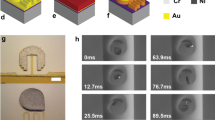
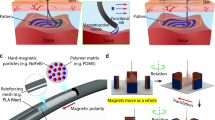
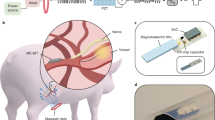
Abstract
Small-scale magnetically actuated catheters capable of remote active navigation have promising applications in minimally invasive surgeries. However, existing fabrication techniques hinder their integration with multimodal sensing components, especially since embedding rigid electronic components within the catheters may diminish their flexibility and controllability. Here we report a magnetically actuated bioelectronic catheter with the in situ multiplexed biosensing of multiple types of metabolite or ion simultaneously. We use four-dimensional multichannel printing to fabricate a flexible multichannel ferromagnetic catheter with a multichannel-sheath structure, comprising six liquid metal microchannels embedded in a polymer matrix for electrical conduction. The catheter can navigate through blood vessels and intestines using magnetically controlled active steering, being used for renal vein or intestines interventional surgeries and in situ multimetabolite sensing on rabbit and porcine models. Overall, the reported magnetically actuated bioelectronic catheter is a promising tool for remotely controlled biosensing and therapies on hard-to-reach lesions during minimally invasive surgery.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data are available in the Article or its Supplementary Information. Source data are provided with this paper.
References
Heikenfeld, J. et al. Accessing analytes in biofluids for peripheral biochemical monitoring. Nat. Biotechnol. 37, 407–419 (2019).
Wang, B. et al. Wearable aptamer-field-effect transistor sensing system for noninvasive cortisol monitoring. Sci. Adv. 8, eabk0967 (2022).
Heitzer, E., Haque, I. S., Roberts, C. E. S. & Speicher, M. R. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat. Rev. Genet. 20, 71–88 (2019).
Loeb, S. et al. Systematic review of complications of prostate biopsy. Eur. Urol. 64, 876–892 (2013).
Li, W. et al. Liquid biopsy in lung cancer: significance in diagnostics, prediction, and treatment monitoring. Mol. Cancer 21, 25 (2022).
Tapper Elliot, B. & Lok Anna, S. F. Use of liver imaging and biopsy in clinical practice. N. Engl. J. Med. 377, 756–768 (2017).
Huang, S. et al. Advances in multifunctional electronic catheters for precise and intelligent diagnosis and therapy in minimally invasive surgery. ACS Nano 18, 18129–18150 (2024).
Lee, S. Y. et al. Needle-compatible miniaturized optoelectronic sensor for pancreatic cancer detection. Sci. Adv. 6, eabc1746 (2020).
Kim, D.-H. et al. Materials for multifunctional balloon catheters with capabilities in cardiac electrophysiological mapping and ablation therapy. Nat. Mater. 10, 316–323 (2011).
Huang, S. et al. Petromyzontidae-biomimetic multimodal microneedles-integrated bioelectronic catheters for theranostic endoscopic surgery. Adv. Funct. Mater. 33, 2214485 (2023).
Kim, Y., Parada, G. A., Liu, S. & Zhao, X. Ferromagnetic soft continuum robots. Sci. Robot. 4, eaax7329 (2019).
Cianchetti, M., Laschi, C., Menciassi, A. & Dario, P. Biomedical applications of soft robotics. Nat. Rev. Mater. 3, 143–153 (2018).
Runciman, M., Darzi, A. & Mylonas, G. P. Soft robotics in minimally invasive surgery. Soft Robot. 6, 423–443 (2019).
Yang, G. Z. et al. The grand challenges of Science Robotics. Sci. Robot. 3, eaar7650 (2018).
Hu, W., Lum, G. Z., Mastrangeli, M. & Sitti, M. Small-scale soft-bodied robot with multimodal locomotion. Nature 554, 81–85 (2018).
Kim, Y., Yuk, H., Zhao, R., Chester, S. A. & Zhao, X. Printing ferromagnetic domains for untethered fast-transforming soft materials. Nature 558, 274–279 (2018).
Hwang, J. et al. An electromagnetically controllable microrobotic interventional system for targeted, real-time cardiovascular intervention. Adv. Healthc. Mater. 11, 2102529 (2022).
Kim, Y. et al. Telerobotic neurovascular interventions with magnetic manipulation. Sci. Robot. 7, eabg9907 (2022).
Ze, Q. et al. Magnetic shape memory polymers with integrated multifunctional shape manipulation. Adv. Mater. 32, 1906657 (2020).
Wang, L., Kim, Y., Guo, C. F. & Zhao, X. Hard-magnetic elastica. J. Mech. Phys. Solids 142, 104045 (2020).
Zhao, R., Kim, Y., Chester, S. A., Sharma, P. & Zhao, X. Mechanics of hard-magnetic soft materials. J. Mech. Phys. Solids 124, 244–263 (2019).
Zhang, Y. et al. Submillimeter multifunctional ferromagnetic fiber robots for navigation, sensing, and modulation. Adv. Healthc. Mater. 12, 2300964 (2023).
Ma, J. et al. Shaping a soft future: patterning liquid metals. Adv. Mater. 35, 2205196 (2023).
Yang, Z. et al. From liquid metal to stretchable electronics: overcoming the surface tension. Sci. China Mater. 65, 2072–2088 (2022).
Zhao, Z., Soni, S., Lee, T., Nijhuis, C. A. & Xiang, D. Smart eutectic gallium–indium: from properties to applications. Adv. Mater. 35, 2203391 (2023).
Zhang, J., Soon, R. H., Wei, Z., Hu, W. & Sitti, M. Liquid metal-elastomer composites with dual-energy transmission mode for multifunctional miniature untethered magnetic robots. Adv. Sci. 9, 2203730 (2022).
Lee, S., Lee, Y. J., Kim, J. H. & Lee, G.-J. Electrochemical detection of H2O2 released from prostate cancer cells using Pt nanoparticle-decorated rGO-CNT nanocomposite-modified screen-printed carbon electrodes. Chemosensors 8, 63 (2020).
Meskher, H. et al. A review on CNTs-based electrochemical sensors and biosensors: unique properties and potential applications. Crit. Rev. Anal. Chem. 54, 2398–2421 (2024).
Wang, L. et al. Functionalized helical fibre bundles of carbon nanotubes as electrochemical sensors for long-term in vivo monitoring of multiple disease biomarkers. Nat. Biomed. Eng. 4, 159–171 (2020).
Serra-Maia, R. et al. Mechanism and kinetics of hydrogen peroxide decomposition on platinum nanocatalysts. ACS Appl. Mater. Interfaces 10, 21224–21234 (2018).
Tao, Z., Si, H., Zhang, X., Liao, J. & Lin, S. Highly sensitive and selective H2O2 sensors based on ZnO TFT using PBNCs/Pt-NPs/TNTAs as gate electrode. Sens. Actuators B 349, 130791 (2021).
Berberich, J. A., Yang, L. W., Bahar, I. & Russell, A. J. A stable three enzyme creatinine biosensor. 2. Analysis of the impact of silver ions on creatine amidinohydrolase. Acta Biomater. 1, 183–191 (2005).
Hsiue, G.-H., Lu, P.-L. & Chen, J.-C. Multienzyme-immobilized modified polypropylene membrane for an amperometric creatinine biosensor. J. Appl. Polym. Sci. 92, 3126–3134 (2004).
Liu, Y. et al. Uric acid and creatinine biosensors with enhanced room-temperature storage stability by a multilayer enzyme matrix. Anal. Chim. Acta 1227, 340264 (2022).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Huang, S. et al. Semi-implantable device based on multiplexed microfilament electrode cluster for continuous monitoring of physiological ions. Bio-Des. Manuf. 7, 88–103 (2024).
Huang, X. et al. 3D-assembled microneedle ion sensor-based wearable system for the transdermal monitoring of physiological ion fluctuations. Microsyst. Nanoeng. 9, 25 (2023).
Gong, X. et al. Polymer hydrogel-based multifunctional theranostics for managing diabetic wounds. Adv. Funct. Mater. 34, 2315564 (2024).
Bandodkar, A. J. et al. Tattoo-based potentiometric ion-selective sensors for epidermal pH monitoring. Analyst 138, 123–128 (2013).
Mandal, R., Panda, S. K. & Nayak, S. Rheology of concrete: critical review, recent advancements, and future prospectives. Constr. Build. Mater. 392, 132007 (2023).
Cánovas, R., Cuartero, M. & Crespo, G. A. Modern creatinine (bio)sensing: challenges of point-of-care platforms. Biosens. Bioelectron. 130, 110–124 (2019).
Kumar, P., Jaiwal, R. & Pundir, C. S. An improved amperometric creatinine biosensor based on nanoparticles of creatininase, creatinase and sarcosine oxidase. Anal. Biochem. 537, 41–49 (2017).
Kassal, P. et al. Smart bandage with wireless connectivity for uric acid biosensing as an indicator of wound status. Electrochem. Commun. 56, 6–10 (2015).
Liu, Z. et al. Integrated multiplex sensing bandage for in situ monitoring of early infected wounds. ACS Sens. 6, 3112–3124 (2021).
Mehmood, N., Hariz, A., Fitridge, R. & Voelcker, N. H. Applications of modern sensors and wireless technology in effective wound management. J. Biomed. Mater. Res. Part B 102B, 885–895 (2014).
Cutting, K. F. Wound exudate: composition and functions. Br. J. Community Nurs. 8, S4–S9 (2003).
Kellum, J. A. et al. Acute kidney injury. Nat. Rev. Dis. Prim. 7, 52 (2021).
Turgut, F., Awad, A. S. & Abdel-Rahman, E. M. Acute kidney injury: medical causes and pathogenesis. J. Clin. Med. 12, 375 (2023).
Kao, C.-C., Yang, W.-S., Fang, J.-T., Liu, K. D. & Wu, V.-C. Remote organ failure in acute kidney injury. J. Formos. Med. Assoc. 118, 859–866 (2019).
Hilton, R. Defining acute renal failure. Can. Med. Assoc. J. 183, 1167–1169 (2011).
Giordano, C., Karasik, O., King-Morris, K. & Asmar, A. Uric acid as a marker of kidney disease: review of the current literature. Dis. Markers 2015, 382918 (2015).
Hilton, R. Acute renal failure. Br. Med. J. 333, 786 (2006).
Acknowledgements
This research was supported by the National Natural Science Foundation of China under grant numbers T2225010 (X.X.), 32171399 (X.X.), 32171456 (H.C.) and 52305442 (Y.Z.); Guangdong Basic and Applied Basic Research Foundation under grant numbers 2023A1515011267 (X.X.), 2023A1515111139 (X.H.) and 2025A1515010188 (Y.Z.); Natural Science Foundation of Guangdong Province under grant number 2022B1515020011 (L.J.); Shenzhen Science and Technology Program under grant numbers JCYJ20220818102201003 (L.J.), KCXFZ20230731094500001 (L.J.) and JCYJ20220818102201002 (L.J.); Science and Technology Program of Guangzhou, China, under grant numbers 2024B03J0121 (X.X.) and 2024B03J1284 (H.C.); and the Fundamental Research Funds for the Central Universities, Sun Yat-sen University, under grant number 24xkjc011 (X.X.).
Author information
Authors and Affiliations
Contributions
X.X. and L.J. conceived and designed the project. J.Y. and Y.Z. performed most of the experiments, analysed the data and prepared the manuscript. L.J. and X.X. supervised the work, guided the research and edited the manuscript. Z.L., S.H. and X.H. helped with the data analysis, references and graphics preparation. S.H., X.H., S.Z. and F.C. contributed to the in vivo experiments and data collection. J.Y., Y.Z. and T.W. developed the eight-axis coil magnetic control system. Y.W., Y.T., M.L., L.X., H.C. and J.L. guided the animal experiments. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Hongsoo Choi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–17, Figs. 1–53, Table 1, captions to Supplementary Videos 1–5 and refs. 1–16.
Supplementary Data 1 (download XLSX )
Source data for Supplementary Figs. 2, 4–6, 8, 9, 12, 15, 17–20, 31, 32, 42 and 45–49.
Supplementary Video 1 (download MP4 )
Demonstration of MMBC lighting up the LED array at different coordinates.
Supplementary Video 2 (download MP4 )
Demonstration of MMBC navigating through a 3D tortuous vascular phantom network.
Supplementary Video 3 (download MP4 )
Experimental demonstration of MMBC selectively steering through a set of rings.
Supplementary Video 4 (download MP4 )
Navigation process of MMBC within the rabbit’s intestines.
Supplementary Video 5 (download MP4 )
Selective navigation of MMBC from the inferior vena cava into the upper branch of the renal vein.
Source data
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2e–t.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 2d–n.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4d–f.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5e–i.
Source Data Fig. 6 (download XLSX )
Source data for Fig. 6i.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, J., Zhang, Y., Liu, Z. et al. Magnetically actuated multimodal bioelectronic catheter for minimally invasive surgery and sensing. Nat. Mater. 24, 2019–2031 (2025). https://doi.org/10.1038/s41563-025-02340-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41563-025-02340-5