Abstract
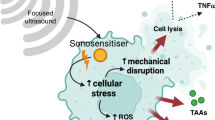
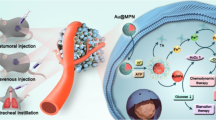
Chemodynamic therapy and sonodynamic therapy are two promising tumour therapeutic strategies. However, lack of highly effective sonosensitizers and control over chemodynamic therapy limit their application. Here we synthesize silver-doped zinc selenide quantum dots with atomically dispersed superficial Fe and show that they act as efficient sonosensitizers, catalysers and immunoreagents. Surface modification with an in situ self-assembly peptide drives accumulation in tumours. Superficial FeIII remains stable and converts to FeII only under ultrasonic processing, reverting to FeIII upon ultrasound cessation. Under ultrasound stimulation, superficial Fe undergoes valence change with concomitant amelioration of the hypoxic tumour microenvironment and production of sonodynamic therapy-beneficial hydroxyl radicals. Furthermore, silver doping suppressed nonradiative recombination of excitons, leading to improved production of singlet oxygen. Meanwhile, selenium promotes robust systemic immune responses for the inhibition of tumour metastases. This nano-platform allows control of valence switching of atomically dispersed catalysts, representing an effective tool for chemodynamic/sonodynamic/immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All relevant data during the study are available from the corresponding authors upon request. Source data are provided with this paper.
References
Chen, R. et al. Photoacoustic molecular imaging-escorted adipose photodynamic–browning synergy for fighting obesity with virus-like complexes. Nat. Nanotechnol. 16, 455–465 (2021).
Wang, D. et al. Sub-nanosheet induced inverse growth of negative valency Au clusters for tumor treatment by enhanced oxidative stress. Angew. Chem. Int. Ed. 63, e202410649 (2024).
Fan, Y., Liu, S., Yi, Y., Rong, H. & Zhang, J. Catalytic nanomaterials toward atomic levels for biomedical applications: from metal clusters to single-atom catalysts. ACS Nano 15, 2005–2037 (2021).
Fan, K. et al. In vivo guiding nitrogen-doped carbon nanozyme for tumor catalytic therapy. Nat. Commun. 9, 1440 (2018).
Li, J., Stephanopoulos, M. F. & Xia, Y. Introduction: heterogeneous single-atom catalysis. Chem. Rev. 120, 11699–11702 (2020).
Ji, S. et al. Matching the kinetics of natural enzymes with a single-atom iron nanozyme. Nat. Catal. 4, 407–417 (2021).
Yin, Y. et al. Boosting Fenton-like reactions via single atom Fe catalysis. Environ. Sci. Technol. 53, 11391–11400 (2019).
Qian, X., Zhang, J., Gu, Z. & Chen, Y. Nanocatalysts-augmented Fenton chemical reaction for nanocatalytic tumor therapy. Biomaterials 211, 1–13 (2019).
Yu, B., Wang, W., Sun, W., Jiang, C. & Lu, L. Defect engineering enables synergistic action of enzyme-mimicking active centers for high-efficiency tumor therapy. J. Am. Chem. Soc. 143, 8855–8865 (2021).
Qin, X. et al. Peroxisome inspired hybrid enzyme nanogels for chemodynamic and photodynamic therapy. Nat. Commun. 12, 5243 (2021).
Huang, P. et al. Metalloporphyrin-encapsulated biodegradable nanosystems for highly efficient magnetic resonance imaging-guided sonodynamic cancer therapy. J. Am. Chem. Soc. 139, 1275–1284 (2017).
Wang, D. et al. Precise magnetic resonance imaging-guided sonodynamic therapy for drug-resistant bacterial deep infection. Biomaterials 264, 120386 (2021).
Wang, D., Zhu, Y., Wan, X., Zhang, X. & Zhang, J. Colloidal semiconductor nanocrystals for biological photodynamic therapy applications: recent progress and perspectives. Prog. Nat. Sci. Mater. Int. 30, 443–455 (2020).
Linterman, M. A. & Denton, A. E. Selenium saves ferroptotic TFH cells to fortify the germinal center. Nat. Immunol. 22, 1074–1076 (2021).
Gao, S. et al. Selenium-containing nanoparticles combine the NK cells mediated immunotherapy with radiotherapy and chemotherapy. Adv. Mater. 32, 1907568 (2020).
Feng, Z., Wang, H., Chen, X. & Xu, B. Self-assembling ability determines the activity of enzyme-instructed self-assembly for inhibiting cancer cells. J. Am. Chem. Soc. 139, 15377–15384 (2017).
Zhang, L. et al. Transformable peptide nanoparticles arrest HER2 signalling and cause cancer cell death in vivo. Nat. Nanotechnol. 15, 145–153 (2020).
Gui, J. et al. Phosphine-initiated cation exchange for precisely tailoring composition and properties of semiconductor nanostructures: old concept, new applications. Angew. Chem. Int. Ed. 127, 3754–3758 (2015).
Di, Q. et al. High-performance quantum dots with synergistic doping and oxide shell protection synthesized by cation exchange conversion of ternary-composition nanoparticles. J. Phys. Chem. Lett. 10, 2606–2615 (2019).
Jing, X. et al. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol. Cancer 18, 157 (2019).
Wu, C. et al. Biodegradable Fe(III)@WS2-PVP nanocapsules for redox reaction and TME-enhanced nanocatalytic, photothermal, and chemotherapy. Adv. Funct. Mater. 29, 1901722 (2019).
Huang, L., Chen, J., Gan, L., Wang, J. & Dong, S. Single-atom nanozymes. Sci. Adv. 5, eaav5490 (2019).
Ma, W. et al. A single-atom Fe–N4 catalytic site mimicking bifunctional antioxidative enzymes for oxidative stress cytoprotection. Chem. Commun. 55, 159–162 (2019).
Bai, J., Jia, X., Zhen, W., Cheng, W. & Jiang, X. A facile ion-doping strategy to regulate tumor microenvironments for enhanced multimodal tumor theranostics. J. Am. Chem. Soc. 140, 106–109 (2018).
Cheng, D.-B. et al. Autocatalytic morphology transformation platform for targeted drug accumulation. J. Am. Chem. Soc. 141, 4406–4411 (2019).
Li, G. et al. Fluorescence and optical activity of chiral CdTe quantum dots in their interaction with amino acids. ACS Nano 14, 4196–4205 (2020).
Branca, C. et al. Role of the OH and NH vibrational groups in polysaccharide-nanocomposite interactions: A FTIR-ATR study on chitosan and chitosan/clay films. Polymer 99, 614–622 (2016).
Pillai, C., Paul, W. & Sharma, C. P. Chitin and chitosan polymers: chemistry, solubility and fiber formation. Prog. Polym. Sci. 34, 641–678 (2009).
Liu, F.-H. et al. Near-infrared laser-driven in situ self-assembly as a general strategy for deep tumor therapy. Nano Lett. 18, 6577–6584 (2018).
Song, X., Feng, L., Liang, C., Yang, K. & Liu, Z. Ultrasound triggered tumor oxygenation with oxygen-shuttle nanoperfluorocarbon to overcome hypoxia-associated resistance in cancer therapies. Nano Lett. 16, 6145–6153 (2016).
Meng, Z. et al. Light-triggered in situ gelation to enable robust photodynamic-immunotherapy by repeated stimulations. Adv. Mater. 31, 1900927 (2019).
Shao, Y. et al. Engineering of upconverted metal–organic frameworks for near-infrared light-triggered combinational photodynamic/chemo-/immunotherapy against hypoxic tumors. J. Am. Chem. Soc. 142, 3939–3946 (2020).
Acknowledgements
This research was supported by the National Natural Science Foundation of China (52272186, 51631001, 51902023, 51702016, 21674027, 21704020, 31870998, 51573032 and 22305139), the National Science Fund for Distinguished Young Scholars (51725302) and the Science Fund for Creative Research Groups of the National Natural Science Foundation of China (11621505).
Author information
Authors and Affiliations
Contributions
D.W., S.B., Z.-Y.Q. and J.Z. conceived and designed the experiments. D.W., L.J., You Li and Z.-Y.Q. performed the experiments. D.W., M.X., H.W., S.B., Z.-Y.Q., J.Z. and Yadong Li analysed the results and discussed the paper. D.W., S.B., Z.-Y.Q. and J.Z. wrote the paper. Z.-Y.Q. and J.Z. supervised the entire project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Kelong Ai, Peng Hou and Huixiong Xu for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Design of the FAQD-1 and the schematic illustration of therapy.
(a) Schematic process of controllable synthesis of FAQD-1. (b) Schematic illustration of multi-enhanced chemodynamic/sonodynamic/immuno therapy of FAQD-1 under US.
Extended Data Fig. 2 ROS detection, cytotoxicity test and retention study in vitro.
(a) Confocal Laser Scanning Microscope images (scale bar = 50 μm) and (b) flow cytometric quantification of Hela cells stained with DCFH-DA after treated by different nanomaterials with 25 μg/mL (n = 3 experimental repeats, mean ± SD, two-way ANOVA; the P value is noted). (c) JC-1 staining (scale bar = 100 μm) and (d) Annexin V-FITC and PI staining (scale bar = 20 μm) of HeLa cells with different treatments (the concentration of the nanomaterials is 25 μg/mL). (e) Cytotoxicity test of Hela cells from under US (1 W/cm2) for 3 min with different formulations (n = 5 experimental repeats, mean ± SD). (f) TEM of MCS treated with FAQD-1 in the presence of MMP, Red arrows indicate the nanoaggregates (scale bar = 500 nm). (g) The retention of Zn element in MCS after co-incubation with FAQD-1 or FAQD-2 in the presence of MMP.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–89 and Tables 1–5.
Supplementary Source Data 1 (download XLSX )
Source data for Supplementary Figs. 3–9, 11–28, 31, 33, 37–41, 44–48, 50, 51, 53, 54, 57–65, 67, 69–74, 77, 79, 81, 83 and 85–89.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, D., Ji, L., Li, Y. et al. Iron–silver-modified quantum dots act as efficient catalysts in anti-cancer multitherapy through controlled, ultrasound-induced oxidation. Nat. Nanotechnol. 20, 1098–1107 (2025). https://doi.org/10.1038/s41565-025-01943-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41565-025-01943-y
This article is cited by
-
Innovative Strategies to Overcome Stability Challenges of Single-Atom Nanozymes
Nano-Micro Letters (2026)
-
Dynamic iron catalysis on quantum dots enables ultrasound-controlled multimodal cancer therapy
Science China Materials (2026)
-
Turning cold tumors into hot tumors to ignite immunotherapy
Molecular Cancer (2025)