Abstract
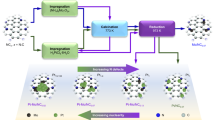
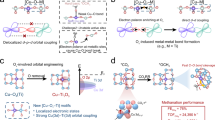
The development of substrates capable of anchoring single-atom catalysts (SACs) while enabling their dynamic reconfiguration into heteronuclear dual-atom catalysts (DACs) holds considerable promise for electrochemical synthesis, yet remains underexplored. Here we show that electrochemical desulfurization of MoS2 generates vacancy-rich 1T′ domains, which support high loadings of Cu (7.9 wt%) and Pt (6.7 wt%) SACs that are well-positioned for dynamic sintering to form DACs. Operando X-ray absorption spectroscopy and density functional theory calculations reveal a voltage-driven, reversible transformation between individual Pt/Cu SACs and Cu–Pt DAC configurations during hydrogen evolution reaction potentials. The electric-field-induced Cu–Pt DACs exhibit superior performance in the selective hydrogenation of alkynes compared with their monometallic SAC counterparts. This work underscores vacancy-enriched 1T′-MoS2 as a versatile platform for high-density SAC deposition, enabling on-demand structural reconfiguration and paving the way for tailored catalyst design in electrosynthesis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available from the authors upon reasonable request. Source data are provided with this paper.
References
Wang, A., Li, J. & Zhang, T. Heterogeneous single-atom catalysis. Nat. Rev. Chem. 2, 65–81 (2018).
Li, X. et al. Functional CeOx nanoglues for robust atomically dispersed catalysts. Nature 611, 284–288 (2022).
Yang, J. et al. Dynamic behavior of single-atom catalysts in electrocatalysis: identification of Cu–N3 as an active site for the oxygen reduction reaction. J. Am. Chem. Soc. 143, 14530–14539 (2021).
Liu, L., Chen, T. & Chen, Z. Understanding the dynamic aggregation in single‐atom catalysis. Adv. Sci. 11, 2308046 (2024).
Hu, J. et al. Sulfur vacancy-rich MoS2 as a catalyst for the hydrogenation of CO2 to methanol. Nat. Catal. 4, 242–250 (2021).
Zheng, J. et al. High loading of transition metal single atoms on chalcogenide catalysts. J. Am. Chem. Soc. 143, 7979–7990 (2021).
Gan, X. et al. 2H/1T phase transition of multilayer MoS2 by electrochemical incorporation of S vacancies. ACS A.E.M. 1, 4754–4765 (2018).
Hou, H. L. et al. High-yield preparation of exfoliated 1T-MoS2 with SERS activity. Chem. Mater. 31, 5725–5734 (2019).
Ophus, C. Four-dimensional scanning transmission electron microscopy (4D-STEM): From scanning nanodiffraction to ptychography and beyond. Microsc. Microanal. 25, 563–582 (2019).
Chen, Z. et al. Electron ptychography achieves atomic-resolution limits set by lattice vibrations. Science 372, 826–831 (2021).
Liu, G. et al. MoS2 monolayer catalyst doped with isolated Co atoms for the hydrodeoxygenation reaction. Nat. Chem. 9, 810–816 (2017).
Li, H. et al. Atomic structure and dynamics of single platinum atom interactions with monolayer MoS2. ACS Nano 11, 3392–3403 (2017).
Qu, W. et al. Single-atom catalysts reveal the dinuclear characteristic of active sites in NO selective reduction with NH3. Nat. Commun. 11, 1532 (2020).
Shi, Z. et al. Phase-dependent growth of Pt on MoS2 for highly efficient H2 evolution. Nature 621, 300–305 (2023).
Xiong, Y. et al. Chemically switchable n-type and p-type conduction in bismuth selenide nanoribbons for thermoelectric energy harvesting. ACS nano 15, 2791–2799 (2021).
Fu, F. et al. Z-contrast and electron energy loss spectroscopy study of passive layer formation at ferroelectric PbTiO3/Pt interfaces. Appl. Phys. Lett. 87, 262904 (2005).
Ahn, C. C. & Krivanek, O. L. EELS Atlas: A Reference Collection of Electron Energy Loss Spectra Covering All Stable Elements (Gatan, Warrendale, PA, 1983).
Chen, Z. X. et al. Addressing the quantitative conversion bottleneck in single-atom catalysis. Nat. Commun. 13, 2807 (2022).
Yan, Q. Q. et al. Reversing the charge transfer between platinum and sulfur-doped carbon support for electrocatalytic hydrogen evolution. Nat. Commun. 10, 4977 (2019).
Huang, F. Low-temperature acetylene semi-hydrogenation over the Pd1–Cu1 dual-atom catalyst. J. Am. Chem. Soc. 144, 18485–18493 (2022).
Kan, M. et al. Structures and phase transition of a MoS2 monolayer. J. Phys. Chem. C 118, 1515–1522 (2014).
Li, W. H. et al. Creating high regioselectivity by electronic metal–support interaction of a single-atomic-site catalyst. J. Am. Chem. Soc. 143, 15453–15461 (2021).
Song, J. et al. Promoting dinuclear‐type catalysis in Cu1–C3N4 single‐atom catalysts. Adv. Mater. 34, 2204638 (2022).
Zhang, X. et al. Platinum–copper single atom alloy catalysts with high performance towards glycerol hydrogenolysis. Nat. Commun. 10, 5812 (2019).
Wang, Z. Y. et al. Pt single-atom electrocatalysts at Cu2O nanowires for boosting electrochemical sensing toward glucose. Chem. Eng. J. 495, 153564 (2024).
Shi, R. et al. Room-temperature electrochemical acetylene reduction to ethylene with high conversion and selectivity. Nat. Catal. 4, 565–574 (2021).
Li, H. et al. σ-Alkynyl adsorption enables electrocatalytic semihydrogenation of terminal alkynes with easy-reducible/passivated groups over amorphous PdSx nanocapsules. J. Am. Chem. Soc. 144, 19456–19465 (2022).
Gu, J. et al. Synergizing metal–support interactions and spatial confinement boosts dynamics of atomic nickel for hydrogenations. Nat. Nanotechnol. 16, 1141–1149 (2021).
Gao, J. et al. Mixed metal-organic framework with multiple binding sites for efficient C2H2/CO2 separation. Angew. Chem. Int. Ed. 11, 4396–4400 (2020).
Wondergem, C. S., Hartman, T. & Weckhuysen, B. M. In situ shell-isolated nanoparticle-enhanced Raman spectroscopy to unravel sequential hydrogenation of phenylacetylene over platinum nanoparticles. ACS Catal. 9, 10794–10802 (2019).
Pei, G. X. et al. Performance of Cu-alloyed Pd single-atom catalyst for semihydrogenation of acetylene under simulated front-end conditions. ACS Catal. 7, 1491–1500 (2017).
Gao, Q. et al. Atomic layers of B2CuPd on Cu nanocubes as catalysts for selective hydrogenation. J. Am. Chem. Soc. 145, 19961–19968 (2023).
Li, B. et al. Introduction of π-complexation into porous aromatic framework for highly selective adsorption of ethylene over ethane. J. Am. Chem. Soc. 136, 8654–8660 (2014).
Tsai, C. et al. Electrochemical generation of sulfur vacancies in the basal plane of MoS2 for hydrogen evolution. Nat. Commun. 8, 15113 (2017).
Acknowledgements
K.P.L. acknowledges funding support from NUS’s Centre for Hydrogen Innovation program (grant no. CHI-P2022-01). Z.C. acknowledges the University Development Fund, Research Start-up Fund (grant no. UDF01002976). M.Y. acknowledges funding support from the Natural Science Foundation of Guangdong (grant no. P0046476). We acknowledge funding support from the Singapore-International Synchrotron Access Programme (SG-ISAP) and National Synchrotron Programme (NSP). Part of this research was performed on the X-ray absorption spectroscopy beamline at the Australian Synchrotron, which is part of ANSTO; we acknowledge their support for operando XAS experiment. We also thank the National Supercomputing Centre of Singapore for providing computational resources, and the Electron Microscopy Facility (EMF) at National University of Singapore, as well as the Facility for Analysis, Characterisation, Testing and Simulation (FACTS) at Nanyang Technological University, for the STEM measurements. We also thank the staff of the BL17B beamline at the National Facility for Protein Science in Shanghai, Shanghai Advanced Research Institute, Chinese Academy of Sciences, for their technical support in XAFS data collection.
Author information
Authors and Affiliations
Contributions
J.W. conceived the research, synthesized the materials, and performed catalytic measurements under the supervision of K.P.L. X-ray absorption spectroscopy measurements and data processing were performed by J.W. and S.X. Molecular dynamics and DFT calculations were performed by K.Y. under the supervision of M.Y. Scanning transmission electron microscopy characterizations (including electron ptychography/4D-STEM) were performed by X.Z. and M.S. In situ Raman measurements were performed by H.L. under the supervision of C.L. and B.Z. Scanning electron microscopy, partial XAS measurements, STEM data analysis and schematic diagrams were performed by Z.W. R.Z. helped to build the model for the XANES simulation. In situ DRIFTS measurement were performed by T.W. under the supervision of N.Y. Q.H. assisted in testing the XAS. The draft was written by J.W. and revised by K.P.L. All authors discussed and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Xin-Hao Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–54 and Tables 1–6.
Supplementary Video 1 (download MP4 )
Molecular dynamics simulation of the protonation process at the negative voltage
Source data
Source Data Fig. 1–5 (download ZIP )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, J., Chen, Z., Yang, K. et al. Electric bias-induced reversible configuration of single and heteronuclear dual-atom catalysts on 1Tʹ-MoS2. Nat. Nanotechnol. 20, 1043–1051 (2025). https://doi.org/10.1038/s41565-025-01934-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41565-025-01934-z
This article is cited by
-
Electrically reconfigurable heteronuclear dual-atom catalysts
Nature Nanotechnology (2025)