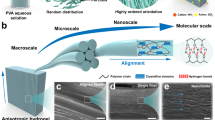
Abstract
Hydrogels are prevalent materials with applications ranging from drug delivery systems, contact lenses and tissue engineering scaffolds. However, they require considerable perturbation to observe their nanoscale, solution-phase structures necessary for predicting bulk properties. Although studies suggest that methylcellulose, a quintessential hydrogel material, can be described by a semiflexible biopolymer network model, there remain demonstrable inconsistencies in the predicted concentration dependence of rheological properties and in the observation of higher-order features. Here we image solvated hydrogels with high spatiotemporal resolution via liquid-phase transmission electron microscopy to avoid desolvation and shear artefacts. Corroborated by scattering and scanning electron microscopy, we observe that methylcellulose hydrogels form a network with high persistence length and micrometre-scale fibril bundles arranged in hierarchical assemblies, providing a more accurate prediction of bulk rheology. In addition, network structures are observed for hydroxypropyl methylcellulose and hydroxypropyl cellulose. These observations across multiple-length scales lead to a clearer understanding of how nanoscale structure impacts microscale structure and macroscopic behaviour, aiding the development of more accurate structure–property relationships for hydrogel materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The raw data used in this Article are publicly available via Figshare at https://doi.org/10.6084/m9.figshare.30855128 (ref. 48).
References
Arvidson, S. A. et al. Interplay of phase separation and thermoreversible gelation in aqueous methylcellulose solutions. Macromolecules 46, 300–309 (2012).
Sarkar, N. Thermal gelation properties of methyl and hydroxypropyl methylcellulose. J. Appl. Polym. Sci. 24, 1073–1087 (1979).
Chang, C. & Zhang, L. Cellulose-based hydrogels: Present status and application prospects. Carbohydr. Polym. 84, 40–53 (2011).
Arca, H. C. et al. Pharmaceutical applications of cellulose ethers and cellulose ether esters. Biomacromolecules 19, 2351–2376 (2018).
Aigoin, J. et al. Comparative analysis of electron microscopy techniques for hydrogel microarchitecture characterization: SEM, cryo-SEM, ESEM, and TEM. ACS Omega 10, 14687–14698 (2025).
Lodge, T. P. et al. Gelation, phase separation, and fibril formation in aqueous hydroxypropylmethylcellulose solutions. Biomacromolecules 19, 816–824 (2018).
Lott, J. R., McAllister, J. W., Arvidson, S. A., Bates, F. S. & Lodge, T. P. Fibrillar structure of methylcellulose hydrogels. Biomacromolecules 14, 2484–2488 (2013).
Lott, J. R. et al. Fibrillar structure in aqueous methylcellulose solutions and gels. Macromolecules 46, 9760–9771 (2013).
McAllister, J. W. et al. Linear and nonlinear rheological behavior of fibrillar methylcellulose hydrogels. ACS Macro Lett. 4, 538–542 (2015).
Schmidt, P. W. et al. Internal structure of methylcellulose fibrils. Macromolecules 53, 398–405 (2020).
Schmidt, P. W. et al. Molecular weight dependence of methylcellulose fibrillar networks. Macromolecules 51, 7767–7775 (2018).
Siepmann, J. & Peppas, N. A. Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Adv. Drug Deliv. Rev. 64, 163–174 (2012).
Toda, I., Shinozaki, N. & Tsubota, K. Hydroxypropyl methylcellulose for the treatment of severe dry eye associated with Sjögren’s syndrome. Cornea 15, 120–128 (1996).
Morozova, S. Methylcellulose fibrils: a mini review. Polym. Int. 69, 125–130 (2019).
Korpanty, J., Parent, L. R., Hampu, N., Weigand, S. & Gianneschi, N. C. Thermoresponsive polymer assemblies via variable temperature liquid-phase transmission electron microscopy and small angle X-ray scattering. Nat. Commun. 12, 6568 (2021).
MacKintosh, F. C., Kas, J. & Janmey, P. A. Elasticity of semiflexible biopolymer networks. Phys. Rev. Lett. 75, 4425–4428 (1995).
Prince, E. & Kumacheva, E. Design and applications of man-made biomimetic fibrillar hydrogels. Nat. Rev. Mater. 4, 99–115 (2019).
Li, L. Thermal Gelation of Methylcellulose in Water, scaling and thermoreversibility. Macromolecules 35, 5990–5998 (2002).
Chen, S. et al. Microstructures and rheological properties of tilapia fish-scale collagen hydrogels with aligned fibrils fabricated under magnetic fields. Acta Biomater. 7, 644–652 (2011).
Guo, Y. Z. et al. Facile preparation of cellulose hydrogel with Achilles tendon-like super strength through aligning hierarchical fibrous structure. Chem. Eng. J. https://doi.org/10.1016/j.cej.2021.1320402022.
Gnanasekaran, K., Vailonis, K. M., Jenkins, D. M. & Gianneschi, N. C. In situ monitoring of the seeding and growth of silver metal–organic nanotubes by liquid-cell transmission electron microscopy. ACS Nano 14, 8735–8743 (2020).
Korpanty, J., Wang, C. & Gianneschi, N. C. Upper critical solution temperature polymer assemblies via variable temperature liquid phase transmission electron microscopy and liquid resonant soft X-ray scattering. Nat. Commun. 14, 3441 (2023).
Parent, L. R. et al. Tackling the challenges of dynamic experiments using liquid-cell transmission electron microscopy. Acc. Chem. Res. 51, 3–11 (2018).
Ross, F. M. Opportunities and challenges in liquid cell electron microscopy. Science 350, aaa9886 (2015).
Vailonis, K. M., Gnanasekaran, K., Powers, X. B., Gianneschi, N. C. & Jenkins, D. M. Elucidating the growth of metal-organic nanotubes combining isoreticular synthesis with liquid-cell transmission electron microscopy. J. Am. Chem. Soc. 141, 10177–10182 (2019).
Morozova, S., Schmidt, P. W., Bates, F. S. & Lodge, T. P. Effect of poly(ethylene glycol) grafting density on methylcellulose fibril formation. Macromolecules 51, 9413–9421 (2018).
Ginzburg, V. V., Sammler, R. L., Huang, W. & Larson, R. G. Anisotropic self-assembly and gelation in aqueous methylcellulose-theory and modeling. J. Polym. Sci. Part B 54, 1624–1636 (2016).
Coughlin, M. L. et al. Methyl cellulose solutions and gels: fibril formation and gelation properties. Progress. Polym. Sci. https://doi.org/10.1016/j.progpolymsci.2020.101324 (2021).
Aime, S., Cipelletti, L. & Ramos, L. Power law viscoelasticity of a fractal colloidal gel. J. Rheol. 62, 12 (2018).
McDowall, D., Adams, D. J. & Seddon, A. M. Using small angle scattering to understand low molecular weight gels. Soft Matter 18, 1577–1590 (2022).
Terech, P. et al. Structural variations in a family of orthodialkoxyarenes organogelators. J. Colloid Interface Sci. 302, 633–642 (2006).
Jensen, G. V. & Barker, J. G. Effects of multiple scattering encountered for various small-angle scattering model functions. J. Appl. Crystallogr. 51, 1455–1466 (2018).
Richardson, S., Andersson, T., Brinkmalm, G. & Wittgren, B. Analytical approaches to improved characterization of substitution in hydroxypropyl cellulose. Anal. Chem. 75, 6077–6083 (2003).
Zierold, K. X-ray microanalysis of freeze-dried and frozen-hydrated cryosections. J. Electron Microsc. Tech. 9, 17 (1988).
Zierold, K. The morphology of ultrathin cryosections. Ultramicroscopy 14, 9 (1984).
Meijering, E. et al. Design and validation of a tool for neurite tracing and analysis in fluorescence microscopy images. Cytometry A 58, 167–176 (2004).
Usov, I. et al. Understanding nanocellulose chirality and structure–properties relationship at the single fibril level. Nat. Commun. 6, 7564 (2015).
Loong, C. K., Zhou, H. X. & Chase, P. B. Persistence length of human cardiac alpha-tropomyosin measured by single molecule direct probe microscopy. PLoS ONE 7, e39676 (2012).
Laour, G., Kirkegaard, J. B., Li, H., Knowles, T. P. J. & Gsponer, J. Easyworm: an open-source software tool to determine the mechanical properties of worm-like chains. Source Code Biol. Med. 9, 16 (2014).
Lian, W. Z. et al. Tough hydrogels with dynamic H-bonds: structural heterogeneities and mechanical performances. Macromolecules https://doi.org/10.1021/acs.macromol.1c01064 (2021).
Sarrigiannidis, S. O. et al. A tough act to follow: collagen hydrogel modifications to improve mechanical and growth factor loading capabilities. Mater. Today Bio 10, 100098 (2021).
Loveday, S. M., Rao, M. A., Creamer, L. K. & Singh, H. Factors affecting rheological characteristics of fibril gels: the case of beta-lactoglobulin and alpha-lactalbumin. J. Food Sci. 74, R47–R55 (2009).
Rehm, C. et al. Design and performance of the variable-wavelength Bonse–Hart ultra-small-angle neutron scattering diffractometer KOOKABURRA at ANSTO. J. Appl. Crystallogr. 51, 1–8 (2018).
Wood, K. et al. QUOKKA, the pinhole small-angle neutron scattering instrument at the OPAL Research Reactor, Australia: design, performance, operation and scientific highlights. J. Appl. Crystallogr. 51, 294–314 (2018).
Kline, S. R. Reduction and analysis of SANS and USANS data using IGOR Pro. J. Appl. Crystallogr. 39, 895–900 (2006).
Dhakal, S., Chen, Z., Estrin, D. & Morozova, S. Spatially-resolved network dynamics of poly(vinyl alcohol) gels measured with dynamic small angle light scattering. Gels https://doi.org/10.3390/gels8070394 (2022).
Hammouda, B. A new Guinier–Porod model. J. Appl. Crystallogr. 43, 716–719 (2010).
Rosenmann, N. D. et al. Raw data for “Prediction of rheological properties via structure elucidation of solvated hydrogels”. Figshare https://doi.org/10.6084/m9.figshare.30855128 (2025).
Acknowledgements
Experimental work was supported by the Department of Defense, Army Research Office (W911NF-17-1-0326) and a joint research grant by the National Science Foundation (CHE-MSN 1905270). Equipment was supported by a DURIP/ARO (LCTEM holder) and the National Institutes of Health (S10-OD026871). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Health. The work made use of the EPIC facility of Northwestern University’s NUANCE Center, which has received support from the SHyNE Resource (NSF ECCS-2025633), the IIN, and Northwestern’s MRSEC programme (NSF DMR-1720139). This work made use of the BioCryo facility (RRID:SCR_021288) of Northwestern University’s NUANCE Center, which has received support from the IIN and Northwestern’s MRSEC program (NSF DMR-2308691). This work made use of the MatCI Facility, which receives support from the MRSEC Program (NSF DMR-1720139) of the Materials Research Center at Northwestern University. This work was performed at the DuPont-Northwestern-Dow Collaborative Access Team (DND-CAT) located at Sector 5 of the Advanced Photon Source (APS). DND-CAT is supported by Northwestern University, the Dow Chemical Company and DuPont de Nemours, Inc. This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. Data were collected using an instrument funded by the National Science Foundation under award no. 0960140. We also acknowledge the support of the Australian Nuclear Science and Technology Organisation (ANSTO) in providing USANS and SANS beam facilities (proposal no. P16668). N.C.G. and N.D.R. thank postdoctoral fellow Y. Chen for graphical depictions shown in Supplementary Figs. 7 and 8. N.C.G. and N.D.R. also thank S. Aman for her assistance with collecting additional SAXS data and assistance with performing additional rheological measurements.
Author information
Authors and Affiliations
Contributions
N.D.R. conceived of the project together with N.C.G. and J.K., and performed all sample preparation, VT-LCTEM, cryo-TEM, SAXS, rheology experiments and data analysis. L.M.I. assisted with estimating persistence length and image analysis. J.K. trained N.D.R. and assisted with VT-LCTEM and cryo-TEM experiments, figure development and writing the manuscript. Both E.W.R. and R.B. assisted with the freeze-drying method development. E.W.R. assisted with freeze-dried SEM imaging, assisted with cryo-TEM experiments and gave guidance with freeze-drying sample preparation. R.B. assisted with freeze-dried SEM imaging. J.P.M. assisted in acquiring USANS data and provided guidance to N.D.R. for USANS and SANS data analysis. K.W. assisted in acquiring SANS data. N.N. performed SALS measurements. B.S.S. aided in data analysis and the project strategy. Y.C. assisted in figure development and LCTEM data analysis. S.J.W. assisted in collecting SAXS data. M.J.A.H. assisted in re-analysing previously published USANS/SANS data, provided guidance to N.D.R. for USANS and SANS data analysis, and analysed SALS measurements. B.S.S. assisted in guidance of data interpretation and aided in writing the manuscript. N.C.G. conceived of and supervised all aspects of the research programme and cowrote the manuscript. All authors have approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Lucy Liberman, Taylor Woehl and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Discussion, Figs. 1–26 and Tables 1–12.
Supplementary Video 1 (download MP4 )
LCTEM movie of the gelation of MC, related to Fig. 3.
Supplementary Video 2 (download MP4 )
LCTEM movie of the gelation of MC, related to Supplementary Figs. 6 and 7.
Supplementary Video 3 (download MP4 )
LCTEM movie of the gelation of MC, related to Supplementary Figs. 6 and 8.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rosenmann, N.D., Irie, L.M., Korpanty, J. et al. Prediction of rheological properties via structure elucidation of solvated hydrogels. Nat. Mater. (2026). https://doi.org/10.1038/s41563-026-02491-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41563-026-02491-z