Abstract
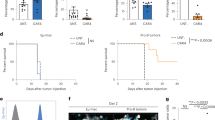
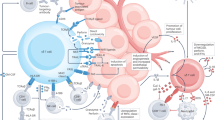
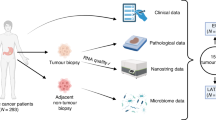
Gamma-delta (γδ) T cell-based cancer immunotherapies represent a promising avenue for cancer treatment. However, their development is challenged by the limited expansion and differentiation of the cells ex vivo. Here we induced the endogenous expansion and activation of γδ T cells through oral administration of garlic-derived nanoparticles (GNPs). We found that GNPs could significantly promote the proliferation and activation of endogenous γδ T cells in the intestine, leading to generation of large amount of interferon-γ (IFNγ). Moreover GNP-treated mice showed increased levels of chemokine CXCR3 in intestinal γδ T cells, which can drive their migration from the gut to the tumour environment. The translocation of γδ T cells and IFNγ from the intestine to extraintestinal subcutaneous tumours remodels the tumour immune microenvironment and synergizes with anti-PD-L1, inducing robust antitumour immunity. Our study delineates mechanistic insight into the complex gut–tumour interactome and provides an alternative approach for γδ T cell-based immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. The authors declare that all other data supporting the findings of this study are available within the paper or Supplementary Information.
Code availability
Single-cell RNA-sequencing data have been deposited at Sequence Read Archive (SRA). SRA records are accessible with the following link: https://www.ncbi.nlm.nih.gov/sra/PRJNA1102105.
References
Duke-Cohan, J. S. et al. Pre-T cell receptor self-MHC sampling restricts thymocyte dedifferentiation. Nature 613, 565–574 (2023).
Silva-Santos, B. & Mensurado, S. γδ T cells maintain sensitivity to immunotherapy in MHC-I-deficient tumors. Nat. Immunol. 24, 387–388 (2023).
de Vries, N. L. et al. γδ T cells are effectors of immunotherapy in cancers with HLA class I defects. Nature 613, 743–750 (2023).
Rancan, C. et al. Exhausted intratumoral Vδ2− γδ T cells in human kidney cancer retain effector function. Nat. Immunol. 24, 612–624 (2023).
Zhu, X. et al. Dectin-1 signaling on colonic γδ T cells promotes psychosocial stress responses. Nat. Immunol. 24, 625–636 (2023).
Wu, Y. et al. An innate-like Vδ1 + γδ T cell compartment in the human breast is associated with remission in triple-negative breast cancer. Sci. Transl. Med. 11, eaax9364 (2019).
Mensurado, S., Blanco-Domínguez, R. & Silva-Santos, B. The emerging roles of γδ T cells in cancer immunotherapy. Nat. Rev. Clin. Oncol. 20, 178–191 (2023).
De Gassart, A. et al. Development of ICT01, a first-in-class, anti-BTN3A antibody for activating Vγ9Vδ2 T cell-mediated antitumor immune response. Sci. Transl. Med. 13, eabj0835 (2021).
Nishimoto, K. P. et al. Allogeneic CD20‐targeted γδ T cells exhibit innate and adaptive antitumor activities in preclinical B‐cell lymphoma models. Clin. Transl. Immunol. 11, e1373 (2022).
Xu, Y. et al. Allogeneic Vγ9Vδ2 T-cell immunotherapy exhibits promising clinical safety and prolongs the survival of patients with late-stage lung or liver cancer. Cell. Mol. Immunol. 18, 427–439 (2021).
Dwivedi, S. F. V. P. Allicin as an adjunct immunotherapy against tuberculosis. J. Cell. Immunol. 2, 78–182 (2020).
Li, Z. et al. Allicin shows antifungal efficacy against Cryptococcus neoformans by blocking the fungal cell membrane. Front. Microbiol. 13, 1012516 (2022).
Catanzaro, E., Canistro, D., Pellicioni, V., Vivarelli, F. & Fimognari, C. Anticancer potential of allicin: a review. Pharmacol. Res. 177, 106118 (2022).
Nantz, M. P. et al. Supplementation with aged garlic extract improves both NK and γδ-T cell function and reduces the severity of cold and flu symptoms: a randomized, double-blind, placebo-controlled nutrition intervention. Clin. Nutr. 31, 337–344 (2012).
Goodridge, H. S. et al. Activation of the innate immune receptor dectin-1 upon formation of a ‘phagocytic synapse’. Nature 472, 471–475 (2011).
Iliev, I. D. et al. Interactions between commensal fungi and the C-type lectin receptor dectin-1 influence colitis. Science 336, 1314–1317 (2012).
Taylor, P. R. et al. Dectin-1 is required for β-glucan recognition and control of fungal infection. Nat. Immunol. 8, 31–38 (2007).
Petermann, F. et al. γδ T cells enhance autoimmunity by restraining regulatory T cell responses via an interleukin-23-dependent mechanism. Immunity 33, 351–363 (2010).
Price, S. J. & Hope, J. C. Enhanced secretion of interferon-γ by bovine γδ T cells induced by coculture with Mycobacterium bovis-infected dendritic cells: evidence for reciprocal activating signals. Immunology 126, 201–208 (2009).
Sedlak, C., Patzl, M., Saalmuller, A. & Gerner, W. IL-12 and IL-18 induce interferon-γ production and de novo CD2 expression in porcine γδ T cells. Dev. Comp. Immunol. 47, 115–122 (2014).
Teo, H. Y. et al. IL-12/18/21 pre-activation enhances the anti-tumor efficacy of expanded γδT cells and overcomes resistance to anti-PD-L1 treatment. Cancer Immunol. Res. 11, 978–999 (2022).
Beziaud, L. et al. IFNγ-induced stem-like state of cancer cells as a driver of metastatic progression following immunotherapy. Cell Stem Cell 30, 818–831.e816 (2023).
Kohlgruber, A. C. et al. γδ T cells producing interleukin-17A regulate adipose regulatory T cell homeostasis and thermogenesis. Nat. Immunol. 19, 464–474 (2018).
Yong, L. et al. Calcium/calmodulin-dependent protein kinase IV promotes imiquimod-induced psoriatic inflammation via macrophages and keratinocytes in mice. Nat. Commun. 13, 4255 (2022).
Nielsen, M. M., Witherden, D. A. & Havran, W. L. γδ T cells in homeostasis and host defence of epithelial barrier tissues. Nat. Rev. Immunol. 17, 733–745 (2017).
Bagheri, H. et al. CXCL-10: a new candidate for melanoma therapy? Cell Oncol. 43, 353–365 (2020).
House, I. G. et al. Macrophage-derived CXCL9 and CXCL10 are required for antitumor immune responses following immune checkpoint blockade. Clin. Cancer Res. 26, 487–504 (2020).
Karin, N. CXCR3 ligands in cancer and autoimmunity, chemoattraction of effector T cells, and beyond. Front. Immunol. 11, 976 (2020).
Ozga, A. J. et al. CXCL10 chemokine regulates heterogeneity of the CD8+ T cell response and viral set point during chronic infection. Immunity 55, 82–97 e88 (2022).
Silva-Santos, B., Serre, K. & Norell, H. γδ T cells in cancer. Nat. Rev. Immunol. 15, 683–691 (2015).
Grasso, C. S. et al. Conserved interferon-γ signaling drives clinical response to immune checkpoint blockade therapy in melanoma. Cancer Cell 38, 500–515 e503 (2020).
Larson, R. C. et al. CAR T cell killing requires the IFNγR pathway in solid but not liquid tumours. Nature 604, 563–570 (2022).
Abiko, K. et al. IFN-γ from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. Br. J. Cancer 112, 1501–1509 (2015).
Papadopoulou, M., Sanchez Sanchez, G. & Vermijlen, D. Innate and adaptive γδ T cells: how, when, and why. Immunol. Rev. 298, 99–116 (2020).
Bordon, Y. γδ T cells mediate immunotherapy responses when cancers lack MHC class I. Nat. Rev. Immunol. 23, 137 (2023).
Lien, S. C. et al. Tumor reactive γδ T cells contribute to a complete response to PD-1 blockade in a Merkel cell carcinoma patient. Nat. Commun. 15, 1094 (2024).
Xu, J. et al. Yeast-derived nanoparticles remodel the immunosuppressive microenvironment in tumor and tumor-draining lymph nodes to suppress tumor growth. Nat. Commun. 13, 110 (2022).
Fan, Q. et al. An implantable blood clot-based immune niche for enhanced cancer vaccination. Sci. Adv. 6, eabb4639 (2020).
Han, X. et al. Red blood cell-derived nanoerythrosome for antigen delivery with enhanced cancer immunotherapy. Sci. Adv. 5, eaaw6870 (2019).
Zhu, H. et al. Human PBMC scRNA-seq-based aging clocks reveal ribosome to inflammation balance as a single-cell aging hallmark and super longevity. Sci. Adv. 9, eabq7599 (2023).
Acknowledgements
This work was supported by the National Key R&D Program of China (grant number 2022YFB3808100) and the National Natural Science Foundation of China (grant numbers 32371476 and T2321005). This work was partly supported by the Collaborative Innovation Center of Suzhou Nano Science and Technology, the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and the 111 Project.
Author information
Authors and Affiliations
Contributions
C.W. and J.X. designed the project. J.X. performed the experiments, collected the data, analysed and interpreted the data, and wrote the first version of the paper. All the authors discussed the results and implications and edited the paper at all stages.
Corresponding authors
Ethics declarations
Competing interests
C.W. and J.X. are inventors on a pending patent related to the technology described here, filed by Soochow University (number CN202310634131.4, filed 12 September 2023). The other authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–45, and Tables 1 and 2.
Source data
Source Data Figs. 1–5 (download XLSX )
Statistical source data for Figs. 1–5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, J., Yu, Y., Zhang, Y. et al. Oral administration of garlic-derived nanoparticles improves cancer immunotherapy by inducing intestinal IFNγ-producing γδ T cells. Nat. Nanotechnol. 19, 1569–1578 (2024). https://doi.org/10.1038/s41565-024-01722-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41565-024-01722-1
This article is cited by
-
Plant-derived exosomes: an emerging delivery platform for cancer therapy
Journal of Nanobiotechnology (2026)
-
Natural compounds as immune checkpoint inhibitors in melanoma: a systematic review
Naunyn-Schmiedeberg's Archives of Pharmacology (2026)
-
Carrier-free nanoparticles—new strategy of improving druggability of natural products
Journal of Nanobiotechnology (2025)
-
Artemisia annua-derived extracellular vesicles reprogram breast tumor immune microenvironment via altering macrophage polarization and synergizing recruitment of T lymphocytes
Chinese Medicine (2025)
-
Internalized polystyrene nanoplastics trigger testicular damage and promote ferroptosis via CISD1 downregulation in mouse spermatocyte
Journal of Nanobiotechnology (2025)