Abstract
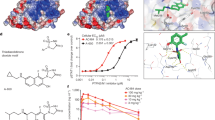
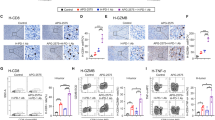
Regulating intracellular phosphorus may affect multiple biosynthetic processes and modulate cancer cell progression. Here we show that exogenous PEGylated black phosphorus nanosheets (BPP) are metabolized into phosphate in tumor cells, where they boost mitochondrial oxidative phosphorylation. This results in the modulation of several signalling pathways, with the attenuation of prosurvival gene expression and reduction in PD-L1 protein expression in melanoma cells, leading to impaired cancer progression. We also reveal that BPP promote the activation of immune regulation, confirmed by the increased proinflammatory cytokine content in serum, high expression of tumour-infiltrating lymphocyte CD8+ T cells and lower expression of CD4+ regulatory T cells in tumour and lymph nodes. In the spleen, BPP mediate a significant increase in the concentration of effector memory CD8+ T cells, inducing a ‘positive regulation’ of the immune microenvironment. The introduction of a PD-1/PD-L1 inhibitor further enhances the immunopotentiation effect. These findings may define BPP as a dual-function tumour chemotherapeutic and immunopotentiator.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
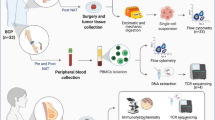
The data that support the findings of this study are available within the Article and its Supplementary Information. The raw data of the transcriptome sequencing have been deposited in The National Center for Biotechnology Information Sequence Read Archive (http://www.ncbi.nlm.nih.gov/sra/) with the accession number PRJNA1287216 for the tumour and PRJNA1286125 for the spleen. The mass spectrometry proteomics data have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD066855. Source data are provided with this paper.
References
Lopaschuk, G. D., Karwi, Q. G., Tian, R., Wende, A. R. & Abel, E. D. Cardiac energy metabolism in heart failure. Circ. Res. 128, 1487–1513 (2021).
Heiden, M. G. V., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Bergers, G. & Fendt, S. M. The metabolism of cancer cells during metastasis. Nat. Rev. Cancer 21, 162–180 (2021).
Kanarek, N., Petrova, B. & Sabatini, D. M. Dietary modifications for enhanced cancer therapy. Nature 579, 507–517 (2020).
Mossmann, D., Park, S. & Hall, M. N. mTOR signalling and cellular metabolism are mutual determinants in cancer. Nat. Rev. Cancer 18, 744–757 (2018).
Liu, G. Y. & Sabatini, D. M. mTOR at the nexus of nutrition, growth, ageing and disease. Nat. Rev. Mol. Cell Biol. 21, 183–203 (2020).
Li, X. Y. et al. Navigating metabolic pathways to enhance antitumour immunity and immunotherapy. Nat. Rev. Clin. Oncol. 16, 425–441 (2019).
Colegio, O. R. et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513, 559–563 (2014).
Zappasodi, R. et al. CTLA-4 blockade drives loss of Treg stability in glycolysis-low tumours. Nature 591, 652–658 (2021).
Calvo, M. S. & Lamberg-Allardt, C. J. Phosphorus. Adv. Nutr. 6, 860–862 (2015).
Boyer, P. D., Falcone, A. B. & Harrison, W. H. Reversal and mechanism of oxidative phosphorylation. Nature 174, 401–402 (1954).
Ubersax, J. A. & Ferrell, J. E. Mechanisms of specificity in protein phosphorylation. Nat. Rev. Mol. Cell Biol. 8, 530–541 (2007).
Gonzalez, P. S. et al. Mannose impairs tumour growth and enhances chemotherapy. Nature 563, 719–723 (2018).
Song, L. T. et al. Proto-oncogene Src links lipogenesis via lipin-1 to breast cancer malignancy. Nat. Commun. 11, 5842 (2020).
Gui, R. J., Jin, H., Wang, Z. H. & Li, J. H. Black phosphorus quantum dots: synthesis, properties, functionalized modification and applications. Chem. Soc. Rev. 47, 6795–6823 (2018).
Li, L. K. et al. Black phosphorus field-effect transistors. Nat. Nanotechnol. 9, 372–377 (2014).
Zhou, W. H. et al. Black phosphorus: bioactive nanomaterials with inherent and selective chemotherapeutic effects. Angew. Chem. Int. Ed. 58, 769–774 (2019).
Zhang, X. G. et al. A targeting black phosphorus nanoparticle based immune cells nano-regulator for photodynamic/photothermal and photo-immunotherapy. Bioact. Mater. 6, 472–489 (2021).
Chen, W. S. et al. Black phosphorus nanosheet-based drug delivery system for synergistic photodynamic/photothermal/chemotherapy of cancer. Adv. Mater. 29, 1603864 (2017).
Liu, J. T. et al. Dual-triggered oxygen self-supply black phosphorus nanosystem for enhanced photodynamic therapy. Biomaterials 172, 83–91 (2018).
Shao, X. M. et al. Intrinsic bioactivity of black phosphorus nanomaterials on mitotic centrosome destabilization through suppression of PLK1 kinase. Nat. Nanotechnol. 16, 1150–1160 (2021).
Cheung, E. C. & Vousden, K. H. The role of ROS in tumour development and progression. Nat. Rev. Cancer 22, 280–297 (2022).
Bian, S. Q. et al. The self-crosslinking smart hyaluronic acid hydrogels as injectable three-dimensional scaffolds for cells culture. Colloids Surf. B Biointerfaces 1, 392–402 (2016).
Hou, J. et al. Treating acute kidney injury with antioxidative black phosphorus nanosheets. Nano Lett. 20, 1447–1454 (2020).
Huang, H. et al. Black phosphorus: a two-dimensional reductant for in situ nanofabrication. npj 2D Mater. Appl. 1, 20 (2017).
Jin, H. J. et al. EGFR activation limits the response of liver cancer to lenvatinib. Nature 595, 730–734 (2021).
Downward, J. Targeting RAS signalling pathways in cancer therapy. Nat. Rev. Cancer 3, 11–22 (2003).
Hoxhaj, G. & Manning, B. D. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism. Nat. Rev. Cancer 20, 74–88 (2020).
Gremke, N. et al. mTOR-mediated cancer drug resistance suppresses autophagy and generates a druggable metabolic vulnerability. Nat. Commun. 11, 4684 (2020).
Wu, Y. Q. et al. ARIH1 signaling promotes anti-tumor immunity by targeting PD-L1 for proteasomal degradation. Nat. Commun. 12, 2346 (2021).
Ding, X. C. et al. The relationship between expression of PD-L1 and HIF-1α in glioma cells under hypoxia. J. Hematol. Oncol. 14, 92 (2021).
Reda, M. et al. Development of a nanoparticle-based immunotherapy targeting PD-L1 and PLK1 for lung cancer treatment. Nat. Commun. 13, 4261 (2022).
Bailey, M. H. et al. Comprehensive characterization of cancer driver genes and mutations. Cell 173, 371–385 (2018).
Chen, G. et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 560, 382–386 (2018).
Dror, S. et al. Melanoma miRNA trafficking controls tumour primary niche formation. Nat. Cell Biol. 18, 1006–1017 (2016).
Poggio, M. et al. PD-L1 suppression of exosomal PD-L1 induces systemic anti-tumor immunity and memory. Cell 177, 414–427 (2019).
Shen, Y. Y. et al. Ursodeoxycholic acid reduces antitumor immunosuppression by inducing CHIP-mediated TGF-β degradation. Nat. Commun. 13, 3419 (2022).
Golan, T. et al. Interactions of melanoma cells with distal keratinocytes trigger metastasis via notch signaling inhibition of MITF. Mol. Cell 59, 664–676 (2015).
Lewis, S. M., Williams, A. & Eisenbarth, S. C. Structure and function of the immune system in the spleen. Sci. Immunol. 4, eaau6085 (2019).
Belz, G. T., Bedoui, S., Kupresanin, F., Carbone, F. R. & Heath, W. R. Minimal activation of memory CD8+ T cell by tissue-derived dendritic cells favors the stimulation of naive CD8+ T cells. Nat. Immunol. 8, 1060–1066 (2007).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Jung, Y. W., Kim, H. G., Perry, C. J. & Kaech, S. M. CCR7 expression alters memory CD8 T-cell homeostasis by regulating occupancy in IL-7-and IL-15-dependent niches. Proc. Natl Acad. Sci. USA 113, 8278–8283 (2016).
Gilchrist, J. J. et al. Natural killer cells demonstrate distinct eQTL and transcriptome-wide disease associations, highlighting their role in autoimmunity. Nat. Commun. 13, 4073 (2022).
Wculek, S. K. et al. Dendritic cells in cancer immunology and immunotherapy. Nat. Rev. Immunol. 20, 7–24 (2020).
Olmeda, D. et al. Whole-body imaging of lymphovascular niches identifies pre-metastatic roles of midkine. Nature 546, 676–680 (2017).
Owens, B. Melanoma. Nature 515, S109 (2014).
Robert, C. et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 20, 1239–1251 (2019).
Zhang, T. et al. Degradation chemistry and stabilization of exfoliated few-layer black phosphorus in water. J. Am. Chem. Soc. 140, 7561–7567 (2018).
Liao, Z., Chua, D. & Tan, N. S. Reactive oxygen species: a volatile driver of field cancerization and metastasis. Mol. Cancer 18, 65 (2019).
Boese, A. C. & Kang, S. Mitochondrial metabolism-mediated redox regulation in cancer progression. Redox Biol. 42, 101870 (2021).
Zhou, C. F. et al. Nynrin preserves hematopoietic stem cell function by inhibiting the mitochondrial permeability transition pore opening. Cell Stem Cell 31, 1359–1375 (2024).
Hou, D. et al. Cationic antimicrobial peptide NRC-03 induces oral squamous cell carcinoma cell apoptosis via CypD-mPTP axis-mediated mitochondrial oxidative stress. Redox Biol. 54, 102355 (2022).
Carne Trecesson, S. et al. BCL-XL directly modulates RAS signalling to favour cancer cell stemness. Nat. Commun. 8, 1123 (2017).
KEGG pathway: map05235. https://www.genome.jp/entry/map05235 (2019).
Liu, Y. et al. Tumors exploit FTO-mediated regulation of glycolytic metabolism to evade immune surveillance. Cell Metab. 33, 1221–1233 (2021).
Reinfeld, B. I., Rathmell, W. K., Kim, T. K. & Rathmell, J. C. The therapeutic implications of immunosuppressive tumor aerobic glycolysis. Cell Mol. Immunol. 19, 46–58 (2022).
Watson, M. J. et al. Metabolic support of tumour-infiltrating regulatory T cells by lactic acid. Nature 591, 645–651 (2021).
Sanmamed, M. F. & Chen, L. A paradigm shift in cancer immunotherapy: from enhancement to normalization. Cell 175, 313–326 (2018).
Propper, D. J. & Balkwill, F. R. Harnessing cytokines and chemokines for cancer therapy. Nat. Rev. Clin. Oncol. 19, 237–253 (2022).
Patsoukis, N., Wang, Q., Strauss, L. & Boussiotis, V. A. Revisiting the PD-1 pathway. Sci. Adv. 6, 114057 (2020).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant numbers 32422044, 32271419 (Y.S.) and 32471416, 51973136 (Y.F.)) and Varnotech-Sichuan University Joint Laboratory on ‘Development and Application of Novel Immunologic Adjuvant’ (grant number 24H0599, Y.S.). We thank Varnotech Biopharm Ltd and its directors H. Wang and G. Liu for their suggestions in the preclinical evaluation of BPP.
Author information
Authors and Affiliations
Contributions
Y.Y. and M.Z. contributed to the execution and analysis of all the experiments. J. Li contributed to the execution and analysis of supplementary experiments. R.X. contributed to the synthesis and preparation of hydrogels and animal studies. J. Liang and Q.J. contributed to the in vitro cell proliferation and tissue processing. X.P., A.T., L.M. and Y.L. contributed to the data analysis and article modification. X.Z. provided research guidance. Y.F. and Y.S. conceived the project and contributed to the study design and result analysis. Y.Y., M.Z., Y.F. and Y.S. prepared the manuscript, with inputs from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Menghuan Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–23, Tables 1 and 2 and materials and methods.
Supplementary Data 1 (download PDF )
In vitro cytotoxicity test—direct-contact test.
Supplementary Data 2 (download PDF )
Bacterial reverse mutation assay.
Supplementary Data 3 (download PDF )
In vitro mammalian cell TK gene mutation test.
Supplementary Data 4 (download PDF )
Test for systemic toxicity: acute systemic toxicity test (direct application—subcutaneous route).
Supplementary Data 5 (download PDF )
Recognition certificate of Sichuan Testing Center for Biomaterials and Medical Devices.
Supplementary Data 6 (download XLSX )
Source data for Supplementary Figures.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2.
Source Data Fig. 2 (download PDF )
Unprocessed western blots for Fig. 2i.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4.
Source Data Fig. 6 (download XLSX )
Statistical source data for Fig. 6.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Y., Zhao, M., Li, J. et al. Black phosphorus nanosheets boost mitochondrial oxidative phosphorylation improving immunotherapy outcomes. Nat. Nanotechnol. 20, 1843–1855 (2025). https://doi.org/10.1038/s41565-025-02022-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41565-025-02022-y