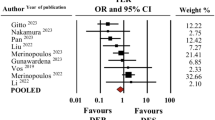
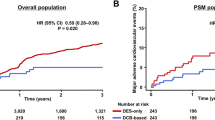
Abstract
Drug-coated balloons (DCBs) are devices used for the treatment of both coronary artery disease (CAD) and peripheral artery disease (PAD). One of the hypothesized advantages of DCB angioplasty over stent implantation is that DCB angioplasty does not result in the presence of a permanent metallic scaffold in the vessel wall. However, DCB angioplasty also has some important limitations, such as a potentially lower efficacy compared with other modalities; therefore, the role of DCBs in the treatment of CAD and PAD is not fully defined. Over the past 20 years, many clinical trials have been performed to investigate the use of these devices for a variety of indications. In this Review, we describe the device design and mechanism of action of DCBs and discuss important procedural considerations for successful DCB use. We summarize the scientific and clinical evidence for DCB angioplasty in CAD and PAD. In addition, we highlight the recommendations for DCB use in clinical practice guidelines and provide perspectives on the potential future role that DCB angioplasty might have in the treatment of CAD and PAD.
Key points
-
Drug-coated balloon (DCB) angioplasty provides an alternative to drug-eluting stents and plain old balloon angioplasty for the treatment of coronary or peripheral artery disease.
-
The absence of a metallic scaffold with DCB angioplasty compared with stenting might confer a biomechanical and physiological benefit and avoids the implantation of additional stent layers in patients with in-stent restenosis.
-
A class effect cannot be assumed for DCBs; device manufacturers are challenged with finding the best combination of antiproliferative agent and excipient to achieve optimal clinical and angiographic outcomes.
-
Several randomized clinical trials have been performed comparing DCB angioplasty with a variety of comparators in both coronary and peripheral artery disease, although more evidence is needed, particularly in de novo coronary artery disease.
-
Positive results from a trial comparing a paclitaxel-coated balloon with an uncoated balloon in patients with coronary in-stent restenosis led to the approval of a DCB for clinical use in the USA in 2024.
-
In long-segment diffuse atherosclerotic disease, DCBs and drug-eluting stents can be considered to be complementary rather than competing strategies, with DCB angioplasty providing an approach to reduce the number of implanted stents.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Scheller, B. et al. Treatment of coronary in-stent restenosis with a paclitaxel-coated balloon catheter. N. Engl. J. Med. 355, 2113–2124 (2006).
Speck, U. et al. Do pharmacokinetics explain persistent restenosis inhibition by a single dose of paclitaxel? Circ. Cardiovasc. Interv. 5, 392–400 (2012).
Scheller, B., Speck, U., Schmitt, A., Böhm, M. & Nickenig, G. Addition of paclitaxel to contrast media prevents restenosis after coronary stent implantation. J. Am. Coll. Cardiol. 42, 1415–1420 (2003).
Axel, D. I. et al. Paclitaxel inhibits arterial smooth muscle cell proliferation and migration in vitro and in vivo using local drug delivery. Circulation 96, 636–645 (1997).
Oberhoff, M. et al. Inhibition of smooth muscle cell proliferation after local drug delivery of the antimitotic drug paclitaxel using a porous balloon catheter. Basic Res. Cardiol. 96, 275–282 (2001).
Scheller, B. et al. Paclitaxel balloon coating, a novel method for prevention and therapy of restenosis. Circulation 110, 810–814 (2004).
Cortese, B. & Bertoletti, A. Paclitaxel coated balloons for coronary artery interventions: a comprehensive review of preclinical and clinical data. Int. J. Cardiol. 161, 4–12 (2012).
Chen, Y. et al. Biolimus-coated versus paclitaxel-coated balloons for coronary in-stent restenosis (BIO ASCEND ISR): a randomised, non-inferiority trial. EuroIntervention 20, e806–e817 (2024).
Katsanos, K., Spiliopoulos, S., Kitrou, P., Krokidis, M. & Karnabatidis, D. Risk of death following application of paclitaxel-coated balloons and stents in the femoropopliteal artery of the leg: a systematic review and meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 7, e011245 (2018).
Nordanstig, J. et al. Mortality with paclitaxel-coated devices in peripheral artery disease. N. Engl. J. Med. 383, 2538–2546 (2020).
Freisinger, E. et al. Mortality after use of paclitaxel-based devices in peripheral arteries: a real-world safety analysis. Eur. Heart J. 41, 3732–3739 (2019).
Scheller, B. et al. Survival after coronary revascularization with paclitaxel-coated balloons. J. Am. Coll. Cardiol. 75, 1017–1028 (2020).
Habara, S. et al. Effectiveness of paclitaxel-eluting balloon catheter in patients with sirolimus-eluting stent restenosis. JACC Cardiovasc. Interv. 4, 149–154 (2011).
Rittger, H. et al. A randomized, multicenter, single-blinded trial comparing paclitaxel-coated balloon angioplasty with plain balloon angioplasty in drug-eluting stent restenosis: the PEPCAD-DES study. J. Am. Coll. Cardiol. 59, 1377–1382 (2012).
Habara, S. et al. A multicenter randomized comparison of paclitaxel-coated balloon catheter with conventional balloon angioplasty in patients with bare-metal stent restenosis and drug-eluting stent restenosis. Am. Heart J. 166, 527–533.e2 (2013).
Rittger, H. et al. Long-term outcomes after treatment with a paclitaxel-coated balloon versus balloon angioplasty: insights from the PEPCAD-DES study (treatment of drug-eluting stent [DES] in-stent restenosis with SeQuent Please paclitaxel-coated percutaneous transluminal coronary angioplasty [PTCA] catheter). JACC Cardiovasc. Interv. 8, 1695–1700 (2015).
Hamm, C. W. et al. A multicentre, randomised controlled clinical study of drug-coated balloons for the treatment of coronary in-stent restenosis. EuroIntervention 16, e328–e334 (2020).
Kufner, S. et al. A prospective trial of a novel low-dose paclitaxel-coated balloon therapy in patients with restenosis in drug-eluting coronary stents Intracoronary Stenting and Angiographic Results: optimizing treatment of Drug Eluting Stent In-stent REstenosis 3A (ISAR-DESIRE 3A). Catheter. Cardiovasc. Interv. 99, 754–762 (2022).
Colleran, R. et al. Comparative efficacy of two paclitaxel-coated balloons with different excipient coatings in patients with coronary in-stent restenosis: a pooled analysis of the Intracoronary Stenting and Angiographic Results: optimizing treatment of Drug Eluting Stent In-Stent Restenosis 3 and 4 (ISAR-DESIRE 3 and ISAR-DESIRE 4) trials. Int. J. Cardiol. 252, 57–62 (2018).
Ninomiya, K. et al. A prospective randomized trial comparing sirolimus-coated balloon with paclitaxel-coated balloon in de novo small vessels. JACC Cardiovasc. Interv. 16, 2884–2896 (2023).
Xu, K. et al. Biolimus-coated balloon in small-vessel coronary artery disease: the BIO-RISE China study. JACC Cardiovasc. Interv. 15, 1219–1226 (2022).
Byrne, R. A. et al. Randomized trial of biolimus DCB for in-stent restenosis: the primary results of the REFORM study. JACC Cardiovasc. Interv. 18, 654–662 (2025).
Malla, S. et al. in Paclitaxel: Sources, Chemistry, Anticancer Actions, and Current Biotechnology (eds Swamy, M. K., Pullaiah, T. & Chen, Z.-S.) 73–127 (Academic Press, 2021).
Solymosi, T. et al. Sirolimus formulation with improved pharmacokinetic properties produced by a continuous flow method. Eur. J. Pharm. Biopharm. 94, 135–140 (2015).
Levin, A. D., Vukmirovic, N., Hwang, C.-W. & Edelman, E. R. Specific binding to intracellular proteins determines arterial transport properties for rapamycin and paclitaxel. Proc. Natl Acad. Sci. USA 101, 9463–9467 (2004).
Clever, Y. P. et al. Paclitaxel and sirolimus differentially affect growth and motility of endothelial progenitor cells and coronary artery smooth muscle cells. EuroIntervention 7, K32–K42 (2011).
Wessely, R. et al. Randomized trial of rapamycin- and paclitaxel-eluting stents with identical biodegradable polymeric coating and design. Eur. Heart J. 28, 2720–2725 (2007).
Stone, G. W. et al. Comparison of an everolimus-eluting stent and a paclitaxel-eluting stent in patients with coronary artery disease: a randomized trial. JAMA 299, 1903–1913 (2008).
Caixeta, A. et al. Clinical follow-up 3 years after everolimus- and paclitaxel-eluting stents: a pooled analysis from the SPIRIT II (a clinical evaluation of the XIENCE V everolimus eluting coronary stent system in the treatment of patients with de novo native coronary artery lesions) and SPIRIT III (a clinical evaluation of the investigational device XIENCE V everolimus eluting coronary stent system [EECSS] in the treatment of subjects with de novo native coronary artery lesions) randomized trials. JACC Cardiovasc. Interv. 3, 1220–1228 (2010).
Stone, G. W. et al. Everolimus-eluting versus paclitaxel-eluting stents in coronary artery disease. N. Engl. J. Med. 362, 1663–1674 (2010).
Kedhi, E. et al. Second-generation everolimus-eluting and paclitaxel-eluting stents in real-life practice (COMPARE): a randomised trial. Lancet 375, 201–209 (2010).
Ahmad, W. A. W. et al. Treatment of coronary de novo lesions by a sirolimus- or paclitaxel-coated balloon. JACC Cardiovasc. Interv. 15, 770–779 (2022).
Radke, P. et al. Vascular effects of paclitaxel following drug-eluting balloon angioplasty in a porcine coronary model: the importance of excipient. EuroIntervention 7, 730–737 (2011).
Loh, J. P., Barbash, I. M. & Waksman, R. The current status of drug-coated balloons in percutaneous coronary and peripheral interventions. EuroIntervention 9, 979–988 (2013).
Serruys, P. W. et al. A comparison of balloon-expandable-stent implantation with balloon angioplasty in patients with coronary artery disease. Benestent Study Group. N. Engl. J. Med. 331, 489–495 (1994).
Fischman, D. L. et al. A randomized comparison of coronary-stent placement and balloon angioplasty in the treatment of coronary artery disease. Stent Restenosis Study Investigators. N. Engl. J. Med. 331, 496–501 (1994).
Tanaka, A. et al. Impact of angiographic result after predilatation on outcome after drug-coated balloon treatment of in-stent coronary restenosis. Am. J. Cardiol. 118, 1460–1465 (2016).
Lee, H. S. et al. Procedural optimization of drug-coated balloons in the treatment of coronary artery disease. Catheter. Cardiovasc. Interv. 98, E43–E52 (2021).
Rhee, T. M. et al. Impact of optimized procedure-related factors in drug-eluting balloon angioplasty for treatment of in-stent restenosis. JACC Cardiovasc. Interv. 11, 969–978 (2018).
Kleber, F. X. et al. Drug-coated balloons for treatment of coronary artery disease: updated recommendations from a consensus group. Clin. Res. Cardiol. 102, 785–797 (2013).
Leimgruber, P. P. et al. Influence of intimal dissection on restenosis after successful coronary angioplasty. Circulation 72, 530–535 (1985).
Cappelletti, A. et al. Short- and long-term evolution of unstented nonocclusive coronary dissection after coronary angioplasty. J. Am. Coll. Cardiol. 34, 1484–1488 (1999).
Lee, T. et al. Predictors of target lesion failure after percutaneous coronary intervention with a drug-coated balloon for de novo lesions. EuroIntervention 20, e818–e825 (2024).
Huber, M. S., Mooney, J. F., Madison, J. & Mooney, M. R. Use of a morphologic classification to predict clinical outcome after dissection from coronary angioplasty. Am. J. Cardiol. 68, 467–471 (1991).
Alfonso, F., Coughlan, J., Giacoppo, D., Kastrati, A. & Byrne, R. A. Management of in-stent restenosis. EuroIntervention 18, e103–e123 (2022).
Kufner, S. et al. Neointimal modification with scoring balloon and efficacy of drug-coated balloon therapy in patients with restenosis in drug-eluting coronary stents: a randomized controlled trial. JACC Cardiovasc. Interv. 10, 1332–1340 (2017).
Gao, X.-F. et al. Intravascular ultrasound vs angiography-guided drug-coated balloon angioplasty. JACC Cardiovasc. Interv. 17, 1519–1528 (2024).
Joner, M. et al. Preclinical evaluation of a novel drug-eluting balloon in an animal model of in-stent stenosis. J. Biomater. Appl. 27, 717–726 (2013).
Pósa, A. et al. Optimization of drug-eluting balloon use for safety and efficacy: evaluation of the 2nd generation paclitaxel-eluting DIOR-balloon in porcine coronary arteries. Catheter. Cardiovasc. Interv. 76, 395–403 (2010).
Ali, R. M. et al. Treatment of coronary drug-eluting stent restenosis by a sirolimus- or paclitaxel-coated balloon. JACC Cardiovasc. Interv. 12, 558–566 (2019).
Liistro, F. et al. Elutax paclitaxel-eluting balloon followed by bare-metal stent compared with Xience V drug-eluting stent in the treatment of de novo coronary stenosis: a randomized trial. Am. Heart J. 166, 920–926 (2013).
Yoshida, R. et al. Impact of adjunctive use of guide extension catheter on midterm outcome of drug-coated balloon angioplasty. EuroIntervention 15, 688–691 (2019).
Kuntz, R. E. & Baim, D. S. Defining coronary restenosis. Newer clinical and angiographic paradigms. Circulation 88, 1310–1323 (1993).
Otsuka, F. et al. Neoatherosclerosis: overview of histopathologic findings and implications for intravascular imaging assessment. Eur. Heart J. 36, 2147–2159 (2015).
Yeh, R. W. et al. Paclitaxel-coated balloon vs uncoated balloon for coronary in-stent restenosis: the AGENT IDE randomized clinical trial. JAMA 331, 1015–1024 (2024).
Boston Scientific. Boston Scientific receives FDA approval for the AGENT™ Drug-Coated Balloon. Boston Scientific https://news.bostonscientific.com/2024-03-01-Boston-Scientific-Receives-FDA-Approval-for-the-AGENT-TM-Drug-Coated-Balloon (2024).
Unverdorben, M. et al. Paclitaxel-coated balloon catheter versus paclitaxel-coated stent for the treatment of coronary in-stent restenosis: the three-year results of the PEPCAD II ISR study. EuroIntervention 11, 926–934 (2015).
Byrne, R. A. et al. Paclitaxel-eluting balloons, paclitaxel-eluting stents, and balloon angioplasty in patients with restenosis after implantation of a drug-eluting stent (ISAR-DESIRE 3): a randomised, open-label trial. Lancet 381, 461–467 (2013).
Adriaenssens, T. et al. Optical coherence tomography study of healing characteristics of paclitaxel-eluting balloons vs. everolimus-eluting stents for in-stent restenosis: the SEDUCE (Safety and Efficacy of a Drug elUting balloon in Coronary artery rEstenosis) randomised clinical trial. EuroIntervention 10, 439–448 (2014).
Alfonso, F. et al. A randomized comparison of drug-eluting balloon versus everolimus-eluting stent in patients with bare-metal stent–in-stent restenosis: the RIBS V clinical trial (Restenosis Intra-stent of Bare Metal Stents: paclitaxel-eluting balloon vs. everolimus-eluting stent). J. Am. Coll. Cardiol. 63, 1378–1386 (2014).
Xu, B. et al. A prospective, multicenter, randomized trial of paclitaxel-coated balloon versus paclitaxel-eluting stent for the treatment of drug-eluting stent in-stent restenosis: results from the PEPCAD China ISR trial. JACC Cardiovasc. Interv. 7, 204–211 (2014).
Alfonso, F. et al. A prospective randomized trial of drug-eluting balloons versus everolimus-eluting stents in patients with in-stent restenosis of drug-eluting stents: the RIBS IV randomized clinical trial. J. Am. Coll. Cardiol. 66, 23–33 (2015).
Pleva, L., Kukla, P., Kusnierova, P., Zapletalova, J. & Hlinomaz, O. Comparison of the efficacy of paclitaxel-eluting balloon catheters and everolimus-eluting stents in the treatment of coronary in-stent restenosis. Circ. Cardiovasc. Interv. 9, e003316 (2016).
Wong, Y. T. A. et al. Comparison of drug-eluting stents and drug-coated balloon for the treatment of drug-eluting coronary stent restenosis: a randomized RESTORE trial. Am. Heart J. 197, 35–42 (2018).
Baan, J. et al. A randomized comparison of paclitaxel-eluting balloon versus everolimus-eluting stent for the treatment of any in-stent restenosis. JACC Cardiovasc. Interv. 11, 275–283 (2018).
Jensen, C. et al. Angiographic and clinical performance of a paclitaxel-coated balloon compared to a second-generation sirolimus-eluting stent in patients with in-stent restenosis: the BIOLUX randomised controlled trial. EuroIntervention 14, 1096–1103 (2018).
Siontis, G. C. et al. Percutaneous coronary interventional strategies for treatment of in-stent restenosis: a network meta-analysis. Lancet 386, 655–664 (2015).
Giacoppo, D. et al. Treatment strategies for coronary in-stent restenosis: systematic review and hierarchical Bayesian network meta-analysis of 24 randomised trials and 4880 patients. BMJ 351, h5392 (2015).
Giacoppo, D. et al. Paclitaxel-coated balloon angioplasty vs. drug-eluting stenting for the treatment of coronary in-stent restenosis: a comprehensive, collaborative, individual patient data meta-analysis of 10 randomized clinical trials (DAEDALUS study). Eur. Heart J. 41, 3715–3728 (2019).
Giacoppo, D. et al. Drug-coated balloon angioplasty versus drug-eluting stent implantation in patients with coronary stent restenosis. J. Am. Coll. Cardiol. 75, 2664–2678 (2020).
Scheller, B. et al. Combined analysis of two parallel randomized trials of sirolimus-coated and paclitaxel-coated balloons in coronary in-stent restenosis lesions. Circ. Cardiovasc. Interv. 15, e012305 (2022).
Byrne, R. A. et al. 2023 ESC Guidelines for the management of acute coronary syndromes: developed by the task force on the management of acute coronary syndromes of the European Society of Cardiology (ESC). Eur. Heart J. 44, 3720–3826 (2023).
Vrints, C. et al. 2024 ESC Guidelines for the management of chronic coronary syndromes: developed by the task force for the management of chronic coronary syndromes of the European Society of Cardiology (ESC) endorsed by the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 45, 3415–3537 (2024).
Frutkin, A. D. et al. Drug-eluting stents and the use of percutaneous coronary intervention among patients with class I indications for coronary artery bypass surgery undergoing index revascularization: analysis from the NCDR (National Cardiovascular Data Registry). JACC Cardiovasc. Interv. 2, 614–621 (2009).
Cortese, B. et al. Drug-coated balloon versus drug-eluting stent for small coronary vessel disease: PICCOLETO II randomized clinical trial. JACC Cardiovasc. Interv. 13, 2840–2849 (2020).
Cortese, B. et al. Paclitaxel-coated balloon versus drug-eluting stent during PCI of small coronary vessels, a prospective randomised clinical trial. The PICCOLETO study. Heart 96, 1291–1296 (2010).
Funatsu, A. et al. A multicenter randomized comparison of paclitaxel-coated balloon with plain balloon angioplasty in patients with small vessel disease. Clin. Res. Cardiol. 106, 824–832 (2017).
Gao, C. et al. Drug-coated balloon angioplasty with rescue stenting versus intended stenting for the treatment of patients with de novo coronary artery lesions (REC-CAGEFREE I): an open-label, randomised, non-inferiority trial. Lancet 404, 1040–1050 (2024).
Gobić, D. et al. Drug-coated balloon versus drug-eluting stent in primary percutaneous coronary intervention: a feasibility study. Am. J. Med. Sci. 354, 553–560 (2017).
Hao, X. et al. Study on the safety and effectiveness of drug-coated balloons in patients with acute myocardial infarction. J. Cardiothorac. Surg. 16, 178 (2021).
Jeger, R. V. et al. Drug-coated balloons for small coronary artery disease (BASKET-SMALL 2): an open-label randomised non-inferiority trial. Lancet 392, 849–856 (2018).
Latib, A. et al. A randomized multicenter study comparing a paclitaxel drug-eluting balloon with a paclitaxel-eluting stent in small coronary vessels: the BELLO (Balloon Elution and Late Loss Optimization) study. J. Am. Coll. Cardiol. 60, 2473–2480 (2012).
Liu, S. et al. Comparison of drug-coated balloon and drug-eluting stent for the treatment of small vessel disease (from the Dissolve SVD randomized trial). Am. J. Cardiol. 211, 29–39 (2024).
Nakamura, M. et al. Drug-coated balloon for the treatment of small vessel coronary artery disease — a randomized non-inferiority trial. Circ. J. 87, 287–295 (2023).
Nishiyama, N. et al. Clinical value of drug-coated balloon angioplasty for de novo lesions in patients with coronary artery disease. Int. J. Cardiol. 222, 113–118 (2016).
Qian, J. et al. Drug-coated balloon for the treatment of small vessel disease: 9 months of angiographic results and 12 months of clinical outcomes of the PEPCAD China SVD study. Catheter. Cardiovasc. Interv. 101, 33–43 (2023).
Rissanen, T. T. et al. Drug-coated balloon for treatment of de-novo coronary artery lesions in patients with high bleeding risk (DEBUT): a single-blind, randomised, non-inferiority trial. Lancet 394, 230–239 (2019).
Tang, Y. et al. Drug-coated balloon versus drug-eluting stent for small-vessel disease: the RESTORE SVD China randomized trial. JACC Cardiovasc. Interv. 11, 2381–2392 (2018).
Vos, N. S. et al. Paclitaxel-coated balloon angioplasty versus drug-eluting stent in acute myocardial infarction: the REVELATION randomized trial. JACC Cardiovasc. Interv. 12, 1691–1699 (2019).
Yu, X. et al. A non-inferiority, randomized clinical trial comparing paclitaxel-coated balloon versus new-generation drug-eluting stents on angiographic outcomes for coronary de novo lesions. Cardiovasc. Drugs Ther. 36, 655–664 (2022).
Spaulding, C. et al. Comparing a strategy of sirolimus-eluting balloon treatment to drug-eluting stent implantation in de novo coronary lesions in all-comers: design and rationale of the SELUTION DeNovo trial. Am. Heart J. 258, 77–84 (2023).
Greco, A. et al. Sirolimus-coated balloon versus everolimus-eluting stent in de novo coronary artery disease: rationale and design of the TRANSFORM II randomized clinical trial. Catheter. Cardiovasc. Interv. 100, 544–552 (2022).
Kereiakes, D. J., Onuma, Y., Serruys, P. W. & Stone, G. W. Bioresorbable vascular scaffolds for coronary revascularization. Circulation 134, 168–182 (2016).
Mitomo, S. et al. Mid-term clinical outcomes after bailout drug-eluting stenting for suboptimal drug-coated balloon results: insights from a Milan registry. Int. J. Cardiol. 263, 17–23 (2018).
Lee, S. Y. et al. Clinical results of drug-coated balloon treatment in a large-scale multicenter Korean registry study. Korean Circ. J. 52, 444–454 (2022).
Basavarajaiah, S. et al. Use of sirolimus-coated balloon in de novo coronary lesions; long-term clinical outcomes from a multi-center real-world population. Catheter. Cardiovasc. Interv. 104, 1159–1167 (2024).
Coughlan, J. & Kastrati, A. Small coronary vessel size: a great need for a standardized definition. J. Soc. Cardiovasc. Angiogr. Interv. 1, 100428 (2022).
Jeger, R. V. et al. Drug-coated balloons for coronary artery disease: third report of the international DCB consensus group. Cardiovasc. Interv. 13, 1391–1402 (2020).
Latib, A. et al. 3-year follow-up of the Balloon Elution and Late Loss Optimization study (BELLO). JACC Cardiovasc. Interv. 8, 1132–1134 (2015).
Jeger, R. V. et al. Long-term efficacy and safety of drug-coated balloons versus drug-eluting stents for small coronary artery disease (BASKET-SMALL 2): 3-year follow-up of a randomised, non-inferiority trial. Lancet 396, 1504–1510 (2020).
Cortese, B., Testa, G., Rivero, F., Erriquez, A. & Alfonso, F. Long-term outcome of drug-coated balloon vs drug-eluting stent for small coronary vessels: PICCOLETO-II 3-year follow-up. JACC Cardiovasc. Interv. 16, 1054–1061 (2023).
Fezzi, S. et al. Individual patient data meta-analysis of paclitaxel-coated balloons vs. drug-eluting stents for small-vessel coronary artery disease: the ANDROMEDA study. Eur. Heart J. 46, 1586–1599 (2025).
Mauri, L. et al. Effects of stent length and lesion length on coronary restenosis. Am. J. Cardiol. 93, 1340–1346 (2004).
Suh, J. et al. The relationship and threshold of stent length with regard to risk of stent thrombosis after drug-eluting stent implantation. JACC Cardiovasc. Interv. 3, 383–389 (2010).
Sawaya, F. J. et al. Contemporary approach to coronary bifurcation lesion treatment. JACC Cardiovasc. Interv. 9, 1861–1878 (2016).
Burzotta, F. et al. Percutaneous coronary intervention for bifurcation coronary lesions: the 15th consensus document from the European Bifurcation Club. EuroIntervention 16, 1307 (2021).
Gao, X. et al. Drug-coated balloon angioplasty of the side branch during provisional stenting: the multicenter randomized DCB-BIF trial. J. Am. Coll. Cardiol. 85, 1–15 (2025).
Kleber, F. X. et al. Drug eluting balloons as stand alone procedure for coronary bifurcational lesions: results of the randomized multicenter PEPCAD-BIF trial. Clin. Res. Cardiol. 105, 613–621 (2016).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/study/NCT06822322 (2025).
Cortese, B., Buccheri, D., Piraino, D. & Silva-Orrego, P. Drug-coated balloon without stent implantation for chronic total occlusion of coronary arteries: description of a new strategy with an optical coherence tomography assistance. Int. J. Cardiol. 191, 75–76 (2015).
Somsen, Y. B. O. et al. Design and rationale of the drug-coated balloon coronary angioplasty versus stenting for treatment of disease adjacent to a chronic total occlusion (Co-CTO) trial. Am. Heart J. 288, 65–76 (2025).
Scheller, B. et al. Bare metal or drug-eluting stent versus drug-coated balloon in non-ST-elevation myocardial infarction: the randomised PEPCAD NSTEMI trial. EuroIntervention 15, 1527–1533 (2020).
Mangner, N. et al. Safety and efficacy of drug-coated balloons versus drug-eluting stents in acute coronary syndromes: a prespecified analysis of BASKET-SMALL 2. Circ. Cardiovasc. Interv. 15, e011325 (2022).
Her, A.-Y. et al. Vasoconstrictor component of atherothrombotic culprit lesions in ST-segment elevation myocardial infarction. J. Saudi Heart Assoc. 31, 114–120 (2019).
Srikanth, S. & Ambrose, J. A. Pathophysiology of coronary thrombus formation and adverse consequences of thrombus during PCI. Curr. Cardiol. Rev. 8, 168–176 (2012).
Giacoppo, D., Saucedo, J. & Scheller, B. Coronary drug-coated balloons for de novo and in-stent restenosis indications. J. Soc. Cardiovasc. Angiogr. Interv. 2, 100625 (2023).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/study/NCT04814212 (2023).
Gouëffic, Y., Brodmann, M., Deloose, K., Dubosq-Lebaz, M. & Nordanstig, J. Drug-eluting devices for lower limb peripheral arterial disease. EuroIntervention 20, e1136–e1153 (2024).
Zeller, T. et al. Drug-eluting balloon versus standard balloon angioplasty for infrapopliteal arterial revascularization in critical limb ischemia: 12-month results from the IN.PACT DEEP randomized trial. J. Am. Coll. Cardiol. 64, 1568–1576 (2014).
Zeller, T. et al. Paclitaxel-coated balloon in infrapopliteal arteries: 12-month results from the BIOLUX P-II randomized trial (BIOTRONIK’S-first in man study of the Passeo-18 LUX drug releasing PTA balloon catheter vs. the uncoated Passeo-18 PTA balloon catheter in subjects requiring revascularization of infrapopliteal arteries). JACC Cardiovasc. Interv. 8, 1614–1622 (2015).
Cassese, S. et al. Drug-coated balloons for revascularization of infrapopliteal arteries: a meta-analysis of randomized trials. JACC Cardiovasc. Interv. 9, 1072–1080 (2016).
Minar, E. et al. Endovascular brachytherapy for prophylaxis of restenosis after femoropopliteal angioplasty: results of a prospective randomized study. Circulation 102, 2694–2699 (2000).
Capek, P., McLean, G. & Berkowitz, H. Femoropopliteal angioplasty. Factors influencing long-term success. Circulation 83, I70–I80 (1991).
Krishnan, P. et al. Determinants of drug-coated balloon failure in patients undergoing femoropopliteal arterial intervention. J. Am. Coll. Cardiol. 80, 1241–1250 (2022).
Zenunaj, G. et al. Primary drug-coated balloon versus drug-eluting stent for native atherosclerotic femoropopliteal lesions: a systematic review and meta-analysis. Ann. Vasc. Surg. 92, 294–303 (2023).
Lukacs, R. A., Weisshaar, L. I., Tornyos, D. & Komocsi, A. Comparing endovascular approaches in lower extremity artery disease: insights from a network meta-analysis. J. Clin. Med. 13, 1024 (2024).
Weintraub, W. S. The pathophysiology and burden of restenosis. Am. J. Cardiol. 100, S3–S9 (2007).
Ullah, W. et al. Safety and efficacy of drug-coated balloon for peripheral artery revascularization — a systematic review and meta-analysis. Catheter. Cardiovasc. Interv. 99, 1319–1326 (2022).
Jia, X. et al. Acotec drug-coated balloon catheter: randomized, multicenter, controlled clinical study in femoropopliteal arteries: evidence from the AcoArt I trial. JACC Cardiovasc. Interv. 9, 1941–1949 (2016).
Krishnan, P. et al. Stellarex drug-coated balloon for treatment of femoropopliteal disease: twelve-month outcomes from the randomized ILLUMENATE pivotal and pharmacokinetic studies. Circulation 136, 1102–1113 (2017).
Rosenfield, K. et al. Trial of a paclitaxel-coated balloon for femoropopliteal artery disease. N. Engl. J. Med. 373, 145–153 (2015).
Scheinert, D., Schulte, K.-L., Zeller, T., Lammer, J. & Tepe, G. Paclitaxel-releasing balloon in femoropopliteal lesions using a BTHC excipient: twelve-month results from the BIOLUX P-I randomized trial. J. Endovasc. Ther. 22, 14–21 (2015).
Tepe, G. et al. 5-year outcomes of drug-coated balloons for peripheral artery in-stent restenosis, long lesions, and CTOs. JACC Cardiovasc. Interv. 16, 1065–1078 (2023).
Werk, M. et al. Paclitaxel-coated balloons reduce restenosis after femoro-popliteal angioplasty: evidence from the randomized PACIFIER trial. Circ. Cardiovasc. Interv. 5, 831–840 (2012).
Scheinert, D. et al. The LEVANT I (Lutonix paclitaxel-coated balloon for the prevention of femoropopliteal restenosis) trial for femoropopliteal revascularization: first-in-human randomized trial of low-dose drug-coated balloon versus uncoated balloon angioplasty. JACC Cardiovasc. Interv. 7, 10–19 (2014).
Laird, J. A. et al. Long-term clinical effectiveness of a drug-coated balloon for the treatment of femoropopliteal lesions. Circ. Cardiovasc. Interv. 12, e007702 (2019).
Ko, Y. G. et al. Intravascular ultrasound-guided drug-coated balloon angioplasty for femoropopliteal artery disease: a clinical trial. Eur. Heart J. 45, 2839–2847 (2024).
Altin, S. E. et al. Long-term patient outcomes after femoropopliteal peripheral vascular intervention in patients with intermittent claudication. JACC Cardiovasc. Interv. 16, 1668–1678 (2023).
Alyavi, B. & Uzokov, J. Peripheral artery disease in the lower extremities: indications for treatment. E-journal Cardiol. Pract. 16, 1–10 (2018).
Tepe, G. et al. Local delivery of paclitaxel to inhibit restenosis during angioplasty of the leg. N. Engl. J. Med. 358, 689–699 (2008).
Tepe, G. et al. Angioplasty of femoral-popliteal arteries with drug-coated balloons: 5-year follow-up of the THUNDER trial. JACC Cardiovasc. Interv. 8, 102–108 (2015).
Tepe, G. et al. Paclitaxel-coated balloon vs uncoated balloon angioplasty for treatment of in-stent restenosis in the superficial femoral and popliteal arteries: the COPA CABANA trial. J. Endovasc. Ther. 27, 276–286 (2020).
Xu, Y. et al. Long-term safety and efficacy of angioplasty of femoropopliteal artery disease with drug-coated balloons from the AcoArt I trial. J. Vasc. Surg. 74, 756–762.e3 (2021).
Albrecht, T. et al. Two-year clinical outcomes of the CONSEQUENT trial: can femoropopliteal lesions be treated with sustainable clinical results that are economically sound? Cardiovasc. Intervent. Radiol. 41, 1008–1014 (2018).
Shishehbor, M. H. et al. Comparison of drug-coated balloons vs bare-metal stents in patients with femoropopliteal arterial disease. J. Am. Coll. Cardiol. 81, 237–249 (2023).
Bausback, Y. et al. Drug-eluting stent versus drug-coated balloon revascularization in patients with femoropopliteal arterial disease. J. Am. Coll. Cardiol. 73, 667–679 (2019).
Fanelli, F. et al. The “DEBELLUM” — lower limb multilevel treatment with drug eluting balloon — randomized trial: 1-year results. J. Cardiovasc. Surg. 55, 207–216 (2014).
Liistro, F. et al. Drug-eluting balloon in peripheral intervention for below the knee angioplasty evaluation (DEBATE-BTK): a randomized trial in diabetic patients with critical limb ischemia. Circulation 128, 615–621 (2013).
Thanigaimani, S. et al. Network meta-analysis of randomised controlled trials comparing the outcomes of different endovascular revascularisation treatments for infra-inguinal peripheral arterial disease causing chronic limb threatening ischaemia. Eur. J. Vasc. Endovasc. Surg. 68, 246–254 (2024).
Zeller, T. et al. The IN.PACT DEEP clinical drug-coated balloon trial: 5-year outcomes. JACC Cardiovasc. Interv. 13, 431–443 (2020).
Kolodgie, F. D. et al. Comparison of particulate embolization after femoral artery treatment with IN.PACT Admiral versus Lutonix 035 paclitaxel-coated balloons in healthy swine. J. Vasc. Interv. Radiol. 27, 1676–1685.e2 (2016).
Boitet, A. et al. An experimental study of paclitaxel embolisation during drug coated balloon angioplasty. Eur. J. Vasc. Endovasc. Surg. 57, 578–586 (2019).
Mazzolai, L. et al. 2024 ESC Guidelines for the management of peripheral arterial and aortic diseases. Eur. Heart J. 45, 3538–3700 (2024).
Kinstner, C. M. et al. Paclitaxel-eluting balloon versus standard balloon angioplasty in in-stent restenosis of the superficial femoral and proximal popliteal artery: 1-year results of the PACUBA trial. JACC Cardiovasc. Interv. 9, 1386–1392 (2016).
Krankenberg, H. et al. Drug-coated balloon versus standard balloon for superficial femoral artery in-stent restenosis: the randomized femoral artery in-stent restenosis (FAIR) trial. Circulation 132, 2230–2236 (2015).
Liao, C. J., Song, S. H., Li, T., Zhang, Y. & Zhang, W. D. Randomized controlled trial of orchid drug-coated balloon versus standard percutaneous transluminal angioplasty for treatment of femoropopliteal artery in-stent restenosis. Int. Angiol. 38, 365–371 (2019).
Ott, I. et al. ISAR-PEBIS (paclitaxel-eluting balloon versus conventional balloon angioplasty for in-stent restenosis of superficial femoral artery): a randomized trial. J. Am. Heart Assoc. 6, e006321 (2017).
Yu, C. et al. Network meta-analysis of all available regimens based on drug-coated balloon angioplasty and laser atherectomy for femoropopliteal in-stent restenosis. J. Endovasc. Ther. 31, 390–399 (2024).
Cassese, S. et al. Drug-coated balloon angioplasty for femoropopliteal in-stent restenosis. Circ. Cardiovasc. Interv. 11, e007055 (2018).
Voll, F. et al. Diabetes mellitus and femoropopliteal in-stent restenosis. Vasa 51, 247–255 (2022).
Angel de Gregorio, M. et al. Drug-coated balloon for the treatment of long-segment femoropopliteal artery disease: pooled analysis from the BIOLUX P-III SPAIN and BIOLUX P-III all-comers registry long lesion subgroup. J. Vasc. Interv. Radiol. 34, 1707–1715.e7 (2023).
Brodmann, M. et al. Long-term outcomes of the 150 mm drug-coated balloon cohort from the IN.PACT Global Study. Cardiovasc. Intervent. Radiol. 45, 1276–1287 (2022).
Micari, A. et al. 2-year results of paclitaxel-coated balloons for long femoropopliteal artery disease: evidence from the SFA-Long study. JACC Cardiovasc. Interv. 10, 728–734 (2017).
Yu, X. et al. One-year outcomes of drug-coated balloon treatment for long femoropopliteal lesions: a multicentre cohort and real-world study. BMC Cardiovasc. Disord. 21, 326 (2021).
Sun, G. et al. Comparison of drug-coated balloon angioplasty versus uncoated balloon angioplasty in treatment of total occlusions with severe femoropopliteal lesions: an additional analysis from the AcoArt I study. Vascular 29, 340–349 (2021).
Neumann, F. J. et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur. Heart J. 40, 87–165 (2019).
Elgendy, I. Y. et al. Drug-eluting balloons versus everolimus-eluting stents for in-stent restenosis: a meta-analysis of randomized trials. Cardiovasc. Revasc. Med. 20, 612–618 (2019).
Lawton, J. S. et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 145, e18–e114 (2022).
Nordanstig, J. et al. Editor’s choice — European Society for Vascular Surgery (ESVS) 2024 clinical practice guidelines on the management of asymptomatic lower limb peripheral arterial disease and intermittent claudication. Eur. J. Vasc. Endovasc. Surg. 67, 9–96 (2024).
Feldman, D. N. et al. SCAI consensus guidelines for device selection in femoral-popliteal arterial interventions. Catheter. Cardiovasc. Interv. 92, 124–140 (2018).
Feldman, D. N. et al. SCAI guidelines on device selection in aorto-iliac arterial interventions. Catheter. Cardiovasc. Interv. 96, 915 (2020).
Conte, M. S. et al. Global vascular guidelines on the management of chronic limb-threatening ischemia. Eur. J. Vasc. Endovasc. Surg. 58, S1–S109.e33 (2019).
Korosoglou, G. et al. Global algorithm for the endovascular treatment of chronic femoropopliteal lesions. JACC Cardiovasc. Interv. 18, 545–557 (2025).
Sato, T. et al. The effect of the debulking by excimer laser coronary angioplasty on long-term outcome compared with drug-coating balloon: insights from optical frequency domain imaging analysis. Lasers Med. Sci. 35, 403–412 (2020).
Liao, C. J., Song, S. H., Li, T. & Zhang, Y. Orchid drug-coated balloon versus standard percutaneous transluminal angioplasty for the treatment of femoropopliteal artery disease: 12-month result of the randomized controlled trial. Vascular 30, 448–454 (2022).
Author information
Authors and Affiliations
Contributions
The authors contributed substantially to all aspects of the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.A.B. does not accept direct or personal payments from the medical device or pharmaceutical industry and reports research funding to his institution from Abbott Vascular, Biosensors, Boston Scientific and Translumina with no effect on his personal remuneration. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Fernando Alfonso, Antonio Colombo, Bernardo Cortese and Bruno Scheller for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
To identify the scientific evidence on the use of DCBs in the setting of CAD and PAD, we performed systematic literature searches. The search strategy is provided in Supplementary Table 6. Literature searches were performed in PubMed (final search on 20 January 2025) to identify clinical trials investigating DCB angioplasty in patients with CAD (Supplementary Table 7) or PAD (Supplementary Table 8). We prioritized evidence from RCTs. The reference lists from the identified papers were also reviewed to identify additional studies.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Durand, R., O’Callaghan, D., Coughlan, J.J. et al. Drug-coated balloon angioplasty for coronary and peripheral artery disease: latest evidence and clinical indications. Nat Rev Cardiol (2026). https://doi.org/10.1038/s41569-026-01262-2
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41569-026-01262-2