting of copper nitride doped with gold nanoparticles and isolated silver atoms, with high selectivity for the formation of C2+ alcohols (Faradic efficiency for ethanol + n-propanol is >70%), within gas-fed flow cells at high current densities. Although active sites are metallic Cu(111) copper atoms derived from copper nitride, gold and silver doping suppresses ethylene formation due to the increased carbophibicity of the catalyst surface, as shown computationally. Overall, these findings open new perspectives regarding the design of catalysts for the production of liquid products from CO.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available in this Article and its Supplementary Information and can be obtained from the corresponding authors upon request.
References
Annual industry outlook. Renewable Fuels Association https://ethanolrfa.org/resources/annual-industry-outlook (2024).
Propanol market size, share & trends analysis report by product by application (n-propanol, isopropyl alcohol), by region, and segment forecasts, 2023–2030. Grand View Research https://www.grandviewresearch.com/industry-analysis/propanol-market (2023).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Fan, L. et al. Strategies in catalysts and electrolyzer design for electrochemical CO2 reduction toward C2+ products. Sci. Adv. 6, eaay3111 (2020).
Hoang, T. T. H. et al. Nanoporous copper–silver alloys by additive-controlled electrodeposition for the selective electroreduction of CO2 to ethylene and ethanol. J. Am. Chem. Soc. 140, 5791–5797 (2018).
Li, Y. C. et al. Binding site diversity promotes CO2 electroreduction to ethanol. J. Am. Chem. Soc. 141, 8584–8591 (2019).
Peng, H.-J., Tang, M. T., Halldin Stenlid, J., Liu, X. & Abild-Pedersen, F. Trends in oxygenate/hydrocarbon selectivity for electrochemical CO(2) reduction to C2 products. Nat. Commun. 13, 1399 (2022).
Kim, J.-Y. et al. Selective hydrocarbon or oxygenate production in CO2 electroreduction over metallurgical alloy catalysts. Nat. Synth. 3, 452–465 (2024).
Clark, E. L., Hahn, C., Jaramillo, T. F. & Bell, A. T. Electrochemical CO2 reduction over compressively strained CuAg surface alloys with enhanced multi-carbon oxygenate selectivity. J. Am. Chem. Soc. 139, 15848–15857 (2017).
Herzog, A. et al. Operando investigation of Ag-decorated Cu2O nanocube catalysts with enhanced CO2 electroreduction toward liquid products. Angew. Chem. Int. Ed. 60, 7426–7435 (2021).
Phong Duong, H. et al. Silver and copper nitride cooperate for CO electroreduction to propanol. Angew. Chem. Int. Ed. 62, e202310788 (2023).
Morales-Guio, C. G. et al. Improved CO2 reduction activity towards C2+ alcohols on a tandem gold on copper electrocatalyst. Nat. Catal. 1, 764–771 (2018).
Jouny, M., Hutchings, G. S. & Jiao, F. Carbon monoxide electroreduction as an emerging platform for carbon utilization. Nat. Catal. 2, 1062–1070 (2019).
Hanselman, S., Koper, M. T. M. & Calle-Vallejo, F. Computational comparison of late transition metal (100) surfaces for the electrocatalytic reduction of CO to C2 species. ACS Energy Lett. 3, 1062–1067 (2018).
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504–507 (2014).
Wang, L. et al. Electrochemically converting carbon monoxide to liquid fuels by directing selectivity with electrode surface area. Nat. Catal. 2, 702–708 (2019).
Jouny, M., Luc, W. & Jiao, F. High-rate electroreduction of carbon monoxide to multi-carbon products. Nat. Catal. 1, 748–755 (2018).
Wang, Y., Raciti, D. & Wang, C. High-flux CO reduction enabled by three-dimensional nanostructured copper electrodes. ACS Catal. 8, 5657–5663 (2018).
Ikeda, M. et al. Context-dependent operation of neural circuits underlies a navigation behavior in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 117, 6178–6188 (2020).
Li, J. et al. Weak CO binding sites induced by Cu–Ag interfaces promote CO electroreduction to multi-carbon liquid products. Nat. Commun. 14, 698 (2023).
Wang, X. et al. Efficient upgrading of CO to C3 fuel using asymmetric C–C coupling active sites. Nat. Commun. 10, 5186 (2019).
Wang, X. et al. Efficient electrosynthesis of n-propanol from carbon monoxide using a Ag–Ru–Cu catalyst. Nat. Energy 7, 170–176 (2022).
Niu, W. et al. Pb-rich Cu grain boundary sites for selective CO-to-n-propanol electroconversion. Nat. Commun. 14, 4882 (2023).
Du, C. et al. Cascade electrocatalysis via AgCu single-atom alloy and Ag nanoparticles in CO2 electroreduction toward multicarbon products. Nat. Commun. 14, 6142 (2023).
Hahn, C. et al. Engineering Cu surfaces for the electrocatalytic conversion of CO2: controlling selectivity toward oxygenates and hydrocarbons. Proc. Natl Acad. Sci. USA 114, 5918–5923 (2017).
Nitopi, S. et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 119, 7610–7672 (2019).
Bobadilla, L. F., Santos, J. L., Ivanova, S., Odriozola, J. A. & Urakawa, A. Unravelling the role of oxygen vacancies in the mechanism of the reverse water–gas shift reaction by operando DRIFTS and ultraviolet–visible spectroscopy. ACS Catal. 8, 7455–7467 (2018).
Ebaid, M. et al. Production of C2/C3 oxygenates from planar copper nitride-derived mesoporous copper via electrochemical reduction of CO2. Chem. Mater. 32, 3304–3311 (2020).
Zhao, C. et al. In situ topotactic transformation of an interstitial alloy for CO electroreduction. Adv. Mater. 32, 2002382 (2020).
Veenstra, F. L. P., Martín, A. J. & Pérez-Ramírez, J. Nitride-derived copper modified with indium as a selective and highly stable catalyst for the electroreduction of carbon dioxide. Chem. Sus. Chem 12, 3501–3508 (2019).
Peng, H. et al. The role of atomic carbon in directing electrochemical CO(2) reduction to multicarbon products. Energy Environ. Sci. 14, 473–482 (2021).
Romero Cuellar, N. S., Wiesner-Fleischer, K., Fleischer, M., Rucki, A. & Hinrichsen, O. Advantages of CO over CO2 as reactant for electrochemical reduction to ethylene, ethanol and n-propanol on gas diffusion electrodes at high current densities. Electrochim. Acta 307, 164–175 (2019).
Sisler, J. et al. Ethylene electrosynthesis: a comparative techno-economic analysis of alkaline vs membrane electrode assembly vs CO2–CO–C2H4 tandems. ACS Energy Lett. 6, 997–1002 (2021).
Huang, W.-D. & Percival Zhang, Y. H. Analysis of biofuels production from sugar based on three criteria: thermodynamics, bioenergetics, and product separation. Energy Environ. Sci. 4, 784–792 (2011).
Overa, S. et al. Enhancing acetate selectivity by coupling anodic oxidation to carbon monoxide electroreduction. Nat. Catal. 5, 738–745 (2022).
Gabardo, C. M. et al. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 3, 2777–2791 (2019).
Luc, W. et al. Two-dimensional copper nanosheets for electrochemical reduction of carbon monoxide to acetate. Nat. Catal. 2, 423–430 (2019).
Schouten, K. J. P., Pérez Gallent, E. & Koper, M. T. M. Structure sensitivity of the electrochemical reduction of carbon monoxide on copper single crystals. ACS Catal. 3, 1292–1295 (2013).
Bagger, A., Ju, W., Varela, A. S., Strasser, P. & Rossmeisl, J. Electrochemical CO2 reduction: classifying Cu facets. ACS Catal. 9, 7894–7899 (2019).
Cheng, D. et al. The nature of active sites for carbon dioxide electroreduction over oxide-derived copper catalysts. Nat. Commun. 12, 395 (2021).
Lum, Y. & Ager, J. W. Evidence for product-specific active sites on oxide-derived Cu catalysts for electrochemical CO2 reduction. Nat. Catal. 2, 86–93 (2019).
Eilert, A. et al. Subsurface oxygen in oxide-derived copper electrocatalysts for carbon dioxide reduction. J. Phys. Chem. Lett. 8, 285–290 (2017).
Feng, X., Jiang, K., Fan, S. & Kanan, M. W. A direct grain–boundary–activity correlation for Coelectroreduction on Cu nanoparticles. ACS Cent. Sci. 2, 169–174 (2016).
Verdaguer-Casadevall, A. et al. Probing the active surface sites for CO reduction on oxide-derived copper electrocatalysts. J. Am. Chem. Soc. 137, 9808–9811 (2015).
Chen, C. et al. The in situ study of surface species and structures of oxide-derived copper catalysts for electrochemical CO2 reduction. Chem. Sci. 12, 5938–5943 (2021).
Ma, G. et al. A hydrophobic Cu/Cu2O sheet catalyst for selective electroreduction of CO to ethanol. Nat. Commun. 14, 501 (2023).
Li, J. et al. Copper adparticle enabled selective electrosynthesis of n-propanol. Nat. Commun. 9, 4614 (2018).
Zhuang, T.-T. et al. Copper nanocavities confine intermediates for efficient electrosynthesis of C3 alcohol fuels from carbon monoxide. Nat. Catal. 1, 946–951 (2018).
Long, C. et al. Steering the reconstruction of oxide-derived Cu by secondary metal for electrosynthesis of n-propanol from CO. J. Am. Chem. Soc. 146, 4632–4641 (2024).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Newville, M. IFEFFIT: interactive XAFS analysis and FEFF fitting. J. Synchrotron Radiat. 8, 322–324 (2001).
Grazulis, S. et al. Crystallography Open Database—an open-access collection of crystal structures. J. Appl. Cryst. 42, 726–729 (2009).
Acknowledgements
This work was financially supported by TotalEnergies SE. The in situ XAS experiments at the SAMBA beamline of Synchrotron SOLEIL have been funded by SOLEIL under User Proposal number 20221501 and by the European Research Council (ERC) Consolidator Grant GENESIS under the European Union’s Horizon 2020 research and innovation programme (grant agreement number 864850). We acknowledge the computing time granted on the high-performance computing (HPC) clusters at TotalEnergies E&P Research & Technology USA. This work was supported by the computational resources provided by Cedar & Cypress. We thank the Houston HPC team for their continuous support and assistance during this project.
Author information
Authors and Affiliations
Contributions
H.P.D. and N.-H.T. contributed to the design of the experiments as well as to acquire and analyse the data. N.-H.T., M.W.S. and M.F. contributed to the conception and design of the work. N.M. performed the TEM and XEDS analyses. J.L. performed the in situ XRD experiments. D.P., A.Z. and Q.A. performed the ex situ and in situ XAS experiments. S.Z. performed the XPS analysis. J.G.R.C. performed the computation work. N.-H.T. and M.F. drafted the work, and M.W.S. substantively revised it.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–29, Tables 1–6 and computational details.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Phong Duong, H., Rivera de la Cruz, J.G., Portehault, D. et al. Incorporation of isolated Ag atoms and Au nanoparticles in copper nitride for selective CO electroreduction to multicarbon alcohols. Nat. Mater. 24, 900–906 (2025). https://doi.org/10.1038/s41563-025-02153-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41563-025-02153-6