Abstract
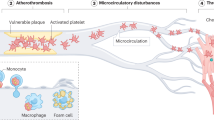
Antiplatelet drugs have represented a milestone in treating patients at high risk of thrombosis. However, their clinical use remains limited by bleeding-associated risk and limited efficacy. Excessive reactive oxygen species (ROS) produced by damaged vascular endothelial cells have been shown to stimulate thrombosis. Here we propose that a ROS-chemotactic nanoscavenger (MDCP), formed by crosslinking melanin and catalase, prevents acute thrombosis by protecting vascular endothelial cells from oxidative stress. We demonstrate that treatment with MDCP inhibits ROS-induced apoptosis of endothelial cells, thereby maintaining endothelial integrity and preventing collagen exposure, which consequently prevents platelet activation and thrombosis. By avoiding direct interference with platelet function, this modulation of vascular redox homeostasis via MDCP provides a promising alternative antithrombotic strategy that addresses the bleeding risk of current clinical antithrombotic drugs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The transcriptomic sequencing data from occlusive coronary thrombi of patients with ST-elevation myocardial infarction and the peripheral blood of healthy donors were retrieved from the GEO under accession number GSE19339. Transcriptome data comparing rat abdominal aortas treated with ferric chloride versus saline control are available in the GEO database under accession number GSE307563. All other data supporting the findings are available within the article and its Supplementary Information, or from the corresponding authors upon request. Source data are provided with this paper.
References
Christensen, H. Long-term disability after transient ischaemic attack or minor stroke. Lancet Neurol. 21, 859–860 (2022).
Koupenova, M., Kehrel, B. E., Corkrey, H. A. & Freedman, J. E. Thrombosis and platelets: an update. Eur. Heart J. 38, 785–791 (2017).
Capodanno, D. et al. Dual-pathway inhibition for secondary and tertiary antithrombotic prevention in cardiovascular disease. Nat. Rev. Cardiol. 17, 242–257 (2020).
McFadyen, J. D., Schaff, M. & Peter, K. Current and future antiplatelet therapies: emphasis on preserving haemostasis. Nat. Rev. Cardiol. 15, 181–191 (2018).
Gorog, D. A. et al. De-escalation or abbreviation of dual antiplatelet therapy in acute coronary syndromes and percutaneous coronary intervention: a Consensus Statement from an international expert panel on coronary thrombosis. Nat. Rev. Cardiol. 20, 830–844 (2023).
Yousuf, O. & Bhatt, D. L. The evolution of antiplatelet therapy in cardiovascular disease. Nat. Rev. Cardiol. 8, 547–559 (2011).
Song, Y. et al. Platelet-targeted thromboprophylaxis with a human serum albumin fusion drug: preventing thrombosis and reducing cardiac ischemia/reperfusion injury without bleeding complications. Theranostics 14, 3267 (2024).
Refaat, A. et al. Activated platelet-targeted IR780 immunoliposomes for photothermal thrombolysis. Adv. Funct. Mater. 33, 2209019 (2023).
Zhang, Y., Murugesan, P., Huang, K. & Cai, H. NADPH oxidases and oxidase crosstalk in cardiovascular diseases: novel therapeutic targets. Nat. Rev. Cardiol. 17, 170–194 (2020).
Liang, Y. et al. Interplay of hypoxia-inducible factors and oxygen therapy in cardiovascular medicine. Nat. Rev. Cardiol. 20, 723–737 (2023).
Violi, F., Pastori, D., Pignatelli, P. & Carnevale, R. Nutrition, thrombosis, and cardiovascular disease. Circ. Res. 126, 1415–1442 (2020).
Lee, J. et al. Thrombus targeting aspirin particles for near infrared imaging and on-demand therapy of thrombotic vascular diseases. J. Control. Release 304, 164–172 (2019).
Ye, H. et al. Thrombus inhibition and neuroprotection for ischemic stroke treatment through platelet regulation and ROS scavenging. ChemMedChem 17, e202200317 (2022).
Wang, Y. et al. A thrombin-triggered self-regulating anticoagulant strategy combined with anti-inflammatory capacity for blood-contacting implants. Sci. Adv. 8, eabm3378 (2022).
Xu, L. et al. Magnetic response combined with bioactive ion therapy: a RONS-scavenging theranostic nanoplatform for thrombolysis and renal ischemia–reperfusion injury. ACS Nano 17, 5695–5712 (2023).
Xi, H. et al. Caspase-1 inflammasome activation mediates homocysteine-induced pyrop-apoptosis in endothelial cells. Circ. Res. 118, 1525–1539 (2016).
Durand, E. et al. In vivo induction of endothelial apoptosis leads to vessel thrombosis and endothelial denudation: a clue to the understanding of the mechanisms of thrombotic plaque erosion. Circulation 109, 2503–2506 (2004).
Furie, B. & Furie, B. C. Mechanisms of thrombus formation. N. Engl. J. Med. 359, 938–949 (2008).
Lippi, G., Franchini, M. & Targher, G. Arterial thrombus formation in cardiovascular disease. Nat. Rev. Cardiol. 8, 502–512 (2011).
Somasundar, A. et al. Positive and negative chemotaxis of enzyme-coated liposome motors. Nat. Nanotechnol. 14, 1129–1134 (2019).
Amioka, N. et al. Pemafibrate protects the rupture of experimental aortic aneurysm in mice through anti-oxidative stress with induced catalase. Eur. Heart J. 41, ehaa946-3789 (2020).
Ciciliano, J. C. et al. Resolving the multifaceted mechanisms of the ferric chloride thrombosis model using an interdisciplinary microfluidic approach. Blood 126, 817–824 (2015).
Nolfi-Donegan, D., Braganza, A. & Shiva, S. Mitochondrial electron transport chain: oxidative phosphorylation, oxidant production, and methods of measurement. Redox Biol. 37, 101674 (2020).
Tyagi, T. et al. A guide to molecular and functional investigations of platelets to bridge basic and clinical sciences. Nat. Cardiovasc. Res. 1, 223–237 (2022).
Ding, Y. et al. E-160 Blood clot identification and composition assessment by fast spin-echo (FSE) T2WI and T2* mapping. J. Neurointerv. Surg. 12, A115–A116 (2020).
Cofiell, R. et al. Eculizumab reduces complement activation, inflammation, endothelial damage, thrombosis, and renal injury markers in aHUS. Blood 125, 3253–3262 (2015).
Amabile, N. et al. Association of circulating endothelial microparticles with cardiometabolic risk factors in the Framingham Heart Study. Eur. Heart J. 35, 2972–2979 (2014).
Abbas, M. et al. Endothelial microparticles from acute coronary syndrome patients induce premature coronary artery endothelial cell aging and thrombogenicity: role of the Ang II/AT1 receptor/NADPH oxidase-mediated activation of MAPKs and PI3-kinase pathways. Circulation 135, 280–296 (2017).
Kawecki, C., Lenting, P. & Denis, C. von Willebrand factor and inflammation. J. Thromb. Haemost. 15, 1285–1294 (2017).
Grover, S. P. & Mackman, N. Tissue factor: an essential mediator of hemostasis and trigger of thrombosis. Arterioscler. Thromb. Vasc. Biol. 38, 709–725 (2018).
Bhatt, D. L. Aspirin—still the GLOBAL LEADER in antiplatelet therapy. Lancet 392, 896–897 (2018).
Tang, Y. D. et al. Randomized comparisons of double-dose clopidogrel or adjunctive cilostazol versus standard dual antiplatelet in patients with high posttreatment platelet reactivity: results of the CREATIVE trial. Circulation 137, 2231–2245 (2018).
Sweeny, J. M., Gorog, D. A. & Fuster, V. Antiplatelet drug ‘resistance’. Part 1: mechanisms and clinical measurements. Nat. Rev. Cardiol. 6, 273–282 (2009).
Kim, Y. W. & Byzova, T. V. Oxidative stress in angiogenesis and vascular disease. Blood 123, 625–631 (2014).
Binder, C. J., Papac-Milicevic, N. & Witztum, J. L. Innate sensing of oxidation-specific epitopes in health and disease. Nat. Rev. Immunol. 16, 485–497 (2016).
Mackman, N., Bergmeier, W., Stouffer, G. A. & Weitz, J. I. Therapeutic strategies for thrombosis: new targets and approaches. Nat. Rev. Drug Discov. 19, 333–352 (2020).
Dayal, S. et al. Hydrogen peroxide promotes aging-related platelet hyperactivation and thrombosis. Circulation 127, 1308–1316 (2013).
Acknowledgements
This work was supported by the National Natural Science Foundation of China 82130060 (G.-J.T.), 61821002 (G.-J.T.), 32030060 (X.-J.L.) and 82302364 (Y.Z.), the National Key Research and Development Programme of China 2018YFA0704100 (G.-J.T.) and 2021YFA1201000 (X.-J.L.), and the National Natural Science Foundation of China International Collaboration Key Project 51861135103 (X.-J.L.). We also acknowledge support from the Chinese Academy of Sciences (CAS-NSTDA) International Partnership Programme 121D11KYSB20210003 (X.-J.L.), the China Postdoctoral Science Foundation 2023M730589 (Y.Z.), the Chongqing Municipal High-Level Medical Talents Programme for Young and Middle-Aged Professionals YXGD202401-58 (Y.Z.), the Programme for Youth Innovation in Future Medicine at Chongqing Medical University W0170 (Y.Z.), the Kuanren Talents Programme kryc-gg-2213 (Y.Z.), and the Kuanren Talents Enhancement Programme and the Doctoral Supervisor Cultivation Programme of the Second Affiliated Hospital of Chongqing Medical University (Y.Z.). We thank F. Jia for his assistance with the fabrication of the microfluidic devices and Shanghai OE Biotech Co., Ltd., for quantitative metabolomics analysis. The funding sources had no role in the writing of the report or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: G.-J.T., Y.Z., Q.N. and X.-J.L. Methodology: Y.Z., Q.N., L.H., G.Q., F.Z., Y.L., H.J. and Z.T. Investigation: Y.Z., Q.N., L.H., G.Q., Z.W. and Y.L. Visualization: Y.Z., H.W., Y.L., H.J. and Z.T. Funding acquisition: G.-J.T., X.-J.L. and Y.Z. Supervision: G.-J.T., H.Z. and X.-J.L. Original draft writing: Q.N., Y.Z. and G.Q. Review and editing: Q.N., G.Q., L.J., G.-J.T., N.G. and X.-J.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Anirban Sen Gupta and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Results of omics analyses and the efficiency of catalase in protecting endothelial cells.
a, Volcano plot illustrating the differential gene expression in occlusive coronary thrombi from patients with ST-elevation myocardial infarction compared with peripheral blood from healthy donors, n = 4. b,c, Volcano plot illustrates differential protein expression in abdominal aorta of rats modelled with saline compared with that of rats modelled with FeCl3 (b), n = 3. These differentially expressed proteins were subsequently enriched in pathways using KEGG database (c). d,e, Heat map illustrates the differential metabolite profiles in the abdominal aorta of rats modelled with saline compared with that of rats modelled with FeCl3 (d), n = 3. These differential metabolites were enriched in the top 20 pathways in KEGG database (e). The colour bar in d represents Z-scores of normalized expression values. f,g, HUVECs were treated with PBS or CAT for 1 hour followed by treatment with XOD for 12 h to assess the efficiency of catalase in preventing XOD-induced apoptosis of endothelial cells. f, Representative CLSM images of endothelial cells stained with Calcein AM/PI, with live cells appearing green (Calcein AM), and dead cells appearing red (propidium iodide, PI). Scale bar, 200 μm. Experiments were replicated three times. g, Representative flow cytometry plots and quantification of the proportion of viable endothelial cells among total cells. Data are presented as mean ± s.d., n = 3 biologically independent samples. Statistical analysis was performed using a two-tailed unpaired Student’s t-test (a,b, and g). Pathway enrichment analysis was performed using the hypergeometric test. Significantly enriched pathways were identified based on a false discovery rate (FDR) < 0.05 (c,e). ns, not significant, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–35 and Supplementary Methods.
Supplementary Data 1 (download XLSX )
Statistical source data for Supplementary Figs. 1–11, 13, 14, 16, 17, 19–21, 23, 26, 31, 33 and 34.
Source data
Source Data Figs. 1–5, Extended Data Fig. 1 (download ZIP )
Statistical source data + flow cytometry data of Fig. 5a,e,f and Extended Data Fig. 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhong, Y., Ni, Q., Huang, L. et al. Prevention of acute thrombosis with vascular endothelium antioxidative nanoscavenger. Nat. Nanotechnol. 20, 1871–1883 (2025). https://doi.org/10.1038/s41565-025-02046-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41565-025-02046-4