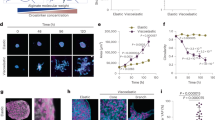
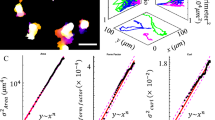
Abstract
Coordinated cell rotation along a curved matrix interface can sculpt epithelial tissues into spherical morphologies. Subsequently, radially oriented invasion of multicellular strands or branches can occur by local remodelling of the confining matrix. These symmetry-breaking transitions emerge from the dynamic reciprocity between cells and matrix but remain poorly understood. Here we show that epithelial cell spheroids collectively transition from circumferential orbiting to radial invasion via bidirectional interactions with the surrounding matrix curvature. Initially, spheroids exhibit an ellipsoidal shape but become rounded as orbiting occurs. In turn, orbiting along sharper curvature results in locally stronger contractile tractions, which gradually align collagen fibres in the radial direction. Thus, the initially elongated morphology primes the matrix towards subsequent invasion of two to four strands that are roughly aligned with its major axis. We then show that orbiting can be arrested and invasion can be reversed using osmotic pressure. We also investigate coordinated orbiting in mosaic spheroids, showing that a small fraction of cells with weakened cell–cell adhesions can impede collective orbiting but still invade into the matrix. This work elucidates how symmetry breaking in tissue morphogenesis is governed by the interplay of collective migration and the local curvature of the cell–matrix interface, with relevance for embryonic development and tumour progression.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The experimental data that support the findings of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper. These data are also available via Figshare at https://doi.org/10.6084/m9.figshare.30686246 (ref. 56). Owing to the file size, computational results in Fig. 6 are available via GitHub at https://github.com/carlesfalco/Spheroid-Orbiting-Model/tree/main/Data%20Fig%206%20(model).
Code availability
All code used for the displacement analysis in Figs. 1–3 and Supplementary Information are available via GitHub at https://github.com/TaeJeong0127/Displacement_Orbiting. All code used for the computational model in Fig. 6 and Supplementary Information are available via GitHub at https://github.com/carlesfalco/Spheroid-Orbiting-Model.
References
Rørth, P. Fellow travellers: emergent properties of collective cell migration. EMBO Rep. 13, 984 (2012).
Tanner, K., Mori, H., Mroue, R., Bruni-Cardoso, A. & Bissell, M. Coherent angular motion in the establishment of multicellular architecture of glandular tissues. Proc. Natl Acad. Sci. USA 109, 1973 (2012).
Wang, H., Lacoche, S., Huang, L., Xue, B. & Muthuswamy, S. Rotational motion during three-dimensional morphogenesis of mammary epithelial acini relates to laminin matrix assembly. Proc. Natl Acad. Sci. USA 110, 163 (2013).
Chin, A. et al. Epithelial cell chirality revealed by three-dimensional spontaneous rotation. Proc. Natl Acad. Sci. USA 115, 12188 (2018).
Fessenden, T. et al. Dia1-dependent adhesions are required by epithelial tissues to initiate invasion. J. Cell Biol. 217, 1485–1502 (2018).
Fernández, P. A. et al. Surface-tension-induced budding drives alveologenesis in human mammary gland organoids. Nat. Phys. 17, 1130 (2021).
Brandstätter, T. et al. Curvature induces active velocity waves in rotating spherical tissues. Nat. Commun. 14, 1643 (2023).
Lu, L. et al. Polarity-driven three-dimensional spontaneous rotation of a cell doublet. Nat. Phys. 20, 1194–1203 (2024).
Haigo, S. L. & Bilder, D. Global tissue revolutions in a morphogenetic movement controlling elongation. Science 331, 1071 (2011).
Cetera, M. et al. Epithelial rotation promotes the global alignment of contractile actin bundles during Drosophila egg chamber elongation. Nat. Commun. 5, 5511 (2014).
Doxzen, K. et al. Guidance of collective cell migration by substrate geometry. Integr. Biol. 5, 1026 (2013).
Segerer, F. J., Thüroff, F., Piera Alberola, A., Frey, E. & Rädler, J. O. Emergence and persistence of collective cell migration on small circular micropatterns. Phys. Rev. Lett. 114, 228102 (2015).
Jain, S. et al. The role of single cell mechanical behavior and polarity in driving collective cell migration. Nat. Phys. 16, 802–809 (2020).
Lo Vecchio, S., Pertz, O., Szopos, M., Navoret, L. & Riveline, D. Spontaneous rotations in epithelia as an interplay between cell polarity and boundaries. Nat. Phys. 20, 322–331 (2024).
Glentis, A. et al. The emergence of spontaneous coordinated epithelial rotation on cylindrical curved surfaces. Sci. Adv. 8, eabn5406 (2022).
Kaufman, L. et al. Glioma expansion in collagen I matrices: analyzing collagen concentration-dependent growth and motility patterns. Biophys. J. 89, 635–650 (2005).
Wolf, K. et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat. Cell Biol. 9, 893 (2007).
Gaggioli, C. et al. Fibroblast-led collective invasion of carcinoma cells with differing roles for rhogtpases in leading and following cells. Nat. Cell Biol. 9, 1392 (2007).
Cheung, K., Gabrielson, E., Werb, Z. & Ewald, A. Collective invasion in breast cancer requires a conserved basal epithelial program. Cell 155, 1639–1651 (2013).
Carey, S., Starchenko, A., McGregor, A. & Reinhart-King, C. Leading malignant cells initiate collective epithelial cell invasion in a three-dimensional heterotypic tumor spheroid model. Clin. Exp. Metastasis 30, 615 (2013).
Kopanska, K. S., Alcheikh, Y., Staneva, R., Vignjevic, D. & Betz, T. Tensile forces originating from cancer spheroids facilitate tumor invasion. PloS ONE 11, e0156442 (2016).
Guzman, A., Alemany, V. S., Nguyen, Y., Zhang, C. R. & Kaufman, L. J. A novel 3D in vitro metastasis model elucidates differential invasive strategies during and after breaching basement membrane. Biomaterials 115, 19 (2017).
Mark, C. et al. Collective forces of tumor spheroids in three-dimensional biopolymer networks. eLife 9, e51912 (2020).
Summerbell, E. et al. Epigenetically heterogeneous tumor cells direct collective invasion through filopodia-driven fibronectin micropatterning. Sci. Adv. 6, eaaz6197 (2020).
Perrin, L., Belova, E., Bayarmagnai, B., Tüzel, E. & Gligorijevic, B. Invadopodia enable cooperative invasion and metastasis of breast cancer cells. Commun. Biol. 5, 758 (2022).
Cai, G. et al. Matrix confinement modulates 3D spheroid sorting and burst-like collective migration. Acta Biomater. 179, 192–206 (2024).
Guo, M. et al. Vimentin intermediate filaments as structural and mechanical coordinators of mesenchymal cells. Nat. Cell Biol. 27, 1210–1218 (2025).
Foty, R. A. & Steinberg, M. S. Cadherin-mediated cell-cell adhesion and tissue segregation in relation to malignancy. Int. J. Dev. Biol. 48, 397 (2004).
Cerchiari, A. et al. A strategy for tissue self-organization that is robust to cellular heterogeneity and plasticity. Proc. Natl Acad. Sci. USA 112, 2287–2292 (2015).
Buttenschön, A. & Edelstein-Keshet, L. Bridging from single to collective cell migration: a review of models and links to experiments. PLoS Comput. Biol. 16, e1008411 (2020).
Trush, O. et al. N-cadherin orchestrates self-organization of neurons within a columnar unit in the Drosophila medulla. J. Neurosci. 39, 5861 (2019).
Falcó, C., Baker, R. E. & Carrillo, J. A. A local continuum model of cell-cell adhesion. SIAM J. Appl. Math. 84, S17–S42 (2024).
D’Orsogna, M. R., Chuang, Y.-L., Bertozzi, A. L. & Chayes, L. S. Self-propelled particles with soft-core interactions: patterns, stability, and collapse. Phys. Rev. Lett. 96, 104302 (2006).
Li, Y.-X., Lukeman, R. & Edelstein-Keshet, L. Minimal mechanisms for school formation in self-propelled particles. Phys. D 237, 699 (2008).
Albi, G., Balague, D., Carrillo, J. A. & von Brecht, J. Stability analysis of flock and mill rings for second order models in swarming. SIAM J. Appl. Math. 74, 794 (2014).
Khoo, A. S. et al. Breast cancer cells transition from mesenchymal to amoeboid migration in tunable three-dimensional silk-collagen hydrogels. ACS Biomater. Sci. Eng. 5, 4341 (2019).
Landauer, A., Patel, M., Henann, D. & Franck, C. A q-factor-based digital image correlation algorithm (qDIC) for resolving finite deformations with degenerate speckle patterns. Exp. Mech. 58, 815 (2018).
Leggett, S. E. et al. Mechanophenotyping of 3D multicellular clusters using displacement arrays of rendered tractions. Proc. Natl Acad. Sci. USA 117, 5655 (2020).
Rittié, L. (ed.) Fibrosis: Methods and Protocols (Humana Press, 2017).
Janmey, P. A. et al. Negative normal stress in semiflexible biopolymer gels. Nat. Mater. 6, 48 (2007).
Wong, I. Y. et al. Collective and individual migration following the epithelial–mesenchymal transition. Nat. Mater. 13, 1063 (2014).
Bertozzi, A. L., Kolokolnikov, T., Sun, H., Uminsky, D. & Brecht, J. V. Ring patterns and their bifurcations in a nonlocal model of biological swarms. Commun. Math. Sci. 13, 955 (2015).
Carrillo, J. A., Colombi, A. & Scianna, M. Adhesion and volume constraints via nonlocal interactions determine cell organisation and migration profiles. J. Theor. Biol. 445, 75 (2018).
Cheng, G., Tse, J., Jain, R. K. & Munn, L. L. Micro-environmental mechanical stress controls tumor spheroid size and morphology by suppressing proliferation and inducing apoptosis in cancer cells. PLoS ONE 4, e4632 (2009).
Mills, K. L., Kemkemer, R., Rudraraju, S. & Garikipati, K. Elastic free energy drives the shape of prevascular solid tumors. PLoS ONE 9, e103245 (2014).
Tang, W. et al. Collective curvature sensing and fluidity in three-dimensional multicellular systems. Nat. Phys. 18, 1371 (2022).
Boghaert, E. et al. Host epithelial geometry regulates breast cancer cell invasiveness. Proc. Natl Acad. Sci. USA 109, 19632 (2012).
Yu, M. et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 339, 580 (2013).
Wittmann, T., Dema, A. & van Haren, J. Lights, cytoskeleton, action: optogenetic control of cell dynamics. Curr. Opin. Cell Biol. 66, 1 (2020).
Di Ciano-Oliveira, C., Thirone, A. C. P., Szászi, K. & Kapus, A. Osmotic stress and the cytoskeleton: the R(h)ole of Rho GTPases. Acta Physiol. 187, 257 (2006).
Araújo, N. A. et al. Steering self-organisation through confinement. Soft Matter 19, 1695 (2023).
Martinson, W. D. et al. Dynamic fibronectin assembly and remodeling by leader neural crest cells prevents jamming in collective cell migration. eLife 12, e83792 (2023).
Dias, C. S., Trivedi, M., Volpe, G., Araújo, N. A. & Volpe, G. Environmental memory boosts group formation of clueless individuals. Nat. Commun. 14, 7324 (2023).
Tsingos, E., Bakker, B. H., Keijzer, K. A., Hupkes, H. J. & Merks, R. M. Hybrid cellular potts and bead-spring modeling of cells in fibrous extracellular matrix. Biophys. J. 122, 2609 (2023).
Joanny, J.-F. & Indekeu, J. O. Statistical physics of active matter, cell division and cell aggregation. Phys. A 631, 129314 (2023).
Kim, J. et al. Collective transitions from orbiting to matrix invasion in three-dimensional multicellular spheroids. figshare https://doi.org/10.6084/m9.figshare.30686246 (2025).
Acknowledgements
We thank S. E. Leggett, R. E. Baker, J. Notbohm, D. Bhaskar, J. Yang and A. McGhee for helpful conversations, as well as J. S. Brugge and D. A. Haber for the gift of stably transfected MCF-10A cell lines. I.Y.W. and J.A.C. also thank P. Kulesa and P. Maini for catalysing this collaboration. We acknowledge funding from Brown University’s Hibbitt Engineering Postdoctoral Fellowship (J.K.), NIH R01GM140108 (J.K., H.J., A.M.H., A.M., H.Y., S.A.A., M.G. and I.Y.W.), ‘la Caixa’ Foundation Fellowship 100010434 with code LCF/BQ/EU21/11890128 (C. Falcó), EPSRC grant EP/R014604/1 (C. Falcó, W.D.M. and J.A.C.), ERC Horizon 2020 Research and Innovation Program Advanced Grant Non-local-CPD 883363 (W.D.M. and J.A.C.), ONR Panther Award N000142212828 (M.A. and C. Franck), MIT School of Engineering Takeda Fellowship (H.Y.) and ARO W911NF2310385 (I.Y.W.). W.D.M. and J.A.C. also thank the Isaac Newton Institute for Mathematical Sciences, Cambridge, for support and hospitality during the programme ‘Mathematics of movement: an interdisciplinary approach to mutual challenges in animal ecology and cell biology’, where work on this paper was undertaken.
Author information
Authors and Affiliations
Contributions
I.Y.W. conceived and supervised the project. J.K. and I.Y.W. designed experimental work. J.K., A.M.H. and A.M. performed spheroid experiments. J.K., H.J., M.A., C. Franck and I.Y.W. analysed collective migration and tractions. J.K., H.Y., V.C.F. and M.G. characterized matrix architecture and rheology. S.A.A. contributed cell lines. C. Falcó, W.D.M., J.A.C. and I.Y.W. designed computational and theoretical work. C. Falcó and W.D.M. implemented theoretical model and performed simulations. J.K., H.J., C. Falcó, W.D.M. and I.Y.W. wrote the paper with feedback from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Physics thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–31 and Tables 1 and 2.
Supplementary Video 1 (download AVI )
Representative video of an orbiting spheroid, corresponding to snapshots in Fig. 1a.
Supplementary Video 2 (download AVI )
Velocity profile corresponding to the snapshots in Fig. 1d,e. Circumferential velocity (left), radial velocity (right).
Supplementary Video 3 (download AVI )
Colour-coded radial deviation and roundness corresponding to the snapshots in Fig. 2a,c. Grey-dotted lines represent the equivalent circle at every time point.
Supplementary Video 4 (download AVI )
Representative visualization of temporal displacement fields of beads and trajectories of cell migration (for 150 min) over time, corresponding to snapshots in Fig. 3a. Quivers indicate the direction of displacement with scaled size, and the colour of each vector indicates the radial components of the displacement vectors. The red–orange colour range indicates the temporal position of trajectories.
Supplementary Video 5 (download AVI )
Representative visualization of cumulative displacement fields of beads and trajectories of cell migration (for 150 min) over time, corresponding to snapshots in Fig. 3b. Quivers indicate the direction of displacement with scaled size, and the colour of each vector indicates the radial components of the displacement vectors. The red–orange colour range indicates the temporal position of trajectories.
Supplementary Video 6 (download AVI )
Representative visualization of cumulative displacement fields for various morphologies of the spheroid over time, corresponding to the results of Supplementary Fig. 7. Quivers indicate the direction of displacement with scaled size, and the colour of each vector indicates the radial components of the displacement vectors. Top left, medium; top right, small; bottom left, large; bottom right, elongated.
Supplementary Video 7 (download AVI )
Representative videos of spheroids under different PEG conditions. 0 → 0% PEG (left), and 0 → 4% PEG (right). Media changed at 48 h.
Supplementary Video 8 (download AVI )
Representative videos of spheroids under different PEG conditions. 4 → 4% PEG (left), and 4 → 0% PEG (right). Media changed at 48 h.
Supplementary Video 9 (download AVI )
Representative video of a 10% Snail mosaic spheroid, corresponding to snapshots in Fig. 5a.
Supplementary Video 10 (download AVI )
Representative video of a 30% Snail mosaic spheroid, corresponding to snapshots in Fig. 5f.
Supplementary Video 11 (download AVI )
Representative video of a GM6001-treated spheroid, corresponding to Supplementary Fig. 15a.
Supplementary Video 12 (download MP4 )
Representative simulations of uncoordinated, global orbiting and peripheral orbiting conditions within a circular domain, corresponding to snapshots in Fig. 6a,b. Adhesive parameters \({f}_{{\rm{a}}}^{{\rm{cc}}}/{F}_{{\rm{c}}},\,{f}_{{\rm{a}}}^{{\rm{cm}}}/{F}_{{\rm{c}}}\), correspond to (0.05,0.45), (0.2,0.2) and (0.2,0.4), respectively.
Supplementary Video 13 (download MP4 )
Representative simulations of one, two or three geometric perturbations of the boundary, corresponding to snapshots in Fig. 6c–f. Boundary perturbation parameters, (h /d, k /d) given from left to right, by (1,1), (1,4) and (2.4,4).
Supplementary Video 14 (download MP4 )
Representative simulations of mosaic populations with varying Snail composition, corresponding to snapshots in Fig. 6g,h. Wildtype (1) and Snail (2) cells adhesive parameters are given by \({f}_{{\rm{a1,1}}}^{{\rm{cc}}}=0.2{F}_{{\rm{c}}},\,{f}_{{\rm{a1,2,}}}^{{\rm{cc}}}{f}_{{\rm{a2,1}}}^{{\rm{cc}}}{f}_{{\rm{a2,2}}}^{{\rm{cc}}}=0,{f}_{{\rm{a1}}}^{{\rm{cm}}}=0.2{F}_{{\rm{c}}}\) and \({f}_{{\rm{a2}}}^{{\rm{cm}}}=0.8{F}_{{\rm{c}}}\).
Source data
Source Data Fig. 1 (download XLSX )
Optical flow results and coordination parameter data.
Source Data Fig. 2 (download XLSX )
Geometrical parameters, rounding times and orbiting time measurements.
Source Data Fig. 3 (download XLSX )
Matrix deformation analysis results (cumulative/temporal) and collagen fibre alignments.
Source Data Fig. 4 (download XLSX )
Projected areas of spheroids under osmotic pressure.
Source Data Fig. 5 (download XLSX )
Optical flow results for mosaic spheroids.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, J., Jeong, H., Falcó, C. et al. Collective transitions from orbiting to matrix invasion in three-dimensional multicellular spheroids. Nat. Phys. 22, 275–286 (2026). https://doi.org/10.1038/s41567-025-03150-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41567-025-03150-x