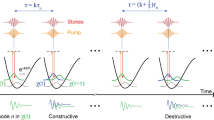
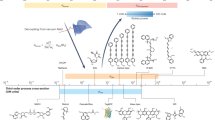
Abstract
Raman spectroscopy is a valuable tool widely used in biology and medicine. While still serving as the mainstream approach for high-fidelity Raman spectral analysis and imaging, spontaneous Raman scattering suffers from low acquisition speed and fluorescence interference. Here we report super-broadband stimulated Raman scattering (SuperB-SRS), a technique able to provide identical spectra to state-of-the-art spontaneous Raman over thousands of wavenumber ranges while being orders of magnitude faster and naturally supporting simultaneous multiplexed fluorescence detection. SuperB-SRS encodes the ideal Raman free induction decays into the stimulated Raman loss through few-cycle laser-induced quantum interference, permitting universal Raman analysis and imaging that unites high spectral fidelity, natural-linewidth-limited spectral resolution, broad bandwidth and state-of-the-art sensitivity. We demonstrate more than a 100-fold increase in speed for imaging of biological tissue samples compared with spontaneous Raman. As a medical application, we detect in clinical serum samples 11 biomarkers widely used to assess liver and kidney functions, cardiovascular diseases and other conditions. SuperB-SRS thus enables generating high-quality, large-scale datasets for data-driven health science, promising wide applications in multiple fields of science.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main paper or supplementary materials or have been deposited via figshare at https://doi.org/10.6084/m9.figshare.30379255 (ref. 51).
References
Raman, C. V. & Krishnan, K. S. A new type of secondary radiation. Nature 121, 501–502 (1928).
Movasaghi, Z., Rehman, S. & Rehman, I. U. Raman spectroscopy of biological tissues. Appl. Spectrosc. Rev. 42, 493–541 (2007).
Rygula, A. et al. Raman spectroscopy of proteins: a review. J. Raman Spectrosc. 44, 1061–1076 (2013).
Czamara, K. et al. Raman spectroscopy of lipids: a review. J. Raman Spectrosc. 46, 4–20 (2015).
Lima, C., Muhamadali, H. & Goodacre, R. The role of Raman spectroscopy within quantitative metabolomics. Annu. Rev. Anal. Chem. 14, 323–345 (2021).
Cheng, J.-X. & Xie, X. S. Vibrational spectroscopic imaging of living systems: an emerging platform for biology and medicine. Science 350, eaaa8870 (2015).
Nie, S. & Emory, S. R. Probing single molecules and single nanoparticles by surface-enhanced Raman scattering. Science 275, 1102–1106 (1997).
Cheng, J.-X., Jia, Y. K., Zheng, G. & Xie, X. S. Laser-scanning coherent anti-Stokes Raman scattering microscopy and applications to cell biology. Biophys. J. 83, 502–509 (2002).
Freudiger, C. W. et al. Label-free biomedical imaging with high sensitivity by stimulated Raman scattering microscopy. Science 322, 1857–1861 (2008).
Min, W., Freudiger, C. W., Lu, S. & Xie, X. S. Coherent nonlinear optical imaging: beyond fluorescence microscopy. Annu. Rev. Phys. Chem. 62, 507–530 (2011).
Evans, C. L. et al. Chemical imaging of tissue in vivo with video-rate coherent anti-Stokes Raman scattering microscopy. Proc. Natl Acad. Sci. USA 102, 16807–16812 (2005).
Saar, B. G. et al. Video-rate molecular imaging in vivo with stimulated Raman scattering. Science 330, 1368–1370 (2010).
Dodo, K., Fujita, K. & Sodeoka, M. Raman spectroscopy for chemical biology research. J. Am. Chem. Soc. 144, 19651–19667 (2022).
Gao, X., Li, X. & Min, W. Absolute stimulated Raman cross sections of molecules. J. Phys. Chem. Lett. 14, 5701–5708 (2023).
De Silvestri, S. et al. Femtosecond time-resolved measurements of optic phonon dephasing by impulsive stimulated Raman scattering in α-perylene crystal from 20 to 300 K. Chem. Phys. Lett. 116, 146–152 (1985).
Selm, R. et al. Ultrabroadband background-free coherent anti-Stokes Raman scattering microscopy based on a compact Er:fiber laser system. Opt. Lett. 35, 3282–3284 (2010).
Ideguchi, T. et al. Coherent Raman spectro-imaging with laser frequency combs. Nature 502, 355–358 (2013).
Camp, C. H. Jr et al. High-speed coherent Raman fingerprint imaging of biological tissues. Nat. Photon. 8, 627–634 (2014).
Yu, Q. et al. Transient stimulated Raman excited fluorescence spectroscopy. J. Am. Chem. Soc. 145, 7758–7762 (2023).
Yu, Q. et al. Transient stimulated Raman scattering spectroscopy and imaging. Light Sci. Appl. 13, 70 (2024).
Kop, R. H., De Vries, P., Sprik, R. & Lagendijk, A. Kramers-Kronig relations for an interferometer. Opt. Commun. 138, 118–126 (1997).
Lenz, G., Eggleton, B., Giles, C., Madsen, C. & Slusher, R. Dispersive properties of optical filters for WDM systems. IEEE J. Quantum Electron. 34, 1390–1402 (1998).
Chia, S.-H. et al. Two-octave-spanning dispersion-controlled precision optics for sub-optical-cycle waveform synthesizers. Optica 1, 315–322 (2014).
Prince, R. C., Frontiera, R. R. & Potma, E. O. Stimulated Raman scattering: from bulk to nano. Chem. Rev. 117, 5070–5094 (2016).
Stolen, R. & Lin, C. Self-phase-modulation in silica optical fibers. Phys. Rev. A 17, 1448 (1978).
Tomlinson, W., Stolen, R. & Shank, C. Compression of optical pulses chirped by self-phase modulation in fibers. JOSA B 1, 139–149 (1984).
Nakamura, T., Ramaiah Badarla, V., Hashimoto, K., Schunemann, P. G. & Ideguchi, T. Simple approach to broadband mid-infrared pulse generation with a mode-locked Yb-doped fiber laser. Opt. Lett. 47, 1790–1793 (2022).
Selm, R., Krauss, G., Leitenstorfer, A. & Zumbusch, A. Simultaneous second-harmonic generation, third-harmonic generation, and four-wave mixing microscopy with single sub-8 fs laser pulses. Appl. Phys. Lett. 99, 181124 (2011).
Xiong, H. et al. Stimulated Raman excited fluorescence spectroscopy and imaging. Nat. Photon. 13, 412–417 (2019).
Palonpon, A. F. et al. Raman and SERS microscopy for molecular imaging of live cells. Nat. Protoc. 8, 677–692 (2013).
Yamakoshi, H. et al. Imaging of EdU, an alkyne-tagged cell proliferation probe, by Raman microscopy. J. Am. Chem. Soc. 133, 6102–6105 (2011).
Kuramochi, H., Takeuchi, S. & Tahara, T. Femtosecond time-resolved impulsive stimulated Raman spectroscopy using sub-7-fs pulses: apparatus and applications. Rev. Sci. Instrum. 87, 043107 (2016).
Paxinos, G. & Franklin, K. B. Paxinos and Franklin’s the Mouse Brain in Stereotaxic Coordinates (Academic Press, 2019).
Rohleder, D., Kiefer, W. & Petrich, W. Quantitative analysis of serum and serum ultrafiltrate by means of Raman spectroscopy. Analyst 129, 906–911 (2004).
Saade, J. et al. Glicemical analysis of human blood serum using FT-Raman: a new approach. Photomed. Laser Surg. 30, 388–392 (2012).
Giansante, S., Giana, H. E., Fernandes, A. B. & Silveira, L. Jr Analytical performance of Raman spectroscopy in assaying biochemical components in human serum. Lasers Med. Sci. 37, 287–298 (2022).
Medipally, D. K. et al. Development of a high throughput (HT) Raman spectroscopy method for rapid screening of liquid blood plasma from prostate cancer patients. Analyst 142, 1216–1226 (2017).
Friedman, A. N. & Fadem, S. Z. Reassessment of albumin as a nutritional marker in kidney disease. J. Am. Soc. Nephrol. 21, 223–230 (2010).
Alves-Bezerra, M. & Cohen, D. E. Triglyceride metabolism in the liver. Compr. Physiol. 8, 1–22 (2018).
Kannel, W. B., Castelli, W. P. & Gordon, T. Cholesterol in the prediction of atherosclerotic disease: new perspectives based on the Framingham study. Ann. Intern. Med. 90, 85–91 (1979).
Don, B. R. & Kaysen, G. Poor nutritional status and inflammation: serum albumin: relationship to inflammation and nutrition. Semin. Dial. 17, 432–437 (2004).
Busher, J. T. in Clinical Methods: The History, Physical, and Laboratory Examinations (eds Walker, H. K. et al.) Ch. 101 (Butterworths, 1990).
Cheng, J.-X. & Xie, X. S. Coherent Raman Scattering Microscopy (CRC Press, 2012).
Camp, C. H. Jr, Lee, Y. J. & Cicerone, M. T. Quantitative, comparable coherent anti-Stokes Raman scattering (CARS) spectroscopy: correcting errors in phase retrieval. J. Raman Spectrosc. 47, 408–415 (2016).
Pollard, W. T. & Mathies, R. A. Analysis of femtosecond dynamic absorption spectra of nonstationary states. Annu. Rev. Phys. Chem. 43, 497–523 (1992).
Kuramochi, H. & Tahara, T. Tracking ultrafast structural dynamics by time-domain Raman spectroscopy. J. Am. Chem. Soc. 143, 9699–9717 (2021).
Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 25, 44–56 (2019).
Liu, X., Shi, L., Zhao, Z., Shu, J. & Min, W. VIBRANT: spectral profiling for single-cell drug responses. Nat. Methods 21, 501–511 (2024).
Liu, Y., Lee, Y. J. & Cicerone, M. T. Broadband CARS spectral phase retrieval using a time-domain Kramers–Kronig transform. Opt. Lett. 34, 1363–1365 (2009).
Vartiainen, E. M., Rinia, H. A., Müller, M. & Bonn, M. Direct extraction of Raman line-shapes from congested CARS spectra. Opt. Express 14, 3622–3630 (2006).
Xiong, H. et al. Super-broadband stimulated Raman scattering spectroscopy and imaging. figshare https://doi.org/10.6084/m9.figshare.30379255 (2025).
Acknowledgements
We thank Y. Li of College of Chemistry and Molecular Engineering at Peking University and the Materials Processing and Analysis Center at Peking University for assistance with Raman characterization. This work is supported by STI2030-Major Projects 2021ZD0202500 (to H.X.) and National Natural Science Foundation of China 32250004 (to H.X.).
Author information
Authors and Affiliations
Contributions
H.X. conceived the concept and designed the system. J.G. constructed the instrument under H.X.’s guidance. W.S. and M.W. collected the serum samples and performed the benchmark biochemistry measurements. Q.Y., J.G. and Y.Q. performed the serum measurements and corresponding data processing. H.Z. built all the electronics for measurements. J.W. and J.G. developed the image acquisition software. J.R. and X.Z. helped prepare the liver tissue samples. Q.Z. and X.Y. prepared the brain tissue samples. J.G., H.X., J.L., H.S., Z.W. and W.Y. analysed the data. H.X. and J.G. wrote the paper with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Photonics thanks Yasuyuki Ozeki and Jian Shu for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 A naïve attempt at broadband transient stimulated Raman scattering (T-SRS).
(a) Combination of the pump pulse and Stokes pulse in conventional SRS setups. DM for dichroic mirror, RF for radio frequency, EOM for electro-optic modulator. (b) Autocorrelations of the few-cycle pulse before and after the reflection from a dichroic mirror with a spectral turning edge near the pulse spectrum. SHG for second harmonic generation. (c) Typical broadband T-SRS spectra of dimethyl sulfoxide (DMSO) and D6-DMSO mixture (1:1 volume ratio) achieved with the naïve design. Spectra acquired at different time delays between the pump pulse and the Stokes pulse are shown here. Red curve in (c) shows significantly reduced bandwidth; orange curve in (c) shows strong spectral distortions and low excitation efficiency (detailed theoretical derivation in SI).
Extended Data Fig. 2 Linewidth of the ring deformation mode of 1-naphthylnitrile.
(a) Time-domain SRL signal of 300-mM 1-naphthylnitrile in DMSO. A 16-ps time-delay range was recorded to achieve the natural linewidth limit of the mode. (b) Corresponding SuperB-SRS spectrum (lower panel) and spontaneous Raman spectrum (upper panel, recorded at ~1.3 cm−1 spectral resolution). The linewidth of the ring deformation mode was determined by least-squares fitting of the corresponding spectral peaks to a Lorentzian lineshape, with a 95% confidence interval provided.
Extended Data Fig. 3 Acquisition speed comparison between SuperB-SRS and spontaneous Raman.
Same sample (that is, the mixture of DMSO and D6-DMSO with a 1:1 volume ratio) was used. Spontaneous Raman was recorded with a commercial confocal spontaneous Raman spectrometer (LabRAM Odyssey, Horiba). ~14-mW/µm2 intensity of 532-nm CW laser was used for spontaneous Raman excitation. To achieve a comparable spectral resolution to that of SuperB-SRS, 3 times grating rotation is required to cover the spectral bandwidth shown above. Pixel dwell time of spontaneous Raman is set to 1 s, so the whole spectral acquisition time for spontaneous Raman is more than 3 s. SuperB-SRS is excited by ~29 mW/µm2 overall pump and Stokes intensity (that is, 14.8-mW Stokes power and 8.5-mW pump power focused by 1.2 N.A. objective). Spectral acquisition time for SuperB-SRS is 8 ms (that is, the time for one round of time delay scanning). Note that the excitation efficiency of SuperB-SRS over the whole spectral band is not homogenous. SNRs of Raman modes at the center of the excitation band (that is, the mode at 1,420 cm−1, etc.) for SuperB-SRS is 7.4 times higher than that of spontaneous Raman. However, SNRs of Raman modes at the excitation band edges (that is, those modes near 500 cm−1) are smaller than that of the spontaneous Raman. The SNRs were calculated as the ratio of the Raman peak amplitudes to the standard deviation of the spectral baseline between 2400 cm⁻¹ and 2500 cm⁻¹.
Extended Data Fig. 4 Sensitivity characterization of SuperB-SRS.
Different concentrations of rhodamine 800 (Rh800) in D6-DMSO (a–c) and the C-H stretching modes of DMSO in D2O (d–f) were tested. All data are measured with a 0.4 N.A. objective. The pump and Stokes powers for Rh800 measurements are 7.6 mW and 13.5 mW, respectively. The pump and Stokes powers for DMSO measurements are 12.7 mW and 22.5 mW, respectively. The error bars in (c) represent the standard deviations of 50 cm−1 range of the off-resonance region around 1649 cm−1. The error bars in (f) represent the standard deviation of 50 cm−1 range of the off-resonance region around 2913 cm−1.
Extended Data Fig. 5 Detection of very low-wavenumber modes by spectral filtering.
(a) Signal origins of the high-wavenumber and low-wavenumber modes (that is, interband and intraband interactions), respectively. The yellow-fill regions show the spectral components that reach the detectors. (b) SuperB-SRS spectrum of benzonitrile that covers the 100 cm−1 to 2500 cm−1 range. The red curve is acquired with spectral filtering, dark blue curve with no spectral filtering, and light blue curve in the upper panel shows the benchmark spontaneous Raman result.
Extended Data Fig. 6 Three replicate experiments of liver imaging.
a–c, Images of the vibrational band at 1657 cm−1 from three independent replicates. d, Representative SuperB-SRS spectra from the dashed box regions in a–c. The image size is 200 × 200 pixels (100 µm × 100 µm). Image in (c) is from the same data set of Fig. 3. All other imaging configurations are the same as Fig. 3. Scale bar: 10 µm.
Extended Data Fig. 7 Single-cell SuperB-SRS spectral analysis for microglia identification.
(a) SuperB-SRS image of a 40 µm thick mouse brain slice. 24 adjacent images of 150 × 150 pixels (6-pixel overlaps on the edges were applied) were captured at a 1-µm pixel size (that is, the image size is 870 µm × 432 µm). the Stokes power was set to 14 mW; the pump powers were set to 5 mW and 3.6 mW for pump bands A and B, respectively. (b) Dual-color two-photon excited (2PE) fluorescence imaging of the same region [DAPI (blue) and Iba1 immunostaining with Alexa Fluor 568 (green)]. (c) Single-cell spectral analysis. Each spectrum represents the average of all pixels within a 12-pixel-diameter circle centered on a DAPI-stained nucleus. (d) Receiver operating characteristic (ROC) curve evaluating the classification model performance. AUC, area under the curve. Scale bar: 100 µm.
Supplementary information
Supplementary Information (download PDF )
Time-delay calibration and SuperB-SRS spectral reconstruction; excitation efficiency of SuperB-SRS; signal generation and detection in SuperB-SRS and spectroscopic CARS; a rigorous comparison between SuperB-SRS and FT-CARS; Figs. 1–6 and Tables 1–4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, J., Wang, M., Yu, Q. et al. Super-broadband stimulated Raman scattering spectroscopy and imaging. Nat. Photon. (2026). https://doi.org/10.1038/s41566-025-01841-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41566-025-01841-8