Abstract
Mg3(Sb, Bi)2 is the most promising candidate as a next-generation thermoelectric cooling material; however, its application is bottlenecked by poor moisture stability. We demonstrate a protection strategy for Mg3(Sb, Bi)2 by constructing anodic phases that are preferentially corroded to protect the cathodic material matrix, as enabled by the in situ formation of uniformly distributed multiscale anodic phases based on a large Pilling–Bedworth ratio, low equilibrium potential, high chemical inertness and rapid oxide/hydroxide coverage ability. Mg17Al12 preferentially corrodes and promotes the formation of a protective film, reducing the average corrosion rate of Mg3(Sb, Bi)2 by 92% to ~95 μm year−1 in air and 86% to ~0.36 μm h−1 in water, achieving excellent corrosion resistance. The cooling performance of the fabricated module is comparable with that of commercial bismuth telluride modules at 300 K, and exceeds them at 325 K and 350 K. Meanwhile, no performance degradation is observed after 28-day aging at 350 K and 70% relative humidity. Our study addresses the issues of moisture stability of Mg3(Sb, Bi)2 during storage, processing and application, and could be extended to other aqueous vapour-sensitive materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in the Article and its Supplementary Information. Source data are provided with this paper.
References
Bian, L. et al. Observation of micro-droplets on metal surface in early atmospheric corrosion. Electrochem. Commun. 7, 1033–1038 (2005).
Xu, N. et al. Laboratory observation of dew formation at an early stage of atmospheric corrosion of metals. Corros. Sci. 44, 163–170 (2002).
Xu, C. et al. Device-level optimization of n-type Mg3(Sb, Bi)2-based thermoelectric modules toward applications: a perspective. Adv. Funct. Mater. 33, 2304173 (2023).
Yang, J. et al. Next-generation thermoelectric cooling modules based on high-performance Mg3(Bi,Sb)2 material. Joule 6, 193–204 (2022).
Li, J. W. et al. Wide-temperature-range thermoelectric n-type Mg3(Sb,Bi)2 with high average and peak zT values. Nat. Commun. 14, 7428 (2023).
Mao, J. et al. High thermoelectric cooling performance of n-type Mg3Bi2-based materials. Science 365, 495–498 (2019).
Zhao, P. et al. Plasticity in single-crystalline Mg3Bi2 thermoelecric material. Nature 631, 777–782 (2024).
Mishra, P. et al. First-principles prediction of electrochemical polarization and mechanical behavior in Mg based intermetallics. Comput. Mater. Sci. 214, 111667 (2022).
Luo, Z. et al. Theoretical analysis of the galvanic corrosion behavior of Mg-Ge binary alloy. J. Electrochem. Soc. 166, C421–C427 (2019).
Wang, Y. et al. First-principles study of water decomposition and hydrogen evolution on MgZn2 Laves phase. Comput. Mater. Sci. 196, 110532 (2021).
Zhu, Q. et al. Towards development of a high-strength stainless Mg alloy with Al-assisted growth of passive film. Nat. Commun. 13, 5838 (2022).
Liu, H. et al. Review of the atmospheric corrosion of magnesium alloys. J. Mater. Sci. Technol. 35, 2003–2016 (2019).
Li, A. et al. Chemical stability and degradation mechanism of Mg3Sb2–xBix thermoelectrics towards room-temperature applications. Acta Mater. 239, 118301 (2022).
Song, G.-L. et al. Recently deepened insights regarding Mg corrosion and advanced engineering applications of Mg alloys. J. Magnes. Alloy. 11, 3948–3991 (2023).
Wu, X. et al. Revealing the chemical instability of Mg3Sb2–xBix-based thermoelectric materials. ACS Appl. Mater. Interfaces 15, 50216–50224 (2023).
Choy, K. L. Chemical vapour deposition of coatings. Prog. Mater Sci. 48, 57–170 (2003).
Wu, X. et al. Interface and surface engineering realized high efficiency of 13% and improved thermal stability in Mg3Sb1.5Bi0.5-based thermoelectric generation devices. Adv. Energy Mater. 12, 2203039 (2022).
Ou, Y. X. et al. Recent advances and strategies for high-performance coatings. Prog. Mater Sci. 136, 101125 (2023).
Yoon, J. et al. Wet-style superhydrophobic antifogging coatings for optical sensors. Adv. Mater. 32, e2002710 (2020).
Duarte, M. J. et al. Element-resolved corrosion analysis of stainless-type glass-forming steels. Science 341, 372–376 (2013).
Williams, D. E. et al. Passivity breakdown and pitting corrosion of binary alloys. Nature 350, 216–219 (1991).
Sedriks, A. J. et al. Electrochemical behavior of Ti-Ni alloys in acidic chloride solutions. Corrosion 28, 137–142 (1972).
Tian, H. et al. Steel anti-corrosion strategy enables long-cycle Zn anode. Adv. Energy Mater. 13, 2202603 (2022).
Wu, Y. et al. State of the art and current trends on the metal corrosion and protection strategies in deep sea. J. Mater. Sci. Technol. 215, 192–213 (2025).
Xu, C. et al. Pilling-Bedworth ratio for oxidation of alloys. Mater. Res. Innovations 3, 231–235 (2000).
Greenwood, N. N. et al. Chemistry of the Elements 2nd edn (Elsevier, 2012).
Dean, J. A. Lange’s Handbook of Chemistry 15th edn (McGraw-Hill, 1999).
Xie, L. et al. Screening strategy for developing thermoelectric interface materials. Science 382, 921–928 (2023).
Yin, L. et al. CALPHAD accelerated design of advanced full-Zintl thermoelectric device. Nat. Commun. 15, 1468 (2024).
Tamaki, H. et al. Isotropic conduction network and defect chemistry in Mg3+δSb2-based layered Zintl compounds with high thermoelectric performance. Adv. Mater. 28, 10182–10187 (2016).
Saal, J. E. et al. Materials design and discovery with high-throughput density functional theory: the Open Quantum Materials Database (OQMD). JOM 65, 1501–1509 (2013).
Kirklin, S. et al. The Open Quantum Materials Database (OQMD): assessing the accuracy of DFT formation energies. npj Comput. Mater. 1, 15010 (2015).
Wang, S. et al. Electrochemical impedance spectroscopy. Nat. Rev. Methods Primers 1, 41 (2021).
Collins, L. et al. Towards nanoscale electrical measurements in liquid by advanced KPFM techniques: a review. Rep. Prog. Phys. 81, 086101 (2018).
Yan, Y. et al. Corrosion behavior of stainless steel-tungsten carbide joints brazed with AgCuX (X = In, Ti) alloys. Corros. Sci. 200, 110231 (2022).
Liu, J. et al. The special role of anodic second phases in the micro-galvanic corrosion of EW75 Mg alloy. Electrochim. Acta 189, 190–195 (2016).
Song, Y. et al. Pitting corrosion of a rare earth Mg alloy GW93. J. Mater. Sci. Technol. 33, 954–960 (2017).
Santamaria, M. et al. Initial surface film on magnesium metal: a characterization by X-ray photoelectron spectroscopy (XPS) and photocurrent spectroscopy (PCS). Electrochim. Acta 53, 1314–1324 (2007).
Naumkin, A. V. et al. NIST X-ray Photoelectron Spectroscopy Database (National Institute of Standards and Technology, 2008).
Zhang, F. et al. Corrosion resistance of Mg-Al-LDH coating on magnesium alloy AZ31. Surf. Coat. Technol. 258, 1152–1158 (2014).
Esmaily, M. et al. Fundamentals and advances in magnesium alloy corrosion. Prog. Mater Sci. 89, 92–193 (2017).
Guo, L. et al. Layered double hydroxide coatings on magnesium alloys: a review. J. Mater. Sci. Technol. 34, 1455–1466 (2018).
Song, G. Recent progress in corrosion and protection of magnesium alloys. Adv. Eng. Mater. 7, 563–586 (2005).
Qu, N. et al. Interfacial design contributing to high conversion efficiency in Mg3(Sb, Bi)2/Bi2Te3 thermoelectric module with superior stability. Adv. Energy Mater. 14, 2302818 (2023).
Uhlig, H. H. & Revie, R. W. Corrosion and Corrosion Control 3rd edn (Wiley, 1985).
Chen, N. et al. Improved figure of merit (z) at low temperatures for superior thermoelectric cooling in Mg3(Bi,Sb)2. Nat. Commun. 14, 4932 (2023).
Zhang, X. et al. High-performance MgAgSb/Mg3(Sb,Bi)2-based thermoelectrics with η = 12% at T ≤ 583K. Joule 8, 3324–3335 (2024).
Ma, X. et al. Elevating thermoelectric performance in the sub-ambient temperature range for electronic refrigeration. Innovation 6, 100864 (2025).
Liu, Z. et al. Maximizing the performance of n-type Mg3Bi2 based materials for room-temperature power generation and thermoelectric cooling. Nat. Commun. 13, 1120 (2022).
Zhang, Q. et al. Enhancing Mg3(Bi, Sb)2 thermoelectric performance via ZrSb1–x modified grain-boundary. Mater. Today Phys. 54, 101733 (2025).
Ying, P. et al. A robust thermoelectric module based on MgAgSb/Mg3(Sb,Bi)2 with a conversion efficiency of 8.5% and a maximum cooling of 72 K. Energy Environ. Sci. 15, 2557–2566 (2022).
Liu, Y. et al. Mg compensating design in the melting-sintering method for high-performance Mg3(Bi, Sb)2 thermoelectric devices. Small 19, 2303840 (2023).
Xu, W. et al. Amorphous carbon-modulated Mg3(Bi,Sb)2 and electron-poor CdSb for ultralow-cost Te-free refrigeration modules. Adv. Funct. Mater. 35, 2414194 (2024).
Xie, L. et al. Highly efficient thermoelectric cooling performance of ultrafine-grained and nanoporous materials. Mater. Today 65, 5–13 (2023).
Acknowledgements
J.S. acknowledges the National Key Research and Development Program of China (grant number 2023YFB3809400) and the National Natural Science Foundation of China (grant numbers U23A20556 and 52130106). W.L. acknowledges the Key-Area Research and Development Program of Guangdong Province (grant number 2024B0101040002).
Author information
Authors and Affiliations
Contributions
Z.Y., F.G., W.L. and J.S. initiated the concept and established the experimental scheme. Z.Y., F.G. and J.S. proposed the screening strategy for the anodic phase. Z.Y., Y.S., Jinsuo Hu and M.L. synthesized the samples and performed the characterizations. Z.Y. and Jin Hu performed the data analysis of corrosion-related characterizations. Z.Y., Haijun Wu and X.Z. performed the scanning TEM characterizations. Z.Y., L.W. and J.Z. provided theoretical support for the thermodynamic data calculations. Z.Y., Y.S., H.T. and Hao Wu measured the module performance. Z.Y., Y.S., F.G., Jin Hu, M.L., Y.Z., Z.L., W.C., W.L. and J.S. analysed the results. Z.Y., F.G., Y.S., W.L. and J.S. completed the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Lars-Gunnar Johansson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–7, Figs. 1–39, Tables 1–7 and references.
Supplementary Video 1 (download MP4 )
In situ-formed protective film of the material exhibits excellent self-healing ability.
Supplementary Video 2 (download MP4 )
Comparison of the corrosion resistance of MB and modifed MBA0.20 samples in water.
Supplementary Video 3 (download MP4 )
Comparison of the corrosion resistance of Fe/MBA0.20/Fe and Mg17 Al12/MBA0.20 /Mg17 Al12 samples in water, highlighting the bulk Mg17Al12 used as an interfacial material for the modified Mg3 (Sb, Bi)2 to further delay corrosion.
Source data
Source Data Fig. 1 (download XLSX )
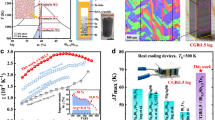
Maximum temperature difference variation data of the Mg17Al12/MBA0.20T/Mg17Al12 module after aging test at 350 K and constant 70% RH plotted in Fig. 1g.
Source Data Fig. 2 (download XLSX )
Equilibrium potential and P–B ratio data of typical metallic elements plotted in Fig. 2a and ranking data of equilibrium potentials for second phases and hydroxide solubility product of different elements plotted in Fig. 2c.
Source Data Fig. 3 (download XLSX )
Potentiodynamic polarization curves data plotted in Fig. 2a, electrochemical impedance spectroscopy Nyquist data plotted in Fig. 2b, SKPFM analysis data of the MBA0.20 alloy plotted in Fig. 2e, and chemical states of O1s, Mg2p and Al2p at different etching depths plotted in Fig. 2i–k.
Source Data Fig. 4 (download XLSX )
Work-function data of MB, MBA0.20 and Mg17Al12 samples plotted in Fig. 2b, coefficient of thermal expansion data of MB, MBA0.20, Fe and Mg17Al12 phases plotted in Fig. 2c, contact resistivity data of the Mg17Al12/MBA0.20/Mg17Al12 joint plotted in Fig. 2d, relative internal resistance change data with aging time of modules plotted in Fig. 2e, zT value data of MBAx and MBA0.20T samples plotted in Fig. 2f, cooling performance data of modules after 28 days of aging plotted in Fig. 2g and maximum temperature difference data of the Mg17Al12/MBA0.20T/Mg17Al12 module, other Mg-based modules and commercial bismuth telluride modules at different hot-side temperatures plotted in Fig. 2h.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Z., Sun, Y., Wu, H. et al. Anodic protection enables moisture-stable Mg3(Sb, Bi)2 for thermoelectric cooling. Nat. Mater. (2026). https://doi.org/10.1038/s41563-026-02563-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41563-026-02563-0