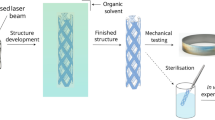
Abstract
Over the past two decades, stents have revolutionized the treatment of cardiovascular diseases, particularly coronary artery and valvular heart disease. In this Review, we evaluate the clinical challenges associated with these diseases, as well as the manufacturing strategies for both coronary and heart valve stents, with a focus on the emerging role of 3D printing. Specifically, we assess the advantages and limitations of clinically available metallic and polymeric stents for coronary artery disease and provide an overview of the metallic and biodegradable polymeric stents developed to treat valvular heart disease. Furthermore, we explore the capabilities of key 3D-printing methods for stent fabrication, highlighting their strengths and drawbacks, and discuss the regulatory considerations that govern the clinical translation of 3D printing-based coronary and valvular stents. Taken together, 3D-printing technologies offer new opportunities to customize geometry, control stent degradation and improve stent implantation approaches for patients with complex anatomies. By merging innovations in material science, manufacturing strategies and bioengineering, 3D-printing technology has a major role in the next generation of personalized and multifunctional cardiovascular stents.
Key points
-
The majority of clinically available coronary artery stents are predominantly manufactured using metals, but these metallic stents are associated with a high risk of restenosis and poor long-term biocompatibility.
-
Newer biodegradable metallic stents aim to reduce the long-term risks of late thrombosis, chronic inflammation and impaired vessel remodelling while promoting natural artery healing.
-
Emerging polymeric stents offer lower profiles and greater flexibility but require improvement in durability and mechanical properties.
-
Both metallic and biodegradable polymeric stents have been explored for the treatment of valvular heart disease, each offering unique benefits, such as high radial strength and proven durability for metallic stents and biocompatibility and gradual resorption for biodegradable polymeric stents, and face distinct challenges, including limited flexibility and potential calcification in metallic stents and insufficient mechanical strength and complex degradation control in polymeric stents.
-
3D-printing technology introduces precision and customization for stent fabrication, potentially surpassing traditional methods such as laser cutting.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Simon-Yarza, T., Bataille, I. & Letourneur, D. Cardiovascular bio-engineering: current state of the art. J. Cardiovasc. Transl. Res. 10, 180–193 (2017).
Lee, J. H., Kim, E. D., Jun, E. J., Yoo, H. S. & Lee, J. W. Analysis of trends and prospects regarding stents for human blood vessels. Biomater. Res. 22, 8 (2018).
Bhatia, S. K. in Biomaterials for Clinical Applications 23–49 (Springer, 2010).
Malakar, A. K. et al. A review on coronary artery disease, its risk factors, and therapeutics. J. Cell. Physiol. 234, 16812–16823 (2019).
Abubakar, M. et al. Advancements in percutaneous coronary intervention techniques: a comprehensive literature review of mixed studies and practice guidelines. Cureus 15, e41311 (2023).
Changal, K. H. et al. Drug-eluting stents versus bare-metal stents in large coronary artery revascularization: systematic review and meta-analysis. Cardiovasc. Revasc. Med. 23, 42–49 (2021).
Korei, N., Solouk, A., Nazarpak, M. H. & Nouri, A. A review on design characteristics and fabrication methods of metallic cardiovascular stents. Mater. Today Commun. 31, 103467 (2022).
Philip, F. et al. Stent thrombosis with second-generation drug-eluting stents compared with bare-metal stents: network meta-analysis of primary percutaneous coronary intervention trials in ST-segment–elevation myocardial infarction. Circ. Cardiovasc. Interv. 7, 49–61 (2014).
Varenhorst, C. et al. Stent thrombosis rates the first year and beyond with new-and old-generation drug-eluting stents compared to bare metal stents. Clin. Res. Cardiol. 107, 816–823 (2018).
Rodriguez-Granillo, A., Rubilar, B., Rodriguez-Granillo, G. & Rodriguez, A. E. Advantages and disadvantages of biodegradable platforms in drug eluting stents. World J. Cardiol. 3, 84 (2011).
Hu, T., Yang, C., Lin, S., Yu, Q. & Wang, G. Biodegradable stents for coronary artery disease treatment: Recent advances and future perspectives. Mater. Sci. Eng. C 91, 163–178 (2018).
Wang, Z., Li, N., Li, R., Li, Y. & Ruan, L. Biodegradable intestinal stents: a review. Prog. Nat. Sci. Mater. Int. 24, 423–432 (2014).
Ormiston, J. A. & Serruys, P. W. Bioabsorbable coronary stents. Circ. Cardiovasc. Interv. 2, 255–260 (2009).
Cabrera, M. S. et al. Computationally designed 3D printed self-expandable polymer stents with biodegradation capacity for minimally invasive heart valve implantation: a proof-of-concept study. 3D Print. Addit. Manuf. 4, 19–29 (2017).
Lisy, M. et al. Allograft heart valves: current aspects and future applications. Biopreserv. Biobank. 15, 148–157 (2017).
Walter, E. D., De By, T., Meyer, R. & Hetzer, R. The future of heart valve banking and of homografts: perspective from the Deutsches Herzzentrum Berlin. HSR Proc. Intensive Care Cardiovasc. Anesth. 4, 97 (2012).
Fiedler, A. G. & Tolis, G. Surgical treatment of valvular heart disease: overview of mechanical and tissue prostheses, advantages, disadvantages, and implications for clinical use. Curr. Treat. Options Cardiovasc. Med. 20, 7 (2018).
Head, S. J., Çelik, M. & Kappetein, A. P. Mechanical versus bioprosthetic aortic valve replacement. Eur. Heart J. 38, 2183–2191 (2017).
Overtchouk, P., Prendergast, B. & Modine, T. Why should we extend transcatheter aortic valve implantation to low-risk patients? A comprehensive review. Arch. Cardiovasc. Dis. 112, 354–362 (2019).
Shen, Y. et al. Development of biodegradable polymeric stents for the treatment of cardiovascular diseases. Biomolecules 12, 1245 (2022).
Malisz, K. & Świeczko-Żurek, B. Vascular stents — materials and manufacturing technologies. Eng. Biomaterials 166, 22–28 (2023).
Li, Y. et al. Additive manufacturing of vascular stents. Acta Biomaterialia 167, 16–37 (2023).
Zheng, Y. & Yang, H. in Metallic Biomaterials Processing and Medical Device Manufacturing (ed Wen, C.) 317–340 (Elsevier, 2020).
Li, Y. et al. Optimizing structural design on biodegradable magnesium alloy vascular stent for reducing strut thickness and raising radial strength. Mater. Des. 220, 110843 (2022).
Nguyen, D. T., Smith, A. F. & Jiménez, J. M. Stent strut streamlining and thickness reduction promote endothelialization. J. R. Soc. Interface 18, 20210023 (2021).
Sullivan, T. M. et al. Effect of endovascular stent strut geometry on vascular injury, myointimal hyperplasia, and restenosis. J. Vasc. Surg. 36, 143–149 (2002).
Foin, N. et al. Impact of stent strut design in metallic stents and biodegradable scaffolds. Int. J. Cardiol. 177, 800–808 (2014).
Pan, C., Han, Y. & Lu, J. Structural design of vascular stents: a review. Micromachines 12, 770 (2021).
Wholey, M. H. & Finol, E. A. Designing the ideal stent. Endovasc. Today 6, 25–34 (2007).
De Santis, G. et al. A computational study of the hemodynamic impact of open- versus closed-cell stent design in carotid artery stenting. Artif. Organs 37, E96–E106 (2013).
Vanaei, S. et al. Manufacturing, processing, and characterization of self-expanding metallic stents: a comprehensive review. Bioengineering 11, 983 (2024).
Colombo, A. et al. Advancements in coronary bifurcation stenting techniques: insights from computational and bench testing studies. Int. J. Numer. Methods Biomed. Eng. 41, e70000 (2025).
Watson, T., Webster, M. W., Ormiston, J. A., Ruygrok, P. N. & Stewart, J. T. Long and short of optimal stent design. Open Heart 4, e000680 (2017).
Ormiston, J. A., Webber, B. & Webster, M. W. Stent longitudinal integrity: bench insights into a clinical problem. JACC Cardiovasc. Interv. 4, 1310–1317 (2011).
Sukmana, I., Chyanegoro, A. G. & Sugiri, A. Biomedical material for stent application: current status and future challenges. J. Appl. Sci. Eng. Technol. https://doi.org/10.47355/aset.v1i2.18 (2021).
Sigwart, U. et al. Prevention of coronary restenosis by stenting. Eur. Heart J. 9, 31–37 (1988).
Hermawan, H. & Mantovani, D. Process of prototyping coronary stents from biodegradable Fe–Mn alloys. Acta Biomaterialia 9, 8585–8592 (2013).
Beshchasna, N. et al. Recent advances in manufacturing innovative stents. Pharmaceutics 12, 349 (2020).
Yue, R. et al. In vitro cytocompatibility, hemocompatibility and antibacterial properties of biodegradable Zn-Cu-Fe alloys for cardiovascular stents applications. Mater. Sci. Eng. C 113, 111007 (2020).
Saraf, A. & Yadav, S. in Functionalised Cardiovascular Stents (eds Wall, J. G., Podbielska, H. & Wawrzyńska, M.) 27–44 (Elsevier, 2018).
Scafa Udriște, A., Niculescu, A.-G., Grumezescu, A. M. & Bădilă, E. Cardiovascular stents: a review of past, current, and emerging devices. Materials 14, 2498 (2021).
Kang, M.-K., Heo, S.-H. & Yoon, J.-K. In-stent re-endothelialization strategies: cells, extracellular matrix, and extracellular vesicles. Tissue Eng. B Rev. 31, 317–330 (2025).
Erne, P., Schier, M. & Resink, T. J. The road to bioabsorbable stents: reaching clinical reality? Cardiovasc. Intervent. Radiol. 29, 11–16 (2006).
Kibos, A., Campeanu, A. & Tintoiu, I. Pathophysiology of coronary artery in-stent restenosis. Acute Card. Care 9, 111–119 (2007).
Buccheri, D., Piraino, D., Andolina, G. & Cortese, B. Understanding and managing in-stent restenosis: a review of clinical data, from pathogenesis to treatment. J. Thorac. Dis. 8, E1150 (2016).
Babapulle, M. N. & Eisenberg, M. J. Coated stents for the prevention of restenosis: part I. Circulation 106, 2734–2740 (2002).
Hong, S.-J. & Hong, M.-K. Drug-eluting stents for the treatment of coronary artery disease: a review of recent advances. Expert Opin. Drug Deliv. 19, 269–280 (2022).
Kuramitsu, S. et al. Drug-eluting stent thrombosis: current and future perspectives. Cardiovasc. Interv. Ther. 36, 158–168 (2021).
Tada, T. et al. Comparative efficacy of 2 zotarolimus-eluting stent generations: resolute versus endeavor stents in patients with coronary artery disease. Am. Heart J. 165, 80–86 (2013).
Mallory, A., Giannopoulos, S., Lee, P., Kokkinidis, D. G. & Armstrong, E. J. Covered stents for endovascular treatment of aortoiliac occlusive disease: a systematic review and meta-analysis. Vasc. Endovasc. Surg. 55, 560–570 (2021).
Udriște, A. S., Burdușel, A. C., Niculescu, A.-G., Rădulescu, M. & Grumezescu, A. M. Coatings for cardiovascular stents — an up-to-date review. Int. J. Mol. Sci. 25, 1078 (2024).
Kilic, I. D. et al. Coronary covered stents. EuroIntervention 12, 1288–1295 (2016).
Raikar, A. S., Priya, S., Bhilegaonkar, S. P., Somnache, S. N. & Kalaskar, D. M. Surface engineering of bioactive coatings for improved stent hemocompatibility: a comprehensive review. Materials 16, 6940 (2023).
Wiyono, A. V. & Ardinal, A. P. Revolutionizing cardiovascular frontiers: a dive into cutting-edge innovations in coronary stent technology. Cardiol. Rev. 10, 1097 (2024).
Brami, P. et al. Evolution of coronary stent platforms: a brief overview of currently used drug-eluting stents. J. Clin. Med. 12, 6711 (2023).
Leone, A. et al. Ultrathin struts drug-eluting stents: a state-of-the-art review. J. Personalized Med. 12, 1378 (2022).
Stevenson, C. L., Santini, J. T. Jr & Langer, R. Reservoir-based drug delivery systems utilizing microtechnology. Adv. Drug Deliv. Rev. 64, 1590–1602 (2012).
Hsiao, H.-M., Chiu, Y.-H., Wu, T.-Y., Shen, J.-K. & Lee, T.-Y. Effects of through-hole drug reservoirs on key clinical attributes for drug-eluting depot stent. Med. Eng. Phys. 35, 884–897 (2013).
Piccolo, R., Franzone, A. & Windecker, S. From bare metal to barely anything: an update on coronary stenting. Heart 104, 533–540 (2018).
Piccolo, R. & Windecker, S. in The Interventional Cardiology Training Manual (eds Myat, A., Clarke, S., Curzen, N., Windecker, S. & Gurbel, P. A.) 137–159 (Springer, 2018).
Carrié, D. The use of the Cre8 Stent in patients with diabetes mellitus. Interv. Cardiol. 11, 47 (2016).
Gibbs, O. et al. Clinical outcomes of Cre8 coronary stent in complex percutaneous coronary intervention. Heart Lung Circ. 27, S442–S443 (2018).
Sinan, U. Y., Serin, E., Keskin-Meric, B. & Arat-Ozkan, A. Cre8 drug eluting stent performance in daily cardiology practice. Rev. Cardiovasc. Med. 24, 53 (2023).
Chiarito, M. et al. Safety and efficacy of polymer-free drug-eluting stents: amphilimus-eluting Cre8 versus biolimus-eluting BioFreedom stents. Circ. Cardiovasc. Interv. 12, e007311 (2019).
Mauler-Wittwer, S. & Garot, P. The biolimus A9-coated BioFreedom™ stent: from clinical efficacy to real-world evidence. Future Cardiol. 17, 239–255 (2021).
Costa, R. A. et al. Polymer-free biolimus A9-coated stents in the treatment of de novo coronary lesions: 4-and 12-month angiographic follow-up and final 5-year clinical outcomes of the prospective, multicenter BioFreedom FIM clinical trial. JACC Cardiovasc. Interv. 9, 51–64 (2016).
Maurina, M. et al. Randomized clinical trial of abluminus DES+ sirolimus-eluting stent versus everolimus-eluting DES for percutaneous coronary intervention in patients with diabetes mellitus: an optical coherence tomography study. Catheter. Cardiovasc. Interv. 102, 1020–1033 (2023).
Goel, R., Chandiramani, R. & Mehran, R. Abluminus DES+ for the treatment of coronary artery disease in patients with diabetes mellitus. Future Cardiol. 16, 613–623 (2020).
Grenacher, L. et al. In vitro comparison of self-expanding versus balloon-expandable stents in a human ex vivo model. Cardiovasc. Intervent. Radiol. 29, 249–254 (2006).
Vishnu, J. et al. Balloon expandable coronary stent materials: a systematic review focused on clinical success. In Vitro Model. 1, 151–175 (2022).
Pyxaras, S. A., Schmitz, T. & Naber, C. K. The STENTYS self-apposing® stent. EuroIntervention 11, V147–V148 (2015).
Pellegrini, D. & Cortese, B. Focus on STENTYS® Xposition S Self-Apposing® stent: a review of available literature. Future Cardiol. 15, 145–159 (2019).
van Geuns, R. J. et al. STENTYS self-apposing sirolimus-eluting stent in ST-segment elevation myocardial infarction: results from the randomised APPOSITION IV trial. EuroIntervention 11, e1267–e1274 (2016).
Grundeken, M. J. et al. APPOSITION V: STENTYS coronary stent system clinical trial in subjects with ST-segment elevation myocardial infarction — rationale and design. Am. Heart J. 168, 652–660 (2014).
Agostoni, P. & Verheye, S. Step-by-step StentBoost-guided small vessel stenting using the self-expandable sparrow stent-in-wire. Catheter. Cardiovasc. Interv. 73, 78–83 (2009).
Jilaihawi, H., Farah, B. & Laborde, J.-C. The use of self-expanding stents in coronary bifurcations and beyond: a paradigm revisited. EuroIntervention 4, 669–675 (2009).
Abizaid, A. C., Costa, J. Jr., Whitbourn, R. J. & Chang, J. C. The CardioMind™ coronary stent delivery system: stent delivery on a. 014 guidewire platform. EuroIntervention 3, 154–157 (2007).
Abizaid, A. C. et al. TCT- 226 CARE II 8 month follow-up results with the CardioMind 0.014 Sparrow sirolimus-eluting nitinol stent system. JACC 56 (Suppl. 13), B53 (2010).
Kume, T. et al. Intravascular ultrasound analysis of small vessel lesions treated with the sparrow coronary stent system: results of the CARE II trial. Catheter. Cardiovasc. Interv. 83, 19–24 (2014).
Verheye, S. et al. Twelve-month clinical and imaging outcomes of the uncaging coronary DynamX bioadaptor system. EuroIntervention 16, e974–e981 (2020).
Webster, M. et al. DynamX bioadaptor, a novel uncaging platform for percutaneous coronary artery revascularisation: 6 month clinical and imaging results. Heart Lung Circ. 30, S300–S301 (2021).
Verheye, S. et al. 24-Month clinical follow-up and mechanistic insights from intravascular imaging following coronary implantation of the novel DynamX bioadaptor platform. Cardiovasc. Revasc. Med. 46, 106–112 (2023).
Verheye, S. et al. Final 36-month outcomes from the multicenter DynamX study evaluating a novel thin-strut novolimus-eluting coronary bioadaptor system and supporting preclinical data. Rev. Cardiovasc. Med. 24, 221 (2023).
Liu, Y. et al. Fundamental theory of biodegradable metals — definition, criteria, and design. Adv. Funct. Mater. 29, 1805402 (2019).
Peuster, M. et al. A novel approach to temporary stenting: degradable cardiovascular stents produced from corrodible metal — results 6–18 months after implantation into New Zealand white rabbits. Heart 86, 563–569 (2001).
Riaz, U., Shabib, I. & Haider, W. The current trends of Mg alloys in biomedical applications — a review. J. Biomed. Mater. Res. B Appl. Biomater. 107, 1970–1996 (2019).
Aljihmani, L. et al. Magnesium-based bioresorbable stent materials: review of reviews. J. Bio Tribo Corrosion 5, 26 (2019).
Heublein, B. et al. Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology? Heart 89, 651–656 (2003).
Bowen, P. K., Drelich, A., Drelich, J. & Goldman, J. Rates of in vivo (arterial) and in vitro biocorrosion for pure magnesium. J. Biomed. Mater. Res. A 103, 341–349 (2015).
Uddin, M., Hall, C. & Murphy, P. Surface treatments for controlling corrosion rate of biodegradable Mg and Mg-based alloy implants. Sci. Technol. Adv. Mater. 16, 053501 (2015).
Song, R. et al. Current development of biodegradable polymeric materials for biomedical applications. Drug Des. Devel. Ther. 12, 3117–3145 (2018).
Cheng, J. et al. Experimental investigation on the properties of poly (L-lactic acid) vascular stent after accelerated in vitro degradation. J. Appl. Polymer Sci. 140, (2022).
Grabow, N., Martin, D. P., Schmitz, K. P. & Sternberg, K. Absorbable polymer stent technologies for vascular regeneration. J. Chem. Technol. Biotechnol. 85, 744–751 (2010).
Smit, N. W. et al. Recombinant human collagen-based microspheres mitigate cardiac conduction slowing induced by adipose tissue-derived stromal cells. PLoS One 12, e0183481 (2017).
Piskin, E. Biodegradable polymers as biomaterials. J. Biomater. Sci. Polym. Ed. 6, 775–795 (1995).
Stack, R. et al. Interventional cardiac catheterization at Duke Medical Center. Am. J. Cardiol. 62, 3F–24F (1988).
Soares, J. S. & Moore, J. E. Biomechanical challenges to polymeric biodegradable stents. Ann. Biomed. Eng. 44, 560–579 (2016).
Berglund, J., Guo, Y. & Wilcox, J. N. Challenges related to development of bioabsorbable vascular stents. EuroIntervention 5, F72–F79 (2009).
Im, S. H. et al. Current status and future direction of metallic and polymeric materials for advanced vascular stents. Prog. Mater. Sci. 126, 100922 (2022).
Ahadi, F., Azadi, M., Biglari, M., Bodaghi, M. & Khaleghian, A. Evaluation of coronary stents: a review of types, materials, processing techniques, design, and problems. Heliyon 9, e13575 (2023).
Wu, W. et al. Three dimensional reconstruction of coronary artery stents from optical coherence tomography: experimental validation and clinical feasibility. Sci. Rep. 11, 12252 (2021).
Kurogi, K., Ishii, M., Yamamoto, N., Yamanaga, K. & Tsujita, K. Optical coherence tomography-guided percutaneous coronary intervention: a review of current clinical applications. Cardiovasc. Interv. Ther. 36, 169–177 (2021).
Commandeur, S., van Beusekom, H. M. & van der Giessen, W. J. Polymers, drug release, and drug-eluting stents. J. Interv. Cardiol. 19, 500–506 (2006).
Colombo, A. & Iakovou, I. Drug-eluting stents: the new gold standard for percutaneous coronary revascularisation. Eur. Heart J. 25, 895–897 (2004).
Kumar, M. et al. Comparison of drug-coated balloon to drug eluting stent in patients with in-stent restenosis: a systematic review and meta-analysis. Am. J. Cardiol. 227, 57–64 (2024).
Yang, F.-F. et al. Five-year outcomes of bioresorbable stent therapy for coronary heart disease: a systematic review and meta-analysis of randomized controlled trials. Rev. Cardiovasc. Med. 25, 238 (2024).
Fioretta, E. S. et al. Next-generation tissue-engineered heart valves with repair, remodelling and regeneration capacity. Nat. Rev. Cardiol. 18, 92–116 (2021).
Oveissi, F., Naficy, S., Lee, A., Winlaw, D. & Dehghani, F. Materials and manufacturing perspectives in engineering heart valves: a review. Mater. Today Bio 5, 100038 (2020).
Santangelo, G. et al. An update on new generation transcatheter aortic valves and delivery systems. J. Clin. Med. 11, 499 (2022).
Steblovnik, K. & Bunc, M. Technical aspects and development of transcatheter aortic valve implantation. J. Cardiovasc. Dev. Dis. 9, 282 (2022).
Pasta, S. & Gandolfo, C. Computational analysis of self-expanding and balloon-expandable transcatheter heart valves. Biomechanics 1, 43–52 (2021).
Li, R. L. et al. Mechanical considerations for polymeric heart valve development: biomechanics, materials, design and manufacturing. Biomaterials 225, 119493 (2019).
Galliazzo, S., Pelitti, V., Campiotti, L., Poli, D. & Squizzato, A. Direct oral anticoagulants in patients with a left-sided bioprosthetic heart valve: a systematic review and meta-analysis. Intern. Emerg. Med. 18, 535–547 (2023).
Bernacca, G., Mackay, T., Wilkinson, R. & Wheatley, D. Calcification and fatigue failure in a polyurethane heart valve. Biomaterials 16, 279–285 (1995).
Bernacca, G., Mackay, T., Wilkinson, R. & Wheatley, D. Polyurethane heart valves: fatigue failure, calcification, and polyurethane structure. J. Biomed. Mater. Res. 34, 371–379 (1997).
Rezvova, M. A., Klyshnikov, K. Y., Gritskevich, A. A. & Ovcharenko, E. A. Polymeric heart valves will displace mechanical and tissue heart valves: a new era for the medical devices. Int. J. Mol. Sci. 24, 3963 (2023).
Bui, H. T. et al. Transcatheter heart valves: a biomaterials perspective. Adv. Healthc. Mater. 10, 2100115 (2021).
Ozturk, S., Ayanoğlu, F. B., Parmaksiz, M., Elçin, A. E. & Elçin, Y. M. in Handbook of Biomaterials Biocompatibility (ed Mozafari, M.) 219–250 (Elsevier, 2020).
Snyder, Y. & Jana, S. Strategies for development of synthetic heart valve tissue engineering scaffolds. Prog. Mater. Sci. 139, 101173 (2023).
Motta, S. E. et al. Combining cell technologies with biomimetic tissue engineering applications: a new paradigm for translational cardiovascular therapies. Stem Cell Transl. Med. 12, 72–82 (2023).
Kathuria, Y. The potential of biocompatible metallic stents and preventing restenosis. Mater. Sci. Eng. A 417, 40–48 (2006).
Muhammad, N. Laser Micromachining of Coronary Stents for Medical Applications. Thesis, Univ. Manchester (2012).
Stoeckel, D., Bonsignore, C. & Duda, S. A survey of stent designs. Minim. Invasive Ther. Allied Technol. 11, 137–147 (2002).
Plaine, A. H., da Silva, M. R. & Bolfarini, C. Tailoring the microstructure and mechanical properties of metastable Ti–29Nb–13Ta-4.6 Zr alloy for self-expansible stent applications. J. Alloys Compd 800, 35–40 (2019).
Schinhammer, M., Hänzi, A. C., Löffler, J. F. & Uggowitzer, P. J. Design strategy for biodegradable Fe-based alloys for medical applications. Acta Biomaterialia 6, 1705–1713 (2010).
Obayi, C. S. et al. Effect of grain sizes on mechanical properties and biodegradation behavior of pure iron for cardiovascular stent application. Biomatter 6, e959874 (2016).
Moravej, M. & Mantovani, D. Biodegradable metals for cardiovascular stent application: interests and new opportunities. Int. J. Mol. Sci. 12, 4250–4270 (2011).
Schmidt, W. et al. Revisiting SFA stent technology: an updated overview on mechanical stent performance. Biomed. Tech. 68, 523–535 (2023).
Nazneen, F. et al. Surface chemical and physical modification in stent technology for the treatment of coronary artery disease. J. Biomed. Mater. Res. B Appl. Biomater. 100, 1989–2014 (2012).
Polanec, B., Kramberger, J. & Glodež, S. A review of production technologies and materials for manufacturing of cardiovascular stents. Adv. Prod. Eng. Manag. 15, 390–402 (2020).
Toong, D. W. Y. et al. Bioresorbable metals in cardiovascular stents: material insights and progress. Materialia 12, 100727 (2020).
Lee, M. S. et al. Stent fracture associated with drug-eluting stents: clinical characteristics and implications. Catheter. Cardiovasc. Interv. 69, 387–394 (2007).
Lee, S.-H. et al. Frequency of stent fracture as a cause of coronary restenosis after sirolimus-eluting stent implantation. Am. J. Cardiol. 100, 627–630 (2007).
Umeda, H. et al. Frequency, predictors and outcome of stent fracture after sirolimus-eluting stent implantation. Int. J. Cardiol. 133, 321–326 (2009).
Guerra, A. J. & Ciurana, J. Stent’s manufacturing field: past, present, and future prospects. Angiography 73817, 41–60 (2019).
Chen, L.-Y., Liang, S.-X., Liu, Y. & Zhang, L.-C. Additive manufacturing of metallic lattice structures: unconstrained design, accurate fabrication, fascinated performances, and challenges. Mater. Sci. Engineering R Rep. 146, 100648 (2021).
Zhou, L. et al. Additive manufacturing: a comprehensive review. Sensors 24, 2668 (2024).
Dilberoglu, U. M., Gharehpapagh, B., Yaman, U. & Dolen, M. Current trends and research opportunities in hybrid additive manufacturing. Int. J. Adv. Manuf. Technol. 113, 623–648 (2021).
Gu, D. et al. Material-structure-performance integrated laser-metal additive manufacturing. Science 372, eabg1487 (2021).
Tofail, S. A. et al. Additive manufacturing: scientific and technological challenges, market uptake and opportunities. Mater. Today 21, 22–37 (2018).
VanKoevering, K. K., Zopf, D. A. & Hollister, S. J. Tissue engineering and 3-dimensional modeling for facial reconstruction. Facial Plastic Surg. Clin. 27, 151–161 (2019).
Du, X. et al. 3D printing of pearl/CaSO4 composite scaffolds for bone regeneration. J. Mater. Chem. B 6, 499–509 (2018).
Martin, D. & Boyle, F. J. Computational structural modelling of coronary stent deployment: a review. Computer Methods Biomech. Biomed. Eng. 14, 331–348 (2011).
Antonini, L., Mandelli, L., Berti, F., Pennati, G. & Petrini, L. Validation of the computational model of a coronary stent: a fundamental step towards in silico trials. J. Mech. Behav. Biomed. Mater. 122, 104644 (2021).
Antonini, L. et al. A computational workflow for modeling complex patient-specific coronary stenting cases. Computer Methods Prog. Biomed. 259, 108527 (2025).
Moore, S. S., O’Sullivan, K. J. & Verdecchia, F. Shrinking the supply chain for implantable coronary stent devices. Ann. Biomed. Eng. 44, 497–507 (2016).
Nouri, A., Shirvan, A. R., Li, Y. & Wen, C. Additive manufacturing of metallic and polymeric load-bearing biomaterials using laser powder bed fusion: a review. J. Mater. Sci. Technol. 94, 196–215 (2021).
Guerra, A., Roca, A. & de Ciurana, J. A novel 3D additive manufacturing machine to biodegradable stents. Procedia Manuf. 13, 718–723 (2017).
Khorasani, M., Gibson, I., Ghasemi, A. H., Hadavi, E. & Rolfe, B. Laser subtractive and laser powder bed fusion of metals: review of process and production features. Rapid Prototyp. J. 29, 935–958 (2023).
Vellaparambil, R. S. Exploring the Potential of Stent-Grafts Derived from Auxetic Unit Cells for Endovascular Applications. Thesis, Ecole Nat. Supérieure Mines Saint-Etienne (2024).
Yasmin, F., Vafadar, A. & Tolouei-Rad, M. Application of additive manufacturing in the development of polymeric bioresorbable cardiovascular stents: a review. Adv. Mater. Technol. 10, 2400210 (2025).
Salmi, M., Ituarte, I. F., Chekurov, S. & Huotilainen, E. Effect of build orientation in 3D printing production for material extrusion, material jetting, binder jetting, sheet object lamination, vat photopolymerisation, and powder bed fusion. Int. J. Collab. Enterp. 5, 218–231 (2016).
Monzón, M. et al. Functionally graded additive manufacturing to achieve functionality specifications of osteochondral scaffolds. Bio-Design Manuf. 1, 69–75 (2018).
Yap, Y. L. et al. Material jetting additive manufacturing: an experimental study using designed metrological benchmarks. Precis. Eng. 50, 275–285 (2017).
Huang, S. H., Liu, P., Mokasdar, A. & Hou, L. Additive manufacturing and its societal impact: a literature review. Int. J. Adv. Manuf. Technol. 67, 1191–1203 (2013).
Kristiawan, R. B., Imaduddin, F., Ubaidillah, D. A. & Arifin, Z. A review on the fused deposition modeling (FDM) 3D printing: filament processing, materials, and printing parameters. Open Eng. 11, 639–649 (2021).
Chai, R. et al. Oxygen-evolving photosynthetic cyanobacteria for 2D bismuthene radiosensitizer-enhanced cancer radiotherapy. Bioact. Mater. 17, 276–288 (2022).
Zaidi, R., Khan, S. U., Farooqi, I. & Azam, A. Rapid adsorption of Pb (II) and Cr (VI) from aqueous solution by Aluminum hydroxide nanoparticles: equilibrium and kinetic evaluation. Mater. Today Proc. 47, 1430–1437 (2021).
Singh, G. et al. Fused filament fabrication of bioresorbable stent on a rotating mandrel. Procedia CIRP 110, 156–161 (2022).
Reeser, K. & Doiron, A. L. Three-dimensional printing on a rotating cylindrical mandrel: a review of additive-lathe 3D printing technology. 3D Print. Addit. Manuf. 6, 293–307 (2019).
Khalaj, R., Tabriz, A. G., Okereke, M. I. & Douroumis, D. 3D printing advances in the development of stents. Int. J. Pharmaceut. 609, 121153 (2021).
Colell Villaró, D. 3D Printing of Bioresorbable Polymeric Cardiovascular Stents. Thesis, Univ. Politècnica Catalunya (2022).
Sousa, A. M., Amaro, A. M. & Piedade, A. P. 3D printing of polymeric bioresorbable stents: a strategy to improve both cellular compatibility and mechanical properties. Polymers 14, 1099 (2022).
Deshpande, A. A. & Pan, Y. Direct ink writing on a rotating mandrel-additive lathe micro-manufacturing. J. Micro Nano-Manuf. 11, 021002 (2023).
Pagac, M. et al. A review of vat photopolymerization technology: materials, applications, challenges, and future trends of 3D printing. Polymers 13, 598 (2021).
Bosch, A. et al. Analysis of printing and post-curing parameters to enhance physicochemical and biomechanical properties of polymeric stents produced by tubular vat photopolymerization. Prog. Addit. Manuf. 10, 5993–6006 (2025).
Hua, W. et al. 3D printing of biodegradable polymer vascular stents: a review. Chin. J. Mech. Eng. Addit. Manuf. Front. 1, 100020 (2022).
Scoutaris, N. et al. Development and biological evaluation of inkjet printed drug coatings on intravascular stent. Mol. Pharmaceut. 13, 125–133 (2016).
U. S. Food & Drug Administration. FDA Medical Device Classification Database: Coronary Stents (Class III Devices) https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpcd/classification.cfm?id=MAF (accessed 2026).
U. S. Food & Drug Administration. Technical Considerations for Additive Manufactured Medical Devices — Guidance for Industry and Food and Drug Administration Staff https://www.fda.gov/regulatory-information/search-fda-guidance-documents/technical-considerations-additive-manufactured-medical-devices (2017).
ISO. ISO 25539-2:2020 — Cardiovascular implants — Endovascular devices — Part 2: Vascular stents. https://www.iso.org/standard/69835.html (2020).
ASTM International. ASTM F2079 — Standard Test Method for Measuring Intrinsic Elastic Recoil of Balloon-Expandable Stents. https://www.astm.org/f2079.html (2022).
ASTM International. ASTM F2477-24 — Standard Test Methods for In Vitro Pulsatile Durability Testing of Vascular Stents and Endovascular Prostheses. https://store.astm.org/f2477-24.html (2024).
European Union. Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on Medical Devices, Amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009, and Repealing Council Directives 90/385/EEC and 93/42/EEC (Medical Device Regulation) https://eur-lex.europa.eu/eli/reg/2017/745/oj/eng (2017).
Zollikofer, C. et al. Arterial stent placement with use of the Wallstent: midterm results of clinical experience. Radiology 179, 449–456 (1991).
Triller, J., Mahler, F., Do, D. & Thalmann, R. in RöFo-Fortschritte auf dem Gebiet der Röntgenstrahlen und der bildgebenden Verfahren 328–334 (Georg Thieme, 1989).
Duprat, G. Jr, Wright, K., Charnsangavej, C., Wallace, S. & Gianturco, C. Self-expanding metallic stents for small vessels: an experimental evaluation. Radiology 162, 469–472 (1987).
Sigwart, U., Puel, J., Mirkovitch, V., Joffre, F. & Kappenberger, L. Intravascular stents to prevent occlusion and re-stenosis after transluminal angioplasty. N. Engl. J. Med. 316, 701–706 (1987).
de Feyter, P. J., de Jaegere, P. P. & Serruys, P. W. Incidence, predictors, and management of acute coronary occlusion after coronary angioplasty. Am. Heart J. 127, 643–651 (1994).
Fischman, D. L. et al. A randomized comparison of coronary-stent placement and balloon angioplasty in the treatment of coronary artery disease. N. Engl. J. Med. 331, 496–501 (1994).
Palmaz, J. C. The balloon-expandable stent. EuroIntervention 2, 416–421 (2007).
Marquis-Gravel, G. & Schatz, R. A. How coronary stents became a reality: a firsthand account by an innovator in the field. Can. J. Cardiol. 38, S3–S4 (2022).
Garriboli, L. & Jannello, A. M. Italian single-center experiences with SMART® Flex. Endovasc. Today Eur. 4, 73–75 (2016).
Goverde, P., Taeymans, K. & Lauwers, K. The S.M.A.R.T.® Flex vascular stent system solution. Endovasc. Today Eur. 2, 24–27 (2014).
Suzuki, K. et al. Long-term results of the SMART ControlTM stent for superficial femoral artery lesions, J-SMART registry. Circulation J. 75, 939–944 (2011).
Bosiers, M. et al. Nitinol stent implantation in long superficial femoral artery lesions: 12-month results of the DURABILITY I study. J. Endovasc. Ther. 16, 261–269 (2009).
Matsumura, J. S. et al. The United States study for evaluating endovascular treatments of lesions in the superficial femoral artery and proximal popliteal by using the Protégé Everflex nitinol stent system II (DURABILITY II). J. Vasc. Surg. 58, 73–83.e1 (2013).
Rocha-Singh, K. J. et al. A single stent strategy in patients with lifestyle limiting claudication: 3-year results from the Durability II trial. Catheter. Cardiovasc. Interv. 86, 164–170 (2015).
Laird, J. R. et al. Nitinol stent implantation versus balloon angioplasty for lesions in the superficial femoral artery and proximal popliteal artery: twelve-month results from the RESILIENT randomized trial. Circ. Cardiovasc. Interv. 3, 267–276 (2010).
Davaine, J.-M. et al. One-year clinical outcome after primary stenting for Trans-Atlantic Inter-Society Consensus (TASC) C and D femoropopliteal lesions (the STELLA “STEnting Long de L’Artere femorale superficielle” cohort). Eur. J. Vasc. Endovasc. Surg. 44, 432–441 (2012).
Maleckis, K. et al. Nitinol stents in the femoropopliteal artery: a mechanical perspective on material, design, and performance. Ann. Biomed. Eng. 46, 684–704 (2018).
Dake, M. D. et al. Paclitaxel-eluting stents show superiority to balloon angioplasty and bare metal stents in femoropopliteal disease: twelve-month Zilver PTX randomized study results. Circ. Cardiovasc. Interv. 4, 495–504 (2011).
Dake, M. D. et al. Durable clinical effectiveness with paclitaxel-eluting stents in the femoropopliteal artery: 5-year results of the Zilver PTX randomized trial. Circulation 133, 1472–1483 (2016).
Fujihara, M., Utsunomiya, M., Higashimori, A., Yokoi, Y. & Nakamura, M. Outcomes of Zilver PTX stent implantation for the treatment of complex femoropopliteal artery disease. Heart Vessels 31, 152–157 (2016).
Hoseini, F., Bellelli, A., Mizzi, L., Pecoraro, F. & Spaggiari, A. Self-expanding nitinol stents for endovascular peripheral applications: a review. Mater. Today Commun. 41, 111042 (2024).
Worthley, S. G. et al. First-in-human evaluation of a novel polymer-free drug-filled stent: angiographic, IVUS, OCT, and clinical outcomes from the RevElution study. JACC Cardiovasc. Interv. 10, 147–156 (2017).
Nogic, J. et al. Novel bioabsorbable polymer and polymer-free metallic drug-eluting stents. J. Cardiol. 71, 435–443 (2018).
Hamon, M. et al. Clinical and angiographic experience with a third-generation drug-eluting Orsiro stent in the treatment of single de novo coronary artery lesions (BIOFLOW-I): a prospective, first-in-man study. EuroIntervention 8, 1006–1011 (2013).
Windecker, S. et al. Comparison of a novel biodegradable polymer sirolimus-eluting stent with a durable polymer everolimus-eluting stent: results of the randomized BIOFLOW-II trial. Circ. Cardiovasc. Interv. 8, e001441 (2015).
von Birgelen, C. et al. Very thin strut biodegradable polymer everolimus-eluting and sirolimus-eluting stents versus durable polymer zotarolimus-eluting stents in allcomers with coronary artery disease (BIO-RESORT): a three-arm, randomised, non-inferiority trial. Lancet 388, 2607–2617 (2016).
Meredith, I. T. et al. Primary endpoint results of the EVOLVE trial: a randomized evaluation of a novel bioabsorbable polymer-coated, everolimus-eluting coronary stent. J. Am. Coll. Cardiol. 59, 1362–1370 (2012).
Sarno, G. et al. Real-life clinical outcomes with everolimus eluting platinum chromium stent with an abluminal biodegradable polymer in patients from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR). Catheter. Cardiovasc. Interv. 90, 881–887 (2017).
Ananthakrishna, R. et al. Incidence and predictors of target lesion failure in a multiethnic Asian population receiving the SYNERGY coronary stent: a prospective all-comers registry. Catheter. Cardiovasc. Interv. 92, 1097–1103 (2018).
Bennett, J. & Dubois, C. A novel platinum chromium everolimus-eluting stent for the treatment of coronary artery disease. Biologics 7, 149–159 (2013).
Schmehl, J. M., Harder, C., Wendel, H. P., Claussen, C. D. & Tepe, G. Silicon carbide coating of nitinol stents to increase antithrombogenic properties and reduce nickel release. Cardiovasc. Revasc. Med. 9, 255–262 (2008).
Dick, P. et al. Balloon angioplasty versus stenting with nitinol stents in intermediate length superficial femoral artery lesions. Catheter. Cardiovasc. Interv. 74, 1090–1095 (2009).
Burket, M. W., Brodmann, M., Metzger, C., Tan, K. & Jaff, M. R. Twelve-month results of the nitinol astron stent in iliac artery lesions. J. Vasc. Interv. Radiol. 27, 1650–1656.e1 (2016).
Morice, M.-C. et al. A randomized comparison of a sirolimus-eluting stent with a standard stent for coronary revascularization. N. Engl. J. Med. 346, 1773–1780 (2002).
Moses, J. W. et al. Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N. Engl. J. Med. 349, 1315–1323 (2003).
Weisz, G. et al. Two-year outcomes after sirolimus-eluting stent implantation: results from the Sirolimus-Eluting Stent in de Novo Native Coronary Lesions (SIRIUS) trial. J. Am. Coll. Cardiol. 47, 1350–1355 (2006).
Weisz, G. et al. Five-year follow-up after sirolimus-eluting stent implantation: results of the SIRIUS (Sirolimus-Eluting Stent in De-Novo Native Coronary Lesions) trial. J. Am. Coll. Cardiol. 53, 1488–1497 (2009).
Mehilli, J. et al. Randomized trial of paclitaxel-versus sirolimus-eluting stents for treatment of coronary restenosis in sirolimus-eluting stents: the ISAR-DESIRE 2 (Intracoronary Stenting and Angiographic Results: Drug Eluting Stents for In-Stent Restenosis 2) study. J. Am. Coll. Cardiol. 55, 2710–2716 (2010).
Kufner, S. et al. Sirolimus-eluting versus paclitaxel-eluting stents in diabetic and non-diabetic patients within sirolimus-eluting stent restenosis: results from the ISAR-DESIRE 2 trial. Cardiovasc. Revasc. Med. 15, 69–75 (2014).
Farooq, V., Okamura, T., Onuma, Y., Gogas, B. D. & Serruys, P. W. Unravelling the complexities of the coronary bifurcation: is this raising a few eyebrows. EuroIntervention 7, 1133–1141 (2012).
Ranade, S. V., Richard, R. E. & Helmus, M. N. Styrenic block copolymers for biomaterial and drug delivery applications. Acta Biomaterialia 1, 137–144 (2005).
Ranade, S. V. et al. Physical characterization of controlled release of paclitaxel from the TAXUS™ Express2™ drug-eluting stent. J. Biomed. Mater. Res. A 71, 625–634 (2004).
Pinto, D. S. et al. Impact of routine angiographic follow-up on the clinical benefits of paclitaxel-eluting stents: results from the TAXUS-IV trial. J. Am. Coll. Cardiol. 48, 32–36 (2006).
Hermiller, J. B. et al. Outcomes with the polymer-based paclitaxel-eluting TAXUS stent in patients with diabetes mellitus: the TAXUS-IV trial. J. Am. Coll. Cardiol. 45, 1172–1179 (2005).
Somaratne, J. B. & Whitbourn, R. J. TAXUS Element stent system. Interv. Cardiol. 3, 641 (2011).
Muni, N. I. & Gross, T. P. Problems with drug-eluting coronary stents — the FDA perspective. N. Engl. J. Med. 351, 1593–1595 (2004).
Lemos, P. A. & Bienert, I. The Supralimus® sirolimus-eluting stent. Expert Rev. Med. Devices 10, 295–300 (2013).
Seth, A., Chandra, P., Chouhan, N. S. & Thakkar, A. S. A first-in-man study of sirolimus-eluting, biodegradable polymer coated cobalt chromium stent in real life patients. Indian Heart J. 64, 547–552 (2012).
Steigerwald, K. et al. The pre-clinical assessment of rapamycin-eluting, durable polymer-free stent coating concepts. Biomaterials 30, 632–637 (2009).
Wessely, R. et al. Inhibition of neointima formation by a novel drug-eluting stent system that allows for dose-adjustable, multiple, and on-site stent coating. Arterioscler. Thromb. Vasc. Biol. 25, 748–753 (2005).
Hausleiter, J. et al. Prevention of restenosis by a novel drug-eluting stent system with a dose-adjustable, polymer-free, on-site stent coating. Eur. Heart J. 26, 1475–1481 (2005).
Mehilli, J. et al. Randomized trial of a nonpolymer-based rapamycin-eluting stent versus a polymer-based paclitaxel-eluting stent for the reduction of late lumen loss. Circulation 113, 273–279 (2006).
Kumar, A. S. & Hariram, V. Indigenous stents: examining the clinical data on new technologies. Interventional Cardiol. 6, 319 (2014).
Ostojic, M. C. et al. The pharmacokinetics of Biolimus A9 after elution from the BioMatrix II stent in patients with coronary artery disease: the Stealth PK Study. Eur. J. Clin. Pharmacol. 67, 389–398 (2011).
Windecker, S. et al. Biolimus-eluting stent with biodegradable polymer versus sirolimus-eluting stent with durable polymer for coronary revascularisation (LEADERS): a randomised non-inferiority trial. Lancet 372, 1163–1173 (2008).
Serruys, P. W. et al. Improved safety and reduction in stent thrombosis associated with biodegradable polymer-based biolimus-eluting stents versus durable polymer-based sirolimus-eluting stents in patients with coronary artery disease: final 5-year report of the LEADERS (Limus Eluted From A Durable Versus ERodable Stent Coating) randomized, noninferiority trial. JACC Cardiovasc. Interv. 6, 777–789 (2013).
Barlis, P. et al. An optical coherence tomography study of a biodegradable vs. durable polymer-coated limus-eluting stent: a LEADERS trial sub-study. Eur. Heart J. 31, 165–176 (2010).
Ng, J. et al. Over-expansion capacity and stent design model: an update with contemporary DES platforms. Int. J. Cardiol. 221, 171–179 (2016).
Stefanini, G. G. et al. Long-term clinical outcomes of biodegradable polymer biolimus-eluting stents versus durable polymer sirolimus-eluting stents in patients with coronary artery disease (LEADERS): 4 year follow-up of a randomised non-inferiority trial. Lancet 378, 1940–1948 (2011).
Kim, S. et al. Coronary stent fracture complicated multiple aneurysms confirmed by 3-dimensional reconstruction of intravascular-optical coherence tomography in a patient treated with open-cell designed drug-eluting stent. Circulation 129, e24–e27 (2014).
Chevalier, B. et al. Randomised comparison of Nobori, biolimus A9-eluting coronary stent with a Taxus (R), paclitaxel-eluting coronary stent in patients with stenosis in native coronary arteries: the Nobori 1 trial. EuroIntervention 2, 426–434 (2007).
Vlachojannis, G. J. et al. Biodegradable polymer biolimus-eluting stents versus durable polymer everolimus-eluting stents in patients with coronary artery disease: final 5-year report from the COMPARE II trial (abluminal biodegradable polymer biolimus-eluting stent versus durable polymer everolimus-eluting stent). Cardiovasc. Interv. 10, 1215–1221 (2017).
Granada, J. F. et al. Development of a novel prohealing stent designed to deliver sirolimus from a biodegradable abluminal matrix. Circ. Cardiovasc. Interv. 3, 257–266 (2010).
Woudstra, P. et al. 1-year results of the REMEDEE registry: clinical outcomes after deployment of the abluminal sirolimus-coated bioengineered (Combo) stent in a multicenter, prospective all-comers registry. JACC Cardiovasc. Interv. 9, 1127–1134 (2016).
Jaguszewski, M. et al. The REMEDEE-OCT study: an evaluation of the bioengineered COMBO dual-therapy CD34 antibody-covered sirolimus-eluting coronary stent compared with a cobalt-chromium everolimus-eluting stent in patients with acute coronary syndromes: insights from optical coherence tomography imaging analysis. JACC Cardiovasc. Interv. 10, 489–499 (2017).
Haude, M. et al. The REMEDEE trial: a randomized comparison of a combination sirolimus-eluting endothelial progenitor cell capture stent with a paclitaxel-eluting stent. JACC Cardiovasc. Interv. 6, 334–343 (2013).
Huang, Y., Ng, H. C. A., Ng, X. W. & Subbu, V. Drug-eluting biostable and erodible stents. J. Controlled Rel. 193, 188–201 (2014).
Perkins, L. E., Boeke-Purkis, K. H., Wang, Q., Stringer, S. K. & Coleman, L. A. XIENCE V™ everolimus-eluting coronary stent system: a preclinical assessment. J. Intervent. Cardiol. 22, S28–S40 (2009).
Serruys, P. W. et al. A randomized comparison of a durable polymer everolimus-eluting stent with a bare metal coronary stent: the SPIRIT first trial. EuroIntervention 1, 58–65 (2005).
Serruys, P. W. et al. A randomised comparison of an everolimus-eluting coronary stent with a paclitaxel-eluting coronary stent: the SPIRIT II trial. EuroIntervention 2, 286–294 (2006).
Gasior, P. et al. Comparison of overexpansion capabilities and thrombogenicity at the side branch ostia after implantation of four different drug eluting stents. Sci. Rep. 10, 20791 (2020).
Ormiston, J. A. et al. Six-month results of the NEVO Res-Elution I (NEVO RES-I) trial: a randomized, multicenter comparison of the NEVO sirolimus-eluting coronary stent with the TAXUS Liberte paclitaxel-eluting stent in de novo native coronary artery lesions. Circ. Cardiovasc. Interv. 3, 556–564 (2010).
Abizaid, A., Costa, J. R. Jr & Feres, F. First nine-month complete invasive assessment (angiography, IVUS, and OCT) of the novel NEVO™ sirolimus-eluting stent with biodegradable polymer. Catheter. Cardiovasc. Interv. 77, 49–51 (2011).
Shiratori, Y. et al. One-year head to head comparison of the neointimal response between sirolimus eluting stent with reservoir technology and everolimus eluting stent: an optical coherence tomography study. Catheter. Cardiovasc. Interv. 82, E428–E436 (2013).
Falotico, R. et al. NEVO™: a new generation of sirolimus-eluting coronary stent. EuroIntervention 5, F88–F93 (2009).
Fajadet, J. et al. Randomized, double-blind, multicenter study of the Endeavor zotarolimus-eluting phosphorylcholine-encapsulated stent for treatment of native coronary artery lesions: clinical and angiographic results of the ENDEAVOR II trial. Circulation 114, 798–806 (2006).
Meredith, I. T. et al. First-in-human study of the Endeavor ABT-578-eluting phosphorylcholine-encapsulated stent system in de novo native coronary artery lesions: Endeavor I Trial. EuroIntervention 1, 157–164 (2005).
Kandzari, D. E. et al. Comparison of zotarolimus-eluting and sirolimus-eluting stents in patients with native coronary artery disease: a randomized controlled trial. J. Am. Coll. Cardiol. 48, 2440–2447 (2006).
Basalus, M. et al. TWENTE study: the real-world endeavor resolute versus xience V drug-eluting stent study in twente: study design, rationale and objectives. Neth. Heart J. 18, 360–364 (2010).
Price, M. J. et al. Safety and efficacy of the next generation resolute onyx zotarolimus-eluting stent: primary outcome of the RESOLUTE ONYX core trial. Catheter. Cardiovasc. Interv. 92, 253–259 (2018).
Tam, C. C. et al. One-year clinical outcomes of patients implanted with a Resolute Onyx™ zotarolimus-eluting stent. J. Int. Med. Res. 46, 457–463 (2018).
Kedhi, E. et al. Rationale and design of the Onyx ONE global randomized trial: a randomized controlled trial of high-bleeding risk patients after stent placement with 1 month of dual antiplatelet therapy. Am. Heart J. 214, 134–141 (2019).
Savvoulidis, P., Perlman, G. & Bagur, R. The EluNIRTM ridaforolimus eluting coronary stent system. Expert Rev. Med. Devices 16, 71–76 (2019).
Kandzari, D. E. et al. Randomized comparison of ridaforolimus-and zotarolimus-eluting coronary stents in patients with coronary artery disease: primary results from the BIONICS Trial (BioNIR Ridaforolimus-Eluting Coronary Stent System in Coronary Stenosis). Circulation 136, 1304–1314 (2017).
Paradies, V. et al. A prospective randomised trial comparing the novel ridaforolimus-eluting BioNIR stent to the zotarolimus-eluting Resolute stent: six-month angiographic and one-year clinical results of the NIREUS trial. EuroIntervention 14, 86–93 (2018).
Jinnouchi, H. et al. TCT-734 Comparison of thromboresistance between everolimus-eluting fluoropolymer stent and ELuNIR ridaforolimus eluting coronary stentRidaforolimus-eluting stent in an ex vivo swine shunt model. J. Am. Coll. Cardiol. 72, B294 (2018).
Serruys, P. W. et al. A randomised comparison of novolimus-eluting and zotarolimus-eluting coronary stents: 9-month follow-up results of the EXCELLA II study. EuroIntervention 6, 195–205 (2010).
Costa, J. R. Jr et al. EXCELLA first-in-man (FIM) study: safety and efficacy of novolimus-eluting stent in de novo coronary lesions. EuroIntervention 4, 53–58 (2008).
Iqbal, J. et al. DESyne novolimus-eluting coronary stent is superior to Endeavor zotarolimus-eluting coronary stent at five-year follow-up: final results of the multicentre EXCELLA II randomised controlled trial. EuroIntervention 12, e1336–e1342 (2016).
Garg, S. & Serruys, P. W. Coronary stents: looking forward. J. Am. Coll. Cardiol. 56, S43–S78 (2010).
Shetty, R., Prajapati, J., Pai, U. & Shetty, K. Preliminary evaluation of clinical and angiographic outcomes with biodegradable polymer coated sirolimus-eluting stent in de novo coronary artery disease: results of the MANIPAL-FLEX study. Scientifica 2016, 9324279 (2016).
Lemos, P. A. et al. Clinical outcomes in 995 unselected real-world patients treated with an ultrathin biodegradable polymer-coated sirolimus-eluting stent: 12-month results from the FLEX Registry. BMJ Open 6, e010028 (2016).
Jiménez, V. A. et al. A randomized comparison of novel bioresorbable polymer sirolimus-eluting stent and durable polymer everolimus-eluting stent in patients with acute coronary syndromes: the CENTURY II high risk ACS substudy. Cardiovasc. Revasc. Med. 17, 355–361 (2016).
Stojkovic, S. et al. Reduced sirolimus systemic exposure and improved bioresorbable polymer properties: new allies for the treatment of patients with coronary artery disease. Fundamental Clin. Pharmacol. 29, 95–105 (2015).
Ribeiro, E. E. et al. First-in-man randomised comparison of a novel sirolimus-eluting stent with abluminal biodegradable polymer and thin-strut cobalt-chromium alloy: INSPIRON-I trial. EuroIntervention 9, 1380–1384 (2014).
Oliveira, M. D. P. et al. Four-year clinical follow-up of the first-in-man randomized comparison of a novel sirolimus eluting stent with abluminal biodegradable polymer and ultra-thin strut cobalt-chromium alloy: the INSPIRON-I trial. Cardiovasc. Diag. Ther. 5, 264 (2015).
Stone, G. W. et al. A prospective, randomized evaluation of a novel everolimus-eluting coronary stent: the PLATINUM (a Prospective, Randomized, Multicenter Trial to Assess an Everolimus-Eluting Coronary Stent System [PROMUS Element] for the Treatment of Up to Two de Novo Coronary Artery Lesions) trial. J. Am. Coll. Cardiol. 57, 1700–1708 (2011).
Kelly, C. R. et al. Long-term safety and efficacy of platinum chromium everolimus-eluting stents in coronary artery disease: 5-year results from the PLATINUM trial. JACC Cardiovasc. Interv. 10, 2392–2400 (2017).
Jobe, R. et al. Clinical outcomes to 4 years in patients implanted with bioabsorbable polymer SYNERGY versus permanent polymer everolimus-eluting stents: the EVOLVE II randomized trial. J. Am. Coll. Cardiol. 71, A1018 (2018).
Kereiakes, D. J. et al. Propensity-matched patient-level comparison of the TAXUS Liberté and TAXUS element (ION) paclitaxel-eluting stents. Am. J. Cardiol. 108, 828–837 (2011).
Kereiakes, D. J. et al. Long-term follow-up of the platinum chromium TAXUS element (ION) stent: The PERSEUS Workhorse and Small Vessel trial five-year results. Catheter. Cardiovasc. Interv. 86, 994–1001 (2015).
Mariano, E., Sangiorgi, G. M. & Fioranelli, M. in Imaging Coronary Arteries 101–113 (Springer, 2013).
Grube, E. et al. Six-month clinical and angiographic results of a dedicated drug-eluting stent for the treatment of coronary bifurcation narrowings. Am. J. Cardiol. 99, 1691–1697 (2007).
Bennett, J. et al. 5-year clinical follow-up of the COBRA (complex coronary bifurcation lesions: randomized comparison of a strategy using a dedicated self-expanding biolimus A9-eluting stent vs. a culotte strategy using everolimus-eluting stents) study. Catheter. Cardiovasc. Interv. 92, E375–E380 (2018).
Wilson, W. M. & Cruden, N. L. Advances in coronary stent technology: current expectations and new developments. Res. Rep. Clin. Cardiol. 2013, 85–96 (2013).
Müller-Hülsbeck, S., Keirse, K., Zeller, T., Schroë, H. & Diaz-Cartelle, J. Twelve-month results from the MAJESTIC trial of the Eluvia paclitaxel-eluting stent for treatment of obstructive femoropopliteal disease. J. Endovasc. Ther. 23, 701–707 (2016).
Müller-Hülsbeck, S., Keirse, K., Zeller, T., Schroë, H. & Diaz-Cartelle, J. Long-term results from the MAJESTIC trial of the Eluvia paclitaxel-eluting stent for femoropopliteal treatment: 3-year follow-up. Cardiovasc. Intervent. Radiol. 40, 1832–1838 (2017).
Gray, W. A. et al. A polymer-coated, paclitaxel-eluting stent (Eluvia) versus a polymer-free, paclitaxel-coated stent (Zilver PTX) for endovascular femoropopliteal intervention (IMPERIAL): a randomised, non-inferiority trial. Lancet 392, 1541–1551 (2018).
Müller-Hülsbeck, S. et al. Eluvia drug-eluting vascular stent system for the treatment of symptomatic femoropopliteal lesions. Future Cardiol. 14, 207–213 (2018).
Patted, S. V. et al. Clinical outcomes of novel long-tapered sirolimus-eluting coronary stent system in real-world patients with long diffused de novo coronary lesions. Cardiol. Res. 9, 350 (2018).
Dani, S. et al. First-in-human evaluation of the novel BioMime sirolimus-eluting coronary stent with bioabsorbable polymer for the treatment of single de novo lesions located in native coronary vessels — results from the meriT-1 trial. EuroIntervention 9, 493–500 (2013).
Seth, A. et al. Late angiographic and clinical outcomes of the novel BioMime™ sirolimus-eluting coronary stent with ultra-thin cobalt–chromium platform and biodegradable polymer for the treatment of diseased coronary vessels: results from the prospective, multicentre meriT-2 clinical trial. AsiaIntervention 2, 19–27 (2016).
Jain, R. K. et al. One-year outcomes of a BioMime™ Sirolimus-Eluting Coronary Stent System with a biodegradable polymer in all-comers coronary artery disease patients: The meriT-3 study. Indian Heart J. 68, 599–603 (2016).
Tijssen, R. Y. et al. Evaluation of the MiStent sustained sirolimus eluting biodegradable polymer coated stent for the treatment of coronary artery disease: does uniform sustained abluminal drug release result in earlier strut coverage and better safety profile? Expert Rev. Med. Devices 14, 325–334 (2017).
Ormiston, J. et al. First-in-human evaluation of a bioabsorbable polymer-coated sirolimus-eluting stent: imaging and clinical results of the DESSOLVE I trial (DES with Sirolimus and a Bioabsorbable Polymer for the Treatment of Patients with De Novo Lesion in the Native Coronary Arteries). JACC Cardiovasc. Interv. 6, 1026–1034 (2013).
Wijns, W. et al. Randomised study of a bioabsorbable polymer-coated sirolimus-eluting stent: results of the DESSOLVE II trial. EuroIntervention 10, 1383–1390 (2015).
Lansky, A. J. et al. Comparison of the absorbable polymer sirolimus-eluting stent (MiStent) to the durable polymer everolimus-eluting stent (Xience) (from the DESSOLVE I/II and ISAR-TEST-4 studies). Am. J. Cardiol. 117, 532–538 (2016).
Wijns, W. et al. Evaluation of a crystalline sirolimus-eluting coronary stent with a bioabsorbable polymer designed for rapid dissolution: two-year outcomes from the DESSOLVE I and II trials. EuroIntervention 11, 352–355 (2015).
Hiremath, M. et al. Real world experience of GenXSync™ sirolimus eluting coronary stent system in patients with long coronary lesions: outcome of the GEL registry. Int. J. Adv. Med. 3, 1029 (2016).
Fisher, L., Mathew, A., Punnose, E., Indani, A. & Bhutada, P. Safety and efficacy of hybrid platform design sirolimus eluting stent system in percutaneous coronary intervention in ST elevation myocardial infarction patients at 1 year after treatment. Int. J. Res. Med. Sci. 4, 4458 (2016).
Peeters, P., Bosiers, M., Verbist, J., Deloose, K. & Heublein, B. Preliminary results after application of absorbable metal stents in patients with critical limb ischemia. J. Endovasc. Ther. 12, 1–5 (2005).
Bosiers, M., Deloose, K., Verbist, J. & Peeters, P. First clinical application of absorbable metal stents in the treatment of critical limb ischemia: 12-month results. Vasc. Dis. Manag. 2, 86–91 (2005).
Bosiers, M. et al. AMS INSIGHT — absorbable metal stent implantation for treatment of below-the-knee critical limb ischemia: 6-month analysis. Cardiovasc. Intervent. Radiol. 32, 424–435 (2009).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT00572494 (2008).
Erbel, R. et al. Temporary scaffolding of coronary arteries with bioabsorbable magnesium stents: a prospective, non-randomised multicentre trial. Lancet 369, 1869–1875 (2007).
Erbel, R. et al. Absorbable coronary stents. New promising technology? [German] Herz 32, 308–319 (2007).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT01610102 (2015).
Haude, M. et al. Safety and performance of the drug-eluting absorbable metal scaffold (DREAMS) in patients with de-novo coronary lesions: 12 month results of the prospective, multicentre, first-in-man BIOSOLVE-I trial. Lancet 381, 836–844 (2013).
Haude, M. et al. Safety and performance of the DRug-Eluting Absorbable Metal Scaffold (DREAMS) in patients with de novo coronary lesions: 3-year results of the prospective, multicentre, first-in-man BIOSOLVE-I trial. EuroIntervention 12, e160–e166 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT01168830 (2015).
Haude, M. et al. Safety and performance of the second-generation drug-eluting absorbable metal scaffold in patients with de-novo coronary artery lesions (BIOSOLVE-II): 6 month results of a prospective, multicentre, non-randomised, first-in-man trial. Lancet 387, 31–39 (2016).
Haude, M. et al. Sustained safety and performance of the second-generation drug-eluting absorbable metal scaffold in patients with de novo coronary lesions: 12-month clinical results and angiographic findings of the BIOSOLVE-II first-in-man trial. Eur. Heart J. 37, 2701–2709 (2016).
Garcia-Garcia, H. M. et al. In vivo serial invasive imaging of the second-generation drug-eluting absorbable metal scaffold (Magmaris—DREAMS 2G) in de novo coronary lesions: insights from the BIOSOLVE-II first-in-man trial. Int. J. Cardiol. 255, 22–28 (2018).
Haude, M. et al. Safety and performance of the second-generation drug-eluting absorbable metal scaffold (DREAMS 2G) in patients with de novo coronary lesions: three-year clinical results and angiographic findings of the BIOSOLVE-II first-in-man trial. EuroIntervention 15, e1375–e1382 (2020).
Haude, M. et al. Safety and clinical performance of a drug eluting absorbable metal scaffold in the treatment of subjects with de novo lesions in native coronary arteries: pooled 12-month outcomes of BIOSOLVE-II and BIOSOLVE-III. Catheter. Cardiovasc. Interv. 92, E502–E511 (2018).
Haude, M. et al. Sustained safety and clinical performance of a drug-eluting absorbable metal scaffold up to 24 months: pooled outcomes of BIOSOLVE-II and BIOSOLVE-III. EuroIntervention 13, 432–439 (2017).
Verheye, S. et al. BIOSOLVE-IV-registry: safety and performance of the Magmaris scaffold: 12-month outcomes of the first cohort of 1,075 patients. Catheter. Cardiovasc. Interv. 98, E1–E8 (2021).
Capodanno, D. Bioresorbable scaffolds in coronary intervention: unmet needs and evolution. Korean Circ. J. 48, 24–35 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT01960504 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT02716220 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT02817802 (2023).
McLennan, D. I. et al. Absorbable metal stents for vascular use in pediatric cardiology: progress and outlook. Front. Cardiovasc. Med. 11, 1410305 (2024).
Sabate, M. et al. Magnesium-based resorbable scaffold versus permanent metallic sirolimus-eluting stent in patients with ST-segment elevation myocardial infarction: the MAGSTEMI randomized clinical trial. Circulation 140, 1904–1916 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT03234348 (2020).
Kereiakes, D. J. et al. 3-year clinical outcomes with everolimus-eluting bioresorbable coronary scaffolds: the ABSORB III trial. J. Am. Coll. Cardiol. 70, 2852–2862 (2017).
Diletti, R. et al. 6-month clinical outcomes following implantation of the bioresorbable everolimus-eluting vascular scaffold in vessels smaller or larger than 2.5 mm. J. Am. Coll. Cardiol. 58, 258–264 (2011).
Bosiers, M., Scheinert, D., Simonton, C. A. & Schwartz, L. B. Coronary and endovascular applications of the Absorb bioresorbable vascular scaffold. Interv. Cardiol. 4, 621 (2012).
Werner, M. et al. Evaluation of the biodegradable peripheral Igaki-Tamai stent in the treatment of de novo lesions in the superficial femoral artery: the GAIA study. JACC Cardiovasc. Interv. 7, 305–312 (2014).
Kuwabara, K. et al. Comparative analysis of the paclitaxel-eluting peripheral Igaki-Tamai stent and the drug-free Igaki-Tamai stent using optical coherence tomography and histological analysis in a porcine iliac artery model. Circ. J. 84, 799–805 (2020).
Cheng, Y. et al. Comparative characterization of biomechanical behavior and healing profile of a novel ultra-high-molecular-weight amorphous poly-l-lactic acid sirolimus-eluting bioresorbable coronary scaffold. Circ. Cardiovasc. Interv. 9, e004253 (2016).
McMahon, S. et al. Bio-resorbable polymer stents: a review of material progress and prospects. Prog. Polym. Sci. 83, 79–96 (2018).
Jeong, G.-W., Kim, T.-H., Nah, J.-W. & Park, J.-K. The developing trend in bioresorbable stent for treatment of coronary artery disease. Appl. Chem. Eng. 29, 497–502 (2018).
Boeder, N. F. et al. Effect of plaque composition, morphology, and burden on DESolve novolimus-eluting bioresorbable vascular scaffold expansion and eccentricity — an optical coherence tomography analysis. Cardiovasc. Revasc. Med. 20, 480–484 (2019).
Verheye, S. et al. A next-generation bioresorbable coronary scaffold system: from bench to first clinical evaluation: 6-and 12-month clinical and multimodality imaging results. JACC Cardiovasc. Interv. 7, 89–99 (2014).
Wiebe, J. et al. Everolimus-versus novolimus-eluting bioresorbable scaffolds for the treatment of coronary artery disease: a matched comparison. JACC Cardiovasc. Interv. 10, 477–485 (2017).
Nef, H. M. et al. A new novolimus-eluting bioresorbable coronary scaffold: present status and future clinical perspectives. Int. J. Cardiol. 227, 127–133 (2017).
Durand, E. et al. Head-to-head comparison of a drug-free early programmed dismantling polylactic acid bioresorbable scaffold and a metallic stent in the porcine coronary artery: six-month angiography and optical coherence tomographic follow-up study. Circ. Cardiovasc. Interv. 7, 70–79 (2014).
Yahagi, K. et al. Comparison of a drug-free early programmed dismantling PDLLA bioresorbable scaffold and a metallic stent in a porcine coronary artery model at 3-year follow-up. J. Am. Heart Assoc. 6, e005693 (2017).
Tenekecioglu, E. et al. Hemodynamic analysis of a novel bioresorbable scaffold in porcine coronary artery model. Catheter. Cardiovasc. Interv. 91, 1084–1091 (2018).
Costa, R. A. et al. TCT-546 6-Month angiographic results of the novel MIRAGE microfiber sirolimus-eluting bioresorbable vascular scaffold — a quantitative coronary angiography analysis from the prospective, randomized mirage clinical trial. J. Am. Coll. Cardiol. 66, B223 (2015).
Tenekecioglu, E. et al. Randomized comparison of absorb bioresorbable vascular scaffold and mirage microfiber sirolimus-eluting scaffold using multimodality imaging. JACC Cardiovasc. Interv. 10, 1115–1130 (2017).
Wu, Y. et al. Six-month outcomes of the X INSORB bioresorbable sirolimus-eluting scaffold in treating single de novo lesions in human coronary artery. Catheter. Cardiovasc. Interv. 87, 630–637 (2016).
Lv, X. et al. Healing score of the Xinsorb scaffold in the treatment of de novo lesions: 6-month imaging outcomes. Int. J. Cardiovasc. Imaging 34, 1009–1016 (2018).
Tenekecioglu, E. et al. Bioresorbable scaffolds: a new paradigm in percutaneous coronary intervention. BMC Cardiovasc. Disord. 16, 38 (2016).
Gasior, P. et al. Two-year longitudinal evaluation of a second-generation thin-strut sirolimus-eluting bioresorbable coronary scaffold with hybrid cell design in porcine coronary arteries. Cardiol. J. 27, 115–125 (2020).
Seth, A. et al. Three-year clinical and two-year multimodality imaging outcomes of a thin-strut sirolimus-eluting bioresorbable vascular scaffold: MeRes-1 trial. EuroIntervention 15, 607–614 (2019).
Abizaid, A. et al. TCT-39 12-Month angiographic and clinical results of the ReZolve sirolimus-eluting bioresorbable coronary scaffold: The RESTORE trial. J. Am. Coll. Cardiol. 62, B13 (2013).
Azzalini, L., L’Allier, P. L. & Tanguay, J.-F. Bioresorbable scaffolds: the revolution in coronary stenting? AIMS Med. Sci. 3, 126–146 (2016).
Simonsen, J. K. et al. Mechanical performance and healing patterns of the novel sirolimus-eluting bioresorbable Fantom scaffold: 6-month and 9-month follow-up by optical coherence tomography in the FANTOM II study. Open Heart 6, e000941 (2019).
Chevalier, B. et al. Clinical and angiographic outcomes with a novel radiopaque sirolimus-eluting bioresorbable vascular scaffold: The FANTOM II study. Circ. Cardiovasc. Interv. 12, e007283 (2019).
Dokollari, A. et al. Benefits and pitfalls of the Perceval sutureless bioprosthesis. Front. Cardiovasc. Med. 8, 789392 (2022).
Szecel, D., Eurlings, R., Rega, F., Verbrugghe, P. & Meuris, B. Perceval sutureless aortic valve implantation: midterm outcomes. Ann. Thorac. Surg. 111, 1331–1337 (2021).
Glauber, M. et al. International expert consensus on sutureless and rapid deployment valves in aortic valve replacement using minimally invasive approaches. Innovations 11, 165–173 (2016).
Okuno, T., Lanz, J. & Pilgrim, T. ACURATE neo: how is this TAVR valve doing to fit into an increasingly crowded field? Curr. Cardiol. Rep. 22, 107 (2020).
Holzamer, A. et al. Valve-in-valve implantation using the ACURATE neo in degenerated aortic bioprostheses: an international multicenter analysis. Cardiovasc. Interv. 12, 2309–2316 (2019).
Schneeberger, Y. et al. TAVI for pure non-calcified aortic regurgitation using a self-expandable transcatheter heart valve. Front. Cardiovascular Med. 8, 743579 (2022).
Seigerman, M. E., Nathan, A. & Anwaruddin, S. The lotus valve system: an in-depth review of the technology. Curr. Cardiol. Rep. 21, 157 (2019).
Rizik, D. G. et al. Long-term outcomes of transcatheter aortic valve replacement with the LOTUS Valve vs CoreValve/EvolutR: a secondary analysis of the REPRISE III randomized clinical trial. JAMA Netw. Open 5, e2238792 (2022).
Solomonica, A., Choudhury, T. & Bagur, R. The mechanically expandable LOTUS Valve and LOTUS Edge transcatheter aortic valve systems. Expert Rev. Med. Devices 15, 763–769 (2018).
Poschner, T. et al. The JenaValve pericardial transcatheter aortic valve replacement system to treat aortic valve disease. Future Cardiol. 18, 101–113 (2021).
Figulla, H. R., Ferrari, M., Carr-Brendel, V. & Lauten, A. in Transcatheter Aortic Valve Implantation: Clinical, Interventional and Surgical Perspectives (eds Giordano, A., Biondi-Zoccai, G. & Frati, G.) 279–286 (Springer, 2019).
Zilla, P., Williams, D. F. & Bezuidenhout, D. TAVR for patients with rheumatic heart disease: opening the door for the many? J. Am. Coll. Cardiol. 77, 1714–1716 (2021).
Jones, T. K. et al. Long-term outcomes after melody transcatheter pulmonary valve replacement in the US investigational device exemption trial. Circ. Cardiovasc. Interv. 15, e010852 (2022).
Houeijeh, A. et al. Long-term outcomes of transcatheter pulmonary valve implantation with melody and SAPIEN valves. Int. J. Cardiol. 370, 156–166 (2023).
Holoshitz, N. & Hijazi, Z. M. Transcatheter pulmonary valve replacement: valves, techniques of implantation and outcomes. Interv. Cardiol. 5, 465 (2013).
Dreger, H. et al. Treatment of severe TRIcuspid regurgitation in patients with advanced heart failure with CAval vein implantation of the Edwards Sapien XT VALve (TRICAVAL): a randomised controlled trial. EuroIntervention 15, 1506–1513 (2020).
Güzeltaş, A. et al. Implantation of the Edwards SAPIEN XT and SAPIEN 3 valves for pulmonary position in enlarged native right ventricular outflow tract. Anatol. J. Cardiol. 25, 96 (2021).
Agrawal, A. Transcatheter heart valves—an update on types of valves available, hardware characteristics, and patient selection: an Indian perspective. Indian J. Clin. Cardiol. 5, 139–149 (2024).
Kodali, S. K. et al. Early safety and feasibility of a first-in-class biomimetic transcatheter aortic valve-DurAVR. EuroIntervention 19, e352 (2023).
Sathananthan, J. et al. TCT-374 Redo transcatheter aortic valve replacement with the novel DurAVR valve: insights from the bench. J. Am. Coll. Cardiol. 82, B149–B150 (2023).
Meduri, C. et al. OR7-3 DurAVR biomimetic transcatheter heart valve: first-in-human study results update. J. Soc. Cardiovasc. Angio. Intervent. 3, 101443 (2024).
Buszman, P. P. et al. Long-term evaluation of biocompatibility and endurance of a novel, balloon expandable transcatheter polymeric aortic valve in the ovine aortic banding model. J. Am. Coll. Cardiol. 75, 1350 (2020).
Kachel, M. et al. Temporal, biomechanical evaluation of a novel, transcatheter polymeric aortic valve in ovine aortic banding model. Front. Cardiovasc. Med. 9, 977006 (2022).
Pinchuk, L., Boden, M. & Bluestein, D. in Macromolecular Engineering (eds Lubnin, A. & Erdodi, G.) 211–235 (Elsevier, 2021).
Bui, H. T., Ishrat, A., James, S. P. & Dasi, L. P. Design consideration of a novel polymeric transcatheter heart valve through computational modeling. J. Mech. Behav. Biomed. Mater. 135, 105434 (2022).
Todesco, M., Lezziero, G., Gerosa, G. & Bagno, A. Polymeric heart valves: do they represent a reliable alternative to current prosthetic devices? Polymers 17, 557 (2025).
Scherman, J. & Zilla, P. Poorly suited heart valve prostheses heighten the plight of patients with rheumatic heart disease. Int. J. Cardiol. 318, 104–114 (2020).
Wang, Q. et al. In-vivo assessment of a novel polymer (SIBS) trileaflet heart valve. J. Heart Valve Dis. 19, 499–505 (2010).
Appa, H. et al. The technological basis of a balloon-expandable TAVR system: non-occlusive deployment, anchorage in the absence of calcification and polymer leaflets. Front. Cardiovasc. Med. 9, 791949 (2022).
Rahmani, B. et al. In vitro hydrodynamic assessment of a new transcatheter heart valve concept (the TRISKELE). J. Cardiovasc. Transl. Res. 10, 104–115 (2017).
Patanè, M., la Spina, K. & La Manna, A. in Percutaneous Treatment of Left Side Cardiac Valves: A Practical Guide for the Interventional Cardiologist (eds Tamburino, C., Barbanti, M. & Capodanno, D.) 443–452 (Springer, 2018).
Jenney, C. et al. Assessment of a siloxane poly (urethane-urea) elastomer designed for implantable heart valve leaflets. Adv. NanoBiomed Res. 1, 2000032 (2021).
Yakubov, S. J., Wittel, J. & Johnson, G. CRT-700.20 Foldax TRIA TAVI: a novel-polymer transcatheter aortic valve: pilot chronic ovine model study. Cardiovasc. Interv. 15, S59–S60 (2022).
Kereiakes, D. J. et al. Preliminary evaluation of a novel polymeric valve following surgical implantation for symptomatic aortic valve disease. Cardiovasc. Interv. 14, 2754–2756 (2021).
Elkoumy, A., Mylotte, D., Elzomor, H., McInerney, A. & Soliman, O. Emerging transcatheter heart valve technologies for severe aortic stenosis. Expert Rev. Med. Devices 20, 1065–1077 (2023).
Ge, J. et al. Preliminary Implantation of a novel TAVR device with polymeric leaflets for symptomatic calcific aortic disease. Case Rep. 17, 101901 (2023).
Wang, Y. et al. Recent advancements in polymeric heart valves: from basic research to clinical trials. Mater. Today Bio 28, 101194 (2024).
Bink, N., Mohan, V. B. & Fakirov, S. Recent advances in plastic stents: a comprehensive review. Int. J. Polymeric Mater. Polymeric Biomater. 70, 54–74 (2021).
Liu, Q., Li, Q., Xu, S., Zheng, Q. & Cao, X. Preparation and properties of 3D printed alginate–chitosan polyion complex hydrogels for tissue engineering. Polymers 10, 664 (2018).
Liu, S.-J., Chiang, F.-J., Hsiao, C.-Y., Kau, Y.-C. & Liu, K.-S. Fabrication of balloon-expandable self-lock drug-eluting polycaprolactone stents using micro-injection molding and spray coating techniques. Ann. Biomed. Eng. 38, 3185–3194 (2010).
Lee, C.-H., Chen, C.-J., Liu, S.-J., Hsiao, C.-Y. & Chen, J.-K. The development of novel biodegradable bifurcation stents for the sustainable release of anti-proliferative sirolimus. Ann. Biomed. Eng. 40, 1961–1970 (2012).
Jiang, W., Zhao, W., Zhou, T., Wang, L. & Qiu, T. A review on manufacturing and post-processing technology of vascular stents. Micromachines 13, 140 (2022).
Ramzipoor, K., Alfred, N., Wang, L. & Lee, C. Y. Stent fabrication via tubular casting processes. US Patent 2017/0157806 A1 (2017).
Martinez, A. W. & Chaikof, E. L. Microfabrication and nanotechnology in stent design. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 3, 256–268 (2011).
Kelly, N. et al. Comparison of computational modelling techniques for braided stent analysis. Computer Methods Biomech. Biomed. Eng. 22, 1334–1344 (2019).
Venkatraman, S., Boey, F. & Lao, L. L. Implanted cardiovascular polymers: natural, synthetic and bio-inspired. Prog. Polym. Sci. 33, 853–874 (2008).
Guidoin, R. et al. in Biotextiles as Medical Implants (eds King, M. W., Gupta, B. S. & Guidoin, R.) 434–484 (Woodhead Publishing, 2013).
Jiang, C., Wang, K., Liu, Y., Zhang, C. & Wang, B. Application of textile technology in tissue engineering: a review. Acta Biomaterialia 128, 60–76 (2021).
Khoo, Z. X. et al. 3D printing of smart materials: a review on recent progresses in 4D printing. Virtual Phys. Prototyp. 10, 103–122 (2015).
Giannopoulos, A. A. et al. Applications of 3D printing in cardiovascular diseases. Nat. Rev. Cardiol. 13, 701–718 (2016).
Zhao, D., Zhou, R., Sun, J., Li, H. & Jin, Y. Experimental study of polymeric stent fabrication using homemade 3D printing system. Polym. Eng. Sci. 59, 1122–1131 (2019).
Demir, A. G. & Previtali, B. Additive manufacturing of cardiovascular CoCr stents by selective laser melting. Mater. Des. 119, 338–350 (2017).
Finazzi, V. et al. Design rules for producing cardiovascular stents by selective laser melting: Geometrical constraints and opportunities. Procedia Struct. Integr. 15, 16–23 (2019).
Omar, M. A., Baharudin, B., Sulaiman, S., Ismail, M. & Omar, M. A. Characterisation of powder and microstructure, density and surface roughness for additively manufactured stent using medical grade ASTM F75 cobalt chromium (CoCrMo) by selective laser melting (SLM) technology. Adv. Mater. Process. Technol. 8, 1655–1666 (2022).
Langi, E. et al. A comparative study of microstructures and nanomechanical properties of additively manufactured and commercial metallic stents. Mater. Today Commun. 31, 103372 (2022).
Chen, K., Wan, H., Fang, X. & Chen, H. Laser additive manufacturing of anti-tetrachiral endovascular stents with negative Poisson’s ratio and favorable cytocompatibility. Micromachines 13, 1135 (2022).
Yan, L. et al. Evaluation and characterization of nitinol stents produced by selective laser melting with various process parameters. Prog. Addit. Manuf. 7, 1141–1153 (2022).
Finazzi, V. et al. Patient-specific cardiovascular superelastic NiTi stents produced by laser powder bed fusion. Procedia CIRP 110, 242–246 (2022).
Jamshidi, P. et al. Development, characterisation, and modelling of processability of nitinol stents using laser powder bed fusion. J. Alloy. Compd. 909, 164681 (2022).
Paul, B. et al. Cell–material interactions in direct contact culture of endothelial cells on biodegradable iron-based stents fabricated by laser powder bed fusion and impact of ion release. ACS Appl. Mater. Interfaces 14, 439–451 (2021).
Wang, Q. et al. Impact of inverse unequal height strut structure on the functional performance of an additively manufactured cardiovascular stent. J. Mech. Behav. Biomed. Mater. 146, 106058 (2023).
Flege, C. et al. Development and characterization of a coronary polylactic acid stent prototype generated by selective laser melting. J. Mater. Science: Mater. Med. 24, 241–255 (2013).
Geng, L., Ruan, X., Wu, W., Xia, R. & Fang, D. Mechanical properties of selective laser sintering (SLS) additive manufactured chiral auxetic cylindrical stent. Exp. Mech. 59, 913–925 (2019).
Jia, H., Gu, S. Y. & Chang, K. 3D printed self-expandable vascular stents from biodegradable shape memory polymer. Adv. Polym. Technol. 37, 3222–3228 (2018).
Wu, Z. et al. Radial compressive property and the proof-of-concept study for realizing self-expansion of 3D printing polylactic acid vascular stents with negative Poisson’s ratio structure. Materials 11, 1357 (2018).
Park, S. A. et al. In vivo evaluation and characterization of a bio-absorbable drug-coated stent fabricated using a 3D-printing system. Mater. Lett. 141, 355–358 (2015).
Guerra, A. J. & Ciurana, J. 3D-printed bioabsordable polycaprolactone stent: the effect of process parameters on its physical features. Mater. Des. 137, 430–437 (2018).
Wang, C., Zhang, L., Fang, Y. & Sun, W. Design, characterization, and 3D printing of cardiovascular stents with zero Poisson’s ratio in longitudinal deformation. Engineering 7, 979–990 (2020).
Somszor, K. et al. Personalized, mechanically strong, and biodegradable coronary artery stents via melt electrowriting. ACS Macro Lett. 9, 1732–1739 (2020).
Shen, Y. et al. 3D printed personalized, heparinized and biodegradable coronary artery stents for rabbit abdominal aorta implantation. Chem. Eng. J. 450, 138202 (2022).
Ni, N. et al. 3D printed peripheral vascular stents based on degradable poly(trimethylene carbonate-b-(L-lactide-ran-glycolide)) terpolymer. Polym. Adv. Technol. 34, 1739–1751 (2023).
Chausse, V. et al. Solvent-cast direct-writing as a fabrication strategy for radiopaque stents. Addit. Manuf. 48, 102392 (2021).
Van Lith, R. et al. 3D-printing strong high-resolution antioxidant bioresorbable vascular stents. Adv. Mater. Technol. 1, 1600138 (2016).
Ware, H. O. T. et al. High-speed on-demand 3D printed bioresorbable vascular scaffolds. Mater. Today Chem. 7, 25–34 (2018).
de Oliveira, M. F., da Silva, L. C. & de Oliveira, M. G. 3D printed bioresorbable nitric oxide-releasing vascular stents. Bioprinting 22, e00137 (2021).
Xiao, R. et al. Direct 3D printing of thin-walled cardiovascular stents with negative Poisson’s ratio (NPR) structure and functional metallic coating. Composite Struct. 306, 116572 (2023).
Condello, F. et al. Stent thrombosis and restenosis with contemporary drug-eluting stents: predictors and current evidence. J. Clin. Med. 12, 1238 (2023).
Acknowledgements
A.E., S.E.M. and M.Y.E. were supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement #852814 TAVI4Life). M.G. was supported by the Mäxi Foundation (grant recipients: S.P.H. and M.Y.E.).
Author information
Authors and Affiliations
Contributions
A.E. and F.B.C. researched data for the manuscript; A.E. and M.Y.E. wrote the manuscript; A.E., M.G., H.D. and M.Y.E. contributed to the discussion of content; and A.E., S.E.M., M.G., H.D., V.F., S.P.H. and M.Y.E. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
S.P.H. is a shareholder at LifeMatrix Technologies and Xeltis BV. M.Y.E. is a shareholder at LifeMatrix Technologies. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Atta Behfar, Giuseppe Biondi-Zoccai, Umberto Morbiducci and Manel Sabaté for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ehterami, A., Motta, S.E., Generali, M. et al. Cardiovascular stent technologies for coronary and valvular heart disease: the potential of 3D printing for stent fabrication. Nat Rev Cardiol (2026). https://doi.org/10.1038/s41569-026-01275-x
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41569-026-01275-x